Abstract
-
Background
- The aim of this study is to evaluate the influence of immunosuppressants on in-hospital mortality from sepsis.
-
Methods
- Using data of the Health Insurance Review & Assessment Service, we collected data from patients who were admitted to the hospital due to sepsis from 2009 to 2013. Based on drugs commonly used for immunosuppression caused by various diseases, patients were divided into three groups; immunosuppressant group, steroid-only group, and control group. Patients with no history of immunosuppressants or steroids were assigned to the control group. To identify risk factors of in-hospital mortality in sepsis, we compared differences in patient characteristics, comorbidities, intensive care unit (ICU) care requirements, and immunodeficiency profiles. Subgroup analysis according to age was also performed.
-
Results
- Of the 185,671 included patients, 13,935 (7.5%) were in the steroid-only group and 2,771 patients (1.5%) were in the immunosuppressant group. The overall in-hospital mortality was 38.9% and showed an increasing trend with age. The steroid-only group showed the lowest in-hospital mortality among the three groups except the patients younger than 30 years. The steroid-only group and immunosuppressant group received ICU treatment more frequently (p < 0.001), stayed longer in the hospital (p < 0.001), and showed higher medical expenditure (p < 0.001) compared to the normal group. Univariate and multivariate analyses revealed that age, male gender, comorbidities (especially malignancy), and ICU treatment had a significant effect on in-hospital mortality.
-
Conclusions
- Despite longer hospital length of stay and more frequent need for ICU care, the in-hospital mortality was lower in patients taking immunosuppressive drugs than in patients not taking immunosuppressive drugs.
-
Keywords: immunosuppression; Korea; mortality; sepsis; steroid
Introduction
Sepsis is a leading cause of complications and death in intensive care settings, and its treatment demands enormous medical resources. The incidence of sepsis is still increasing, and the efforts to improve outcomes for sepsis patients are underway in medical institutions worldwide. Among them are the Surviving Sepsis Campaign and the Global Sepsis Alliance.[1,2] As a result, the mortality associated with sepsis has significantly declined.[1,3] However, the absolute mortality rate is still high, hovering between 25% and 40%.[4-6]
The risk factors of sepsis mortality include old age, chronic comorbidity, immunosuppression, blood-borne pathogens and non-antibiotic therapy.[6-9] In particular, the presence of immunosuppression has steadily increased in sepsis patients over the last two decades, calling for investigations.[7,8] However, such investigations have limitations to conduct because the cause of immunoSuppression varies according to the associated disease and medication. Previous researchers therefore focused on a single disease or medication in their studies concerning immunosuppression in spite of limitations posed by their approach.[10,11]
Authors in this study investigated the incidence of in-hospital mortality and associated risk factors in sepsis patients based on epidemiological data of the two groups of sepsis patients on either steroid or non-steroid suppressive drugs and those not on suppressive drugs.
Materials and Methods
This study involved the analysis of medical information of sepsis patients who were hospitalized from January 2009 to December 2013, according to Health Insurance Review & Assessment Service (HIRA)’s database. Korea’s national health insurance system is mandatory and requires all Korean citizens to join either employee health insurance or self-employed health insurance.[12,13] All insurance claims made by medical institutions are reported to the state-run HIRA which conducts the review of medical costs incurred under the national insurance scheme, making HIRA’s database a very useful data source for medical research.
Based on patient information from the HIRA database, sepsis patients (A40 or A41) aged 18 or older were selected from those who were hospitalized from January 2009 to December 2013. Patients divided into three groups: those on suppression medications within a year before hospital admission (Suppression group) those who had steroids only (Steroid group) and those not on any suppression medication. The immunosupressants, commonly prescribed in Korea, include cyclosporine, tacrolimus, azathioprine, mycophenolate mofetil (mycophenolic acid), sirolimus, everolimus, rapamycin, methotrexate, muronomab-CD3, basiliximab, daclizumab, alemtuzumab and rituximab. These suppressive drugs are divided into either steroids or non-steroids in this study. Also, the reason underlying suppressive drug use was also identified for the Suppression and Steroid groups.
Patients in each group were further divided according to age with 10-year intervals, and the gender of each subgroup was identified to investigate distribution of immunosuppressive state and in-hospital mortality. Based on clinical characteristics of patients, risk factors associated with in-hospital mortality were determined, and involved medical resources and length of hospital stay were analyzed.
Statistical analysis was performed using SAS 9.2 software (SAS insititute Inc, Cary, NC). Pearson chi-square test was used to compare gender distribution and intensive care unit (ICU) admission among the groups. ANOVA test was used to compare hospital costs and length of hospital stay. Results are considered statistically significant when p value is less than 0.05. Multiple logistic regression analysis was performed to identify risk factors associated with mortality.
This study was approved by the Research Ethics Committee of Institutional Review Board of National Evidence-based Healthcare Collaborating Agency (IRB No.: NECAIRB14-018).
Results
A total of 185,671 sepsis patients were admitted to hospital from 2009 to 2013. Of those, 13,935 (7.5%) were on steroids only while 2,771 (1.5%) were on non-steroid suppressive drugs. The mean age of patients was 68.4, and the ratio of male to female was 1.1:1. By comorbidity in a population of patients, diabetes (42.6%) was the most common comorbid condition, followed by hypertension (39.4%). Malignant tumor was also found in 6.5% of patients. By the reason underlying suppressive drug use, leukemia (9.1%) was the most common reason, followed by organ transplantation, rheumatoid arthritis, Crohn’s disease, ulcerative colitis and atopic dermatitis (Table 1). The mean age of the Suppression group was lower at 51.2 years, but its ratio of male to female was higher at 1.3:1, compared with the Steroid and control groups. The distribution of comorbidities also showed differences among the groups: the presence of hypertension, diabetes, cerebrovascular disease was higher in the Steroid group whereas chronic liver and kidney diseases along with malignant tumor were significant in the Suppression group (Table 2).
The analysis of distribution of immunosuppressive state showed 3,800 cases of immunosuppression in the 18-29 age group, 57,905 cases in the 70s age group and 45,281 cases in the 80+ age group from 2009 to 2013. The number of male patients was slightly higher than the number of female patients in all age groups except the 80+ age group (Table 3).
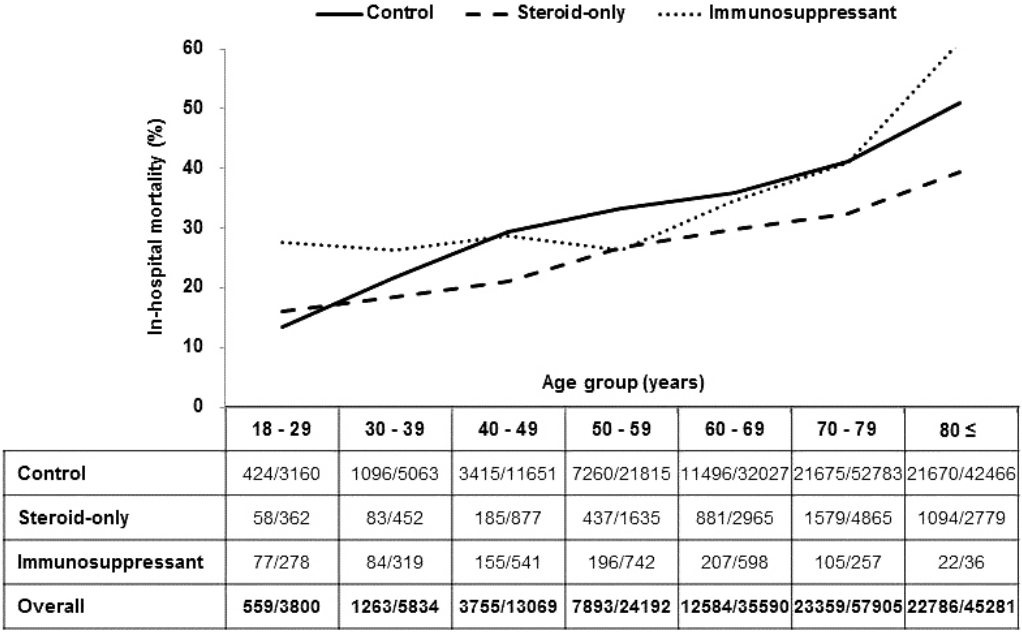
Of total patients, 38.9% had in-hospital mortality, which can be further broken down into 31.0% in the Steroid group, 30.5% in the Suppression group and 39.7% in the control group. In-hospital mortality increased with age in the all groups. In the 18-29 age sub-groups, the in-hospital mortality rate was 16.0%, 27.7% and 13.4% respectively, in the Steroid, Suppression and Control groups. In the 80+ age group, the in-hospitality mortality rate was 39.4%, 61.1% and 51.0%, respectively in the three groups in the same order. The in-hospital mortality was lower in the Steroid group, compared with the Suppression and Control groups, in older groups beyond the 18-29 age group (p < 0.001) (Fig. 1).
Of total patients, 54,119 (29.2%) were admitted to ICU, and the proportion of patients admitted to ICU was significantly higher in the Steroid and Suppression groups than the control group (p < 0.001). While the mean length of stay was 23.7 days for total patients, the Steroid and Suppression groups showed 39.1 and 39.3 days, respectively, meaning that their hospital stay was significantly higher than the control group. The mean total hospital cost of these two groups was also as much as 2-4 times higher than the control group (p < 0.001) (Table 4).
Among the variables of age, gender, comorbidity, ICU admission and history of suppressive drug uses, identified by univariate analysis, all variables except history of suppressive drug use were associated with in-hospital mortality, according to multivariate regression analysis. Comorbidities included diabetes, cardiovascular disease, chronic pulmonary disease, chronic kidney disease, cerebrovascular disease and malignant tumor. The odds ratio of malignancy was higher than those for other comorbidities. The use of suppressive drugs including steroids turned out to be a protective factor reducing the risk of hospital mortality (Table 5).
Discussion
This study explored the presence of immunosuppression and subsequent mortality in sepsis patients on suppressive drugs based on nationwide medical information collected into the HIRA database under the national medical system.
Based on medical information for the period from 2009 to 2013, the total hospital mortality was 38.9% in total sepsis patients. By group, the mortality was 31.0% and 30.5%, respectively, in the Steroid and Suppression groups, which were significantly lower than 39.7% of patients in the control group. However, the admission to ICU, length of stay and total hospital cost were significantly higher in patients on suppressive drugs than those in the control group. According to large scale international studies, sepsis has a mortality rate of 25% to 40% in 2000. A wide range in mortality reflects different levels of sepsis found in the respective studies.[4-6,14-16] This study came up with a relatively higher sepsis mortality rate despite the fact that it was conducted somewhat lately and it included all patients presenting sepsis regardless of their severity.
Sepsis patients in an immunosuppressed state tend to develop non-specific and diverse signs and symptoms, compared to non-immunospressed patients, making it hard to define their severity based on the screening criteria. For instance, these patients did not increase white blood cells or C-reactive protein, which are commonly found in sepsis patients, resulting in failure of early diagnosis and poor prognosis.[17,18] Tolsma suggested that an immunosuppressed state increases the risk of short-term mortality in sepsis patients. His study was more comprehensive than other studies because it addressed immunosuppressed states caused by a wide range of diseases, including white blood cell, inborn immune deficiency, solid tumor and leukemia and suggested an immunosuppressed state as a risk factor influencing prognosis for sepsis.[19]
Steroid drug is commonly prescribed for a variety of conditions due to is anti-inflammatory and immunosuppressive properties, which inhibit the synthesis of proinflammatory cytokines or cyclo-oxygenase. However, prolonged use or overuse of steroid drug can result in excessive immunosuppression, which in turn increases the risk of infection. However, early signs of infection are not easily detectable.[20,21] Steroid-induced risk of various infections such as tuberculosis and oral candidiasis has been reported. A study reported the risk of infection was 1.6 times higher in patients on steroid drug, compared with the control group. [20,22-24] The findings of these studies cannot be compared with this study because we did not investigate clinical characteristics of the Steroid group separately.
Nonetheless, it is true that most of previous studies of sepsis in immunosuppressed patients reported their immunosuppressive state as a risk factor. However, this study found lower mortality rates in sepsis patients on steroid and non-steroid suppressive drugs. This contrasting result may be not supported by a view of literature and pathophysiology. However, we consider the possibility that suppressive drugs helped restore a normal balance between the proinflammatory and anti-inflammatory response. Immune response to sepsis is divided into phases: an early proinflammatory response and late anti-inflammatory response, and imbalance between pro- and anti-inflammatory responses result in death. Excessive pro-inflammatory activity boosts the inflammatory reaction, and excessive anti-inflammatory activity accelerates an immunosuppressed state called immunoparalysis, causing death in both cases.[5,25,26] We assume that suppressive drugs taken before the onset of sepsis can inhibit the early release of mediators such as cytokine, and continuous use of suppressive drugs can also prevent a sudden increase in the anti-inflammatory response, leading to a balance between pro- and anti-inflammatory responses and an increase in survival. Another assumption we make based on the history of suppressive drugs used for patients is that physicians may use a wide range of antibiotics for patients being suspected of sepsis in early stages. With the early use of antibiotics, mortality of sepsis patients decreased in previous studies, and that is also supported by the Surviving Sepsis Campaign.[4,9,27,28] We also assume that early use of antibiotics is more responsible than anti-inflammatory effects of antibiotics for lower mortality in the Steroid and Suppression groups. As mentioned earlier, previous studies described immunosuppressive state induced by conditions such as solid tumor or leukemia as a risk factor for sepsis patients, and their findings are not consistent with the results of this study because we addressed drug-induced immunosuppression.[8,9,29] The protective role of suppressive drugs in sepsis patients in this study can be interpreted as a possible difference between drug- and condition-induced immunosuppressive states in prognosis for sepsis.
The increase in the mean hospital costs as a result of longer period of stay can be explained in the same way. Also, the cause of death in sepsis patients may be other conditions (aggravated comorbidity or complication) than sepsis itself because the cause of death is not correctly described in some cases of insurance claim. That is, sepsis mortality may be more affected by condition-dependent immune deficiency than drug-induced immunosuppression, although further studies are necessary to prove this assumption.
In this study, in-hospital mortality increased with age in all groups, although the presence of immunosuppression varied among different age groups. This finding is in agreement with previous studies in which the onset and severity of sepsis and sepsis mortality were positively correlated with age although actual rates vary among countries.[14,15,30]
This study however has the following limitations: first, to select subjects, this study relied on the category code of sepsis that physicians enter for patients to be discharged. However, entered codes are not monitored, and physicians may not add a new code for patients who were already assigned another category code based on principal diagnosis. This means that the sepsis mortality found in this study may be underestimated because patients who developed sepsis after infection or immune-related comorbidity may not be correctly compiled due to human negligence. Secondly, the results of this study cannot be generalized to a population taking suppressive drugs to determine their sepsis mortality because the study population was defined as sepsis patients taking suppressive drugs and their in-hospital mortality was investigated. We could not confirm the well-established conclusion that immunosuppressed patients are more vulnerable to sepsis than those with healthy immune systems. Thirdly, this study addressed drug-induced immunosuppression only and did not explore condition-induced immunosuppression. Fourthly, the presence of accompanying septic shock and severity of sepsis in each group were not analyzed due to a lack of necessary information. Lastly, we cannot assure that all patients were given a professional care according to standard treatment guidelines such as the Survival Sepsis Guidelines because we cannot confirm such information from the database.
In conclusion, of total sepsis patients, 9.0% were on either steroid or non-steroid suppressive drugs, and their in-hospital mortality was nearly 31%. However, this study did not find a close relationship between immunosuppressed drug use and mortality that is well established in the literature. Further studies are necessary to verify effects of immunosuppressed states.
NOTES
-
No potential conflict of interest relevant to this article was reported.
Fig. 1.In-hospital mortality of sepsis patients. The hospital mortality was positively correlated with age in all age groups, and the mortality rate was lower in the Steroid group, compared with the Suppression and control groups, in the older groups beyond the 18-29 age group, (p < 0.001).

Table 1.Characteristics of sepsis patients
|
Total |
2009 |
2010 |
2011 |
2012 |
2013 |
|
(n = 185,671) |
(n = 37,879) |
(n = 38,739) |
(n = 36,547) |
(n = 38,988) |
(n = 33,518) |
|
Age |
68.4 ± 15.2 |
66.9 ± 15.3 |
67.7 ± 15.2 |
68.5 ± 15.2 |
69.2 ± 15.1 |
69.6 ± 14.9 |
|
Gender (M:F) |
1.1:1 |
1.1:1 |
1.1:1 |
1.1:1 |
1.1:1 |
1.1:1 |
|
Comorbidities |
|
|
|
|
|
|
|
Hypertension |
73,182 (39.4) |
14,432 (38.1) |
15,123 (39.0) |
14,187 (38.8) |
15,692 (40.3) |
13,748 (41.0) |
|
Diabetes |
79,125 (42.6) |
16,261 (42.9) |
16,508 (42.6) |
15,121 (41.4) |
16,703 (42.8) |
14,532 (43.4) |
|
Coronary artery disease |
20,363 (11.0) |
5,288 (14.0) |
4,353 (11.2) |
3,628 (9.9) |
3,915 (10.0) |
3,179 (9.5) |
|
Chronic pulmonary disease |
16,743 (9.0) |
3,460 (9.1) |
3,487 (9.0) |
3,159 (8.6) |
3,587 (9.2) |
3,050 (9.1) |
|
Chronic liver disease |
6,074 (3.3) |
1,339 (3.5) |
1,338 (3.5) |
1,199 (3.3) |
1,219 (3.1) |
979 (2.9) |
|
Chronic kidney disease |
13,854 (7.5) |
2,752 (7.3) |
2,711 (7.0) |
2,691 (7.4) |
3,006 (7.7) |
2,694 (8.0) |
|
Cerebrovascular disease |
21,483 (11.6) |
4,560 (12) |
4,555 (11.8) |
4,188 (11.5) |
4,590 (11.8) |
3,590 (10.7) |
|
Malignancy |
12,038 (6.5) |
2,674 (7.1) |
2,688 (6.9) |
2,297 (6.3) |
2,367 (6.1) |
2,012 (6) |
|
Immunosuppressants |
|
|
|
|
|
|
|
Leukemia |
1,516 (9.1) |
340 (9.8) |
358 (10.2) |
259 (7.9) |
314 (8.8) |
245 (8.6) |
|
Organ transplantation |
1,077 (6.5) |
228 (6.5) |
239 (6.8) |
245 (7.5) |
198 (5.6) |
167 (5.8) |
|
Rheumatoid arthritis |
760 (4.6) |
177 (5.1) |
151 (4.3) |
139 (4.2) |
159 (4.5) |
134 (4.7) |
|
Crohn’s disease |
42 (0.3) |
2 (0.1) |
16 (0.5) |
8 (0.2) |
10 (0.3) |
6 (0.2) |
|
Ulcerative colitis |
65 (0.4) |
17 (0.5) |
14 (0.4) |
15 (0.5) |
14 (0.4) |
5 (0.2) |
|
Atopic dermatitis |
126 (0.8) |
35 (1.0) |
23 (0.7) |
19 (0.6) |
23 (0.6) |
26 (0.9) |
|
Multiple sclerosis |
32 (0.2) |
13 (0.4) |
4 (0.1) |
3 (0.1) |
6 (0.2) |
6 (0.2) |
|
Others |
14,664 (87.8) |
3,039 (87.2) |
3,060 (87.5) |
2,865 (87.1) |
3,168 (88.8) |
2,532 (88.4) |
Table 2.Characteristics of sepsis patients
|
Total |
Control |
Steroid-only |
Immunosuppressant |
p-value |
|
(n = 185,671) |
(n = 168,965) |
(n = 13,935) |
(n = 2,771) |
|
Age |
68.4 ± 15.2 |
68.7 ± 15.0 |
67.7 ± 14.8 |
51.2 ± 15.0 |
< 0.001*
|
|
Gender (M:F) |
1.1:1 |
1.1:1 |
1:1 |
1.3:1 |
< 0.001 |
|
Comorbidities |
|
|
|
|
|
|
Hypertension |
73,182 (39.4) |
65,177 (38.57) |
7,089 (50.87) |
916 (33.06) |
< 0.001 |
|
Diabetes |
79,125 (42.6) |
71,290 (42.19) |
6,747 (48.42) |
1,088 (39.26) |
< 0.001 |
|
Coronary artery disease |
20,363 (11.0) |
18,192 (10.77) |
1,985 (14.24) |
186 (6.71) |
< 0.001 |
|
Chronic pulmonary disease |
16,743 (9.02) |
13,870 (8.21) |
2,742 (19.68) |
131 (4.73) |
< 0.001 |
|
Chronic liver disease |
6,074 (3.27) |
5,521 (3.27) |
332 (2.38) |
221 (7.98) |
< 0.001 |
|
Chronic kidney disease |
13,854 (7.46) |
12,212 (7.23) |
1,366 (9.80) |
276 (9.96) |
< 0.001 |
|
Cerebrovascular disease |
21,483 (11.57) |
19,644 (11.63) |
1,696 (12.17) |
143 (5.16) |
< 0.001 |
|
Malignancy |
12,038 (6.48) |
9,535 (5.64) |
1,473 (10.57) |
1,030 (37.17) |
< 0.001 |
Table 3.Distribution of immunosuppression in sepsis patients
|
|
Total |
Control |
Steroid-only |
Immunosuppressant |
p-value |
|
(n = 185,671) |
(n = 168,965) |
(n = 13,935) |
(n = 2,771) |
|
18 ≤ age < 30 |
Male |
2,015 |
1,663 (82.5) |
191 (9.5) |
161 (8.0) |
0.240 |
|
Female |
1,785 |
1,497 (83.9) |
171 (9.6) |
117 (6.6) |
|
|
Total |
3,800 |
3,160 (83.2) |
362 (9.5) |
278 (7.3) |
|
|
30 ≤ age < 40 |
Male |
3,065 |
2,661 (86.8) |
232 (7.6) |
172 (5.6) |
0.770 |
|
Female |
2,769 |
2,402 (86.7) |
220 (7.9) |
147 (5.3) |
|
|
Total |
5,834 |
5,063 (86.8) |
452 (7.7) |
319 (5.5) |
|
|
40 ≤ age < 50 |
Male |
7,916 |
7,136 (90.1) |
476 (6.0) |
304 (3.8) |
< 0.001 |
|
Female |
5,153 |
4,515 (87.6) |
401 (7.8) |
237 (4.6) |
|
|
Total |
13,069 |
11,651 (89.1) |
877 (6.7) |
541 (4.1) |
|
|
50 ≤ age < 60 |
Male |
15,408 |
14,052 (91.2) |
922 (6) |
434 (2.8) |
< 0.001 |
|
Female |
8,784 |
7,763 (88.4) |
713 (8.1) |
308 (3.5) |
|
|
Total |
24,192 |
21,815 (90.2) |
1,635 (6.8) |
742 (3.1) |
|
|
60 ≤ age < 70 |
Male |
21,124 |
19,141 (90.6) |
1,642 (7.8) |
341 (1.6) |
< 0.001 |
|
Female |
14,466 |
12,886 (89.1) |
1,323 (9.1) |
257 (1.8) |
|
|
Total |
35,590 |
32,027 (90.0) |
2,965 (8.3) |
598 (1.7) |
|
|
70 ≤ age < 80 |
Male |
29,510 |
26,991 (91.5) |
2,386 (8.1) |
133 (0.5) |
0.020 |
|
Female |
28,395 |
25,792 (90.8) |
2,479 (8.7) |
124 (0.4) |
|
|
Total |
57,905 |
52,783 (91.2) |
4,865 (8.4) |
257 (0.4) |
|
|
80 ≤ age |
Male |
17,570 |
16,369 (93.2) |
1,179 (6.7) |
22 (0.1) |
< 0.001 |
|
Female |
27,711 |
26,097 (94.2) |
1,600 (5.8) |
14 (0.1) |
|
|
Total |
45,281 |
42,466 (93.8) |
2,779 (6.1) |
36 (0.1) |
|
|
Total |
Male |
96,608 |
88,013 (91.1) |
7,028 (7.3) |
1,567 (1.6) |
< 0.001 |
|
Female |
89,063 |
80,952 (90.9) |
6,907 (7.8) |
1,204 (1.4) |
|
Table 4.ICU admission, length of stay and total cost of sepsis patients
|
Total |
Control |
Steroid-only |
Immunosuppressant |
p-value |
|
(n = 185,671) |
(n = 168,965) |
(n = 13,935) |
(n = 2,771) |
|
ICU admission |
54,119 (29.2) |
47,129 (27.9) |
5,819 (41.8) |
1,171 (42.3) |
< 0.001 |
|
Length of stay (days) |
23.7 ± 31.3 |
22.2 ± 29.4 |
39.1 ± 44.4 |
39.3 ± 35.0 |
< 0.001*
|
|
Total cost (1,000 KWN) |
8,440 ± 12,591 |
7,630 ± 10,940 |
14,100 ± 17,162 |
29,366 ± 34,703 |
< 0.001*
|
Table 5.Logistic regression analysis of risk factors for in-hospital mortality of admitted sepsis patients
|
Factors |
Univariate Factors
|
Multivariate
|
|
OR |
95% CI |
p-value |
OR |
95% CI |
p-value |
|
Age (categorical variable) |
|
|
|
|
|
|
|
18 ≤ age < 30 |
1.00 |
- |
|
1.00 |
- |
|
|
30 ≤ age < 40 |
1.60 |
(1.44, 1.79) |
< 0.001 |
1.58 |
(1.42, 1.77) |
< 0.001 |
|
40 ≤ age < 50 |
2.34 |
(2.12, 2.58) |
< 0.001 |
2.22 |
(2.01, 2.45) |
< 0.001 |
|
50 ≤ age < 60 |
2.81 |
(2.56, 3.08) |
< 0.001 |
2.64 |
(2.40, 2.90) |
< 0.001 |
|
60 ≤ age < 70 |
3.17 |
(2.89, 3.48) |
< 0.001 |
3.05 |
(2.78, 3.35) |
< 0.001 |
|
70 ≤ age < 80 |
3.92 |
(3.58, 4.29) |
< 0.001 |
4.07 |
(3.71, 4.46) |
< 0.001 |
|
80 ≤ age |
5.87 |
(5.36, 6.44) |
< 0.001 |
6.71 |
(6.12, 7.37) |
< 0.001 |
|
Gender (male) |
1.21 |
(1.19, 1.23) |
< 0.001 |
1.28 |
(1.25, 1.31) |
< 0.001 |
|
Comorbidities |
|
|
|
|
|
|
|
Hypertension |
0.87 |
(0.85, 0.88) |
< 0.001 |
0.72 |
(0.71, 0.74) |
< 0.001 |
|
Diabetes |
1.07 |
(1.05, 1.09) |
< 0.001 |
1.07 |
(1.05, 1.09) |
< 0.001 |
|
Coronary artery disease |
1.14 |
(1.11, 1.18) |
< 0.001 |
1.04 |
(1.01, 1.07) |
0.014 |
|
Chronic pulmonary disease |
1.38 |
(1.34, 1.43) |
< 0.001 |
1.19 |
(1.15, 1.23) |
< 0.001 |
|
Chronic liver disease |
1.27 |
(1.21, 1.34) |
< 0.001 |
1.44 |
(1.37, 1.52) |
< 0.001 |
|
Chronic kidney disease |
1.24 |
(1.20, 1.28) |
< 0.001 |
1.35 |
(1.30, 1.40) |
< 0.001 |
|
Cerebrovascular disease |
1.12 |
(1.09, 1.15) |
< 0.001 |
1.07 |
(1.03, 1.10) |
< 0.001 |
|
Malignancy |
1.71 |
(1.65, 1.78) |
< 0.001 |
2.03 |
(1.95, 2.11) |
< 0.001 |
|
ICU admission |
1.41 |
(1.38, 1.44) |
< 0.001 |
1.50 |
(1.47, 1.53) |
< 0.001 |
|
Immunosuppressants |
|
|
|
|
|
|
|
Control |
1.00 |
- |
|
1.00 |
- |
|
|
Steroid-only |
0.68 |
(0.66, 0.71) |
<0.001 |
0.64 |
(0.62, 0.66) |
< 0.001 |
|
Immunosuppressant |
0.67 |
(0.62, 0.73) |
<0.001 |
0.74 |
(0.68, 0.81) |
< 0.001 |
References
- 1. Castellanos-Ortega A, Suberviola B, García-Astudillo LA, Holanda MS, Ortiz F, Llorca J, et al. Impact of the Surviving Sepsis Campaign protocols on hospital length of stay and mortality in septic shock patients: results of a three-year follow-up quasi-experimental study. Crit Care Med 2010;38:1036-43.ArticlePubMed
- 2. Dellinger RP, Levy MM, Rhodes A, Annane D, Gerlach H, Opal SM, et al. Surviving Sepsis Campaign: international guidelines for management of severe sepsis and septic shock, 2012. Intensive Care Med 2013;39:165-228.ArticlePubMedPMC
- 3. Shiramizo SC, Marra AR, Durão MS, Paes ÂT, Edmond MB, Pavão dos Santos OF. Decreasing mortality in severe sepsis and septic shock patients by implementing a sepsis bundle in a hospital setting. PLoS One 2011;6:e26790.ArticlePubMedPMC
- 4. Dellinger RP, Levy MM, Carlet JM, Bion J, Parker MM, Jaeschke R, et al. Surviving Sepsis Campaign: international guidelines for management of severe sepsis and septic shock: 2008. Crit Care Med 2008;36:296-327.ArticlePubMed
- 5. Leentjens J, Kox M, van der Hoeven JG, Netea MG, Pickkers P. Immunotherapy for the adjunctive treatment of sepsis: from immunosuppression to immunostimulation. Time for a paradigm change? Am J Respir Crit Care Med 2013;187:1287-93.ArticlePubMed
- 6. Galbois A, Aegerter P, Martel-Samb P, Housset C, Thabut D, Offenstadt G, et al. Improved prognosis of septic shock in patients with cirrhosis: a multicenter study*. Crit Care Med 2014;42:1666-75.ArticlePubMed
- 7. Brun-Buisson C, Doyon F, Carlet J, Dellamonica P, Gouin F, Lepoutre A, et al. Incidence, risk factors, and outcome of severe sepsis and septic shock in adults. A multicenter prospective study in intensive care units. French ICU Group for Severe Sepsis. JAMA 1995;274:968-74.ArticlePubMed
- 8. Linden PK. Approach to the immunocompromised host with infection in the intensive care unit. Infect Dis Clin North Am 2009;23:535-56.ArticlePubMed
- 9. Weiss SL, Fitzgerald JC, Balamuth F, Alpern ER, Lavelle J, Chilutti M, et al. Delayed antimicrobial therapy increases mortality and organ dysfunction duration in pediatric sepsis. Crit Care Med 2014;42:2409-17.ArticlePubMedPMC
- 10. Kroschinsky F, Weise M, Illmer T, Haenel M, Born-haeuser M, Hoeffken G, et al. Outcome and prognostic features of intensive care unit treatment in patients with hematological malignancies. Intensive Care Med 2002;28:1294-300.ArticlePubMed
- 11. Japiassú AM, Amâncio RT, Mesquita EC, Medeiros DM, Bernal HB, Nunes EP, et al. Sepsis is a major determinant of outcome in critically ill HIV/AIDS patients. Crit Care 2010;14:R152. ArticlePubMedPMC
- 12. Choi NK, Chang Y, Choi YK, Hahn S, Park BJ. Signal detection of rosuvastatin compared to other statins: data-mining study using national health insurance claims database. Pharmacoepidemiol Drug Saf 2010;19:238-46.ArticlePubMed
- 13. Koo BK, Lee CH, Yang BR, Hwang SS, Choi NK. The incidence and prevalence of diabetes mellitus and related atherosclerotic complications in Korea: a National Health Insurance Database Study. PLoS One 2014;9:e110650.ArticlePubMedPMC
- 14. Shen HN, Lu CL, Yang HH. Epidemiologic trend of severe sepsis in Taiwan from 1997 through 2006. Chest 2010;138:298-304.ArticlePubMed
- 15. Kumar G, Kumar N, Taneja A, Kaleekal T, Tarima S, McGinley E, et al. Nationwide trends of severe sepsis in the 21st century (2000-2007). Chest 2011;140:1223-31.ArticlePubMed
- 16. Kübler A, Adamik B, Durek G, Mayzner-Zawadzka E, Gaszyński W, Karpel E, et al. Results of the severe sepsis registry in intensive care units in Poland from 2003-2009. Anaesthesiol Intensive Ther 2015;47:7-13.ArticlePubMed
- 17. Bouza E, Loeches B, Muñoz P. Fever of unknown origin in solid organ transplant recipients. Infect Dis Clin North Am 2007;21:1033. -54. .ArticlePubMed
- 18. Kieslichova E, Rocen M, Merta D, Kudla M, Splichal I, Cap J, et al. The effect of immunosuppression on manifestations of sepsis in an animal model of cecal ligation and puncture. Transplant Proc 2013;45:770-7.ArticlePubMed
- 19. Tolsma V, Schwebel C, Azoulay E, Darmon M, Souweine B, Vesin A, et al. Sepsis severe or septic shock: outcome according to immune status and immunodeficiency profile. Chest 2014;146:1205-13.ArticlePubMed
- 20. Schäcke H, Döcke WD, Asadullah K. Mechanisms involved in the side effects of glucocorticoids. Pharmacol Ther 2002;96:21-43.Article
- 21. Buttgereit F, Burmester GR, Lipworth BJ. Optimised glucocorticoid therapy: the sharpening of an old spear. Lancet 2005;365:801-3.ArticlePubMed
- 22. Senderovitz T, Viskum K. Corticosteroids and tuberculosis. Respir Med 1994;88:561-5.ArticlePubMed
- 23. Cline JC, Davis SM. Risks of infection or reactivation of tuberculosis associated with chronic corticosteroid therapy. Ann Pharmacother 1997;31:775-6.PubMed
- 24. Kennedy WA, Laurier C, Gautrin D, Ghezzo H, Paré M, Malo JL, et al. Occurrence and risk factors of oral candidiasis treated with oral antifungals in seniors using inhaled steroids. J Clin Epidemiol 2000;53:696-701.ArticlePubMed
- 25. Girardin E, Grau GE, Dayer JM, Roux-Lombard P, Lambert PH. Tumor necrosis factor and interleukin-1 in the serum of children with severe infectious purpura. N Engl J Med 1988;319:397-400.ArticlePubMed
- 26. Hotchkiss RS, Karl IE. The pathophysiology and treatment of sepsis. N Engl J Med 2003;348:138-50.ArticlePubMed
- 27. Ferrer R, Martin-Loeches I, Phillips G, Osborn TM, Townsend S, Dellinger RP, et al. Empiric antibiotic treatment reduces mortality in severe sepsis and septic shock from the first hour: results from a guideline-based performance improvement program. Crit Care Med 2014;42:1749-55.ArticlePubMed
- 28. Yokota PK, Marra AR, Martino MD, Victor ES, Durão MS, Edmond MB, et al. Impact of appropriate antimicrobial therapy for patients with severe sepsis and septic shock--a quality improvement study. PLoS One 2014;9:e104475.ArticlePubMedPMC
- 29. Poutsiaka DD, Davidson LE, Kahn KL, Bates DW, Snydman DR, Hibberd PL. Risk factors for death after sepsis in patients immunosuppressed before the onset of sepsis. Scand J Infect Dis 2009;41:469-79.ArticlePubMedPMC
- 30. Dombrovskiy VY, Martin AA, Sunderram J, Paz HL. Rapid increase in hospitalization and mortality rates for severe sepsis in the United States: a trend analysis from 1993 to 2003. Crit Care Med 2007;35:1244-50.ArticlePubMed
Citations
Citations to this article as recorded by

- Predictive performance of NEWS and qSOFA in immunocompromised sepsis patients at the emergency department
Lisanne Boekhoud, Helena M. E. A. Schaap, Rick L. Huizinga, Tycho J. Olgers, Jan C. ter Maaten, Douwe F. Postma, Hjalmar R. Bouma
Infection.2024;[Epub] CrossRef - Effects of Early Initiation of High-Dose Dexamethasone Therapy on Pro-Inflammatory Cytokines and Mortality in LPS-Challenged Mice
Ji-young Son, Won Gun Kwack, Eun Kyoung Chung, Sooyoung Shin, Yeo Jin Choi
Healthcare.2022; 10(7): 1247. CrossRef - Alleviation of LPS-Induced Inflammation and Septic Shock by Lactiplantibacillus plantarum K8 Lysates
Gayoung Kim, Kyeong-Hun Choi, Hangeun Kim, Dae-Kyun Chung
International Journal of Molecular Sciences.2021; 22(11): 5921. CrossRef - Critical Care Research Using “Big Data”: A Reality in the Near Future
Kwangha Lee
Acute and Critical Care.2018; 33(4): 269. CrossRef - Should Very Old Patients Be Admitted to the Intensive Care Units?
Jun Kwon Cha, In-Ae Song
The Korean Journal of Critical Care Medicine.2017; 32(4): 376. CrossRef - Sepsis in Immunocompromised Patients: Current Status in Korea
Kwangha Lee
The Korean Journal of Critical Care Medicine.2015; 30(4): 239. CrossRef









 KSCCM
KSCCM
 PubReader
PubReader ePub Link
ePub Link Cite
Cite