Articles
- Page Path
- HOME > Acute Crit Care > Volume 33(2); 2018 > Article
- Original Article Current Status and Survival Impact of Infectious Disease Consultation for Multidrug-Resistant Bacteremia in Ventilated Patients: A Single-Center Experience in Korea
-
Insu Kim, Won-Young Kim, Eun Suk Jeoung, Kwangha Lee

-
Acute and Critical Care 2018;33(2):73-82.
DOI: https://doi.org/10.4266/acc.2017.00591
Published online: April 26, 2018
Division of Pulmonary, Allergy and Critical Care Medicine, Department of Internal Medicine, Pusan National University School of Medicine, Busan, Korea
- Correspondence to: Kwangha Lee, Division of Pulmonary, Allergy and Critical Care Medicine, Department of Internal Medicine, Pusan National University School of Medicine, 179 Gudeok-ro, Seo-gu, Busan 49241, Korea Tel: +82-51-240-7743, Fax: +82-52-245-3127, E-mail: jubilate@pusan.ac.kr
Copyright © 2018 The Korean Society of Critical Care Medicine
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
-
Background
- We evaluated the current status and survival impact of infectious disease consultation (IDC) in ventilated patients with multidrug-resistant (MDR) bacteremia.
-
Methods
- One hundred sixty-one consecutive patients from a single tertiary care hospital were enrolled over a 5-year period. Patients with at least one of the following six MDR bacteremias were included: methicillin-resistant Staphylococcus aureus, extended-spectrum β-lactamase-producing gram-negative bacteria (Escherichia coli and Klebsiella pneumonia), carbapenem-resistant gram-negative rods (Acinetobacter baumannii and Pseudomonas aeruginosa), and vancomycin-resistant Enterococcus faecium.
-
Results
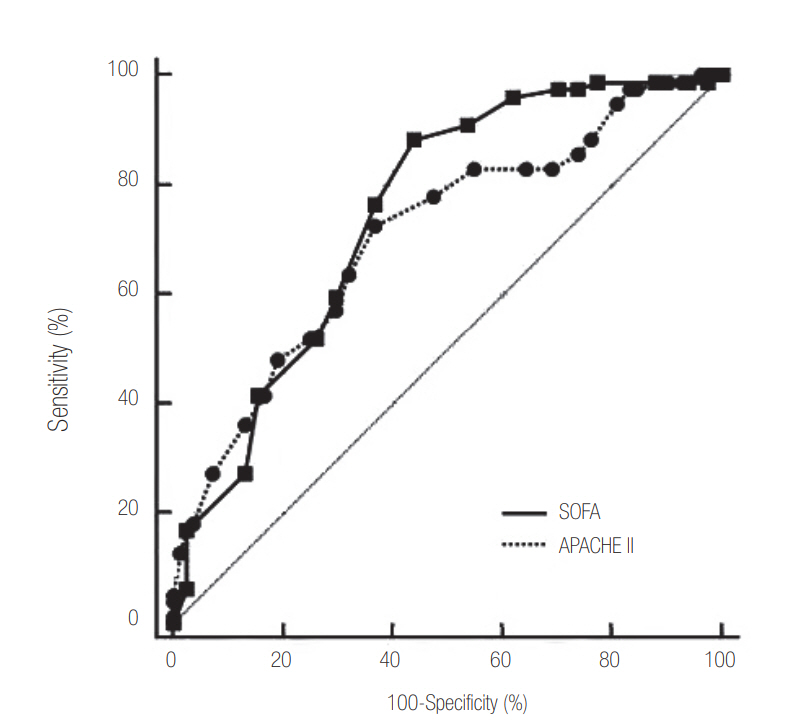
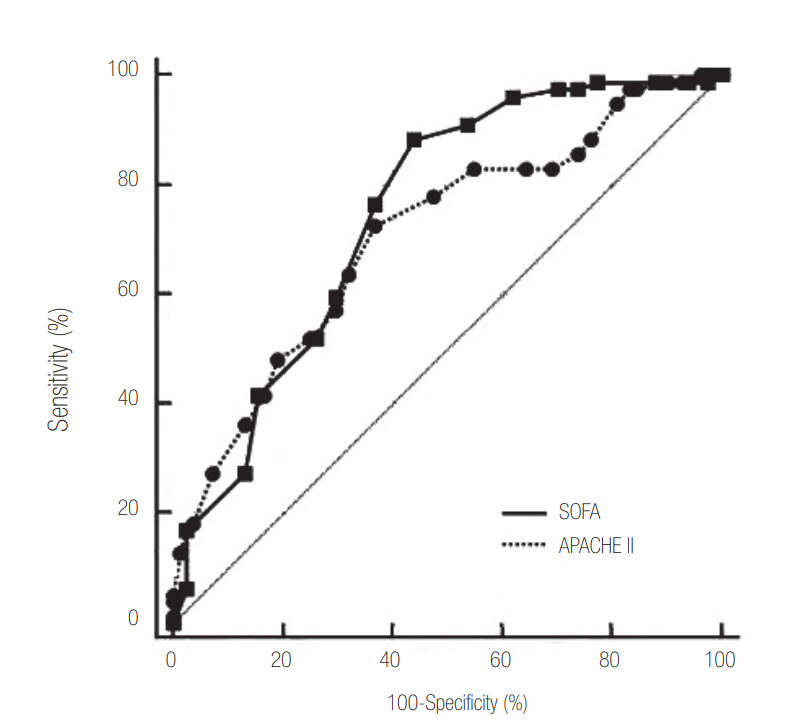
- Median patient age was 66 years (range, 18 to 95), and 57.8% of subjects were male. The 28-day mortality after the day of blood culture was 52.2%. An IDC was requested for 96 patients based on a positive blood culture (59.6%). Patients without IDC had significantly higher rate of hemato-oncologic diseases as a comorbidity (36.9% vs. 11.5%, P < 0.001). Patients without an IDC had higher Acute Physiology and Chronic Health Evaluation (APACHE) II score (median, 20; range, 8 to 38 vs. median, 16; range, 5 to 34, P < 0.001) and Sequential Organ Failure Assessment (SOFA) score (median, 9; range, 2 to 17 vs. median, 7; range, 2 to 20; P = 0.020) on the day of blood culture and a higher 28-day mortality rate (72.3% vs. 38.5%, P < 0.001). In patients with SOFA ≥9 (cut-off level based on Youden’s index) on the day of blood culture and gram-negative bacteremia, IDC was also significantly associated with lower 28-day mortality (hazard ratio [HR], 0.298; 95% confidence interval [CI], 0.167 to 0.532 and HR, 0.180; 95% CI, 0.097 to 0.333; all P < 0.001) based on multivariate Cox regression analysis.
-
Conclusions
- An IDC for MDR bacteremia was requested less often for ventilated patients with greater disease severity and higher 28-day mortality after blood was drawn. In patients with SOFA ≥9 on the day of blood culture and gram-negative bacteremia, IDC was associated with improved 28-day survival after blood draw for culture.
Introduction
MATERIALS AND METHODS
RESULTS
DISCUSSION
Acknowledgments

| Characteristics | Total (n = 161) | IDC (n = 96) | No IDC (n = 65) | P-value |
|---|---|---|---|---|
| Age (yr) | 66 (18–95) | 62 (20–91) | 66 (18–95) | 0.526 |
| Male sex | 93 (57.8) | 56 (58.3) | 37 (56.7) | 0.872 |
| APACHE II scorea | 18 (5–38) | 16 (5–34) | 20 (8–38) | <0.001 |
| SOFA scorea | 8 (2–20) | 7 (2–20) | 9 (2–17) | 0.020 |
| ICU LOS (day) | 23 (1–430) | 34 (2–430) | 16 (1–177) | 0.003 |
| Hospital LOS (day) | 43 (2–431) | 58 (10–431) | 21 (2–422) | <0.001 |
| Total MV (day) | 19 (1–430) | 23 (1–430) | 12 (1–118) | 0.012 |
| Comorbidity | ||||
| Diabetes | 46 (28.6) | 28 (29.2) | 18 (27.7) | 0.861 |
| Cardiovascular disease | 37 (23.0) | 19 (19.8) | 18 (27.7) | 0.258 |
| Hemato-oncologic disease | 35 (21.7) | 11 (11.5) | 24 (36.9) | <0.001 |
| Cerebrovascular disease | 27 (16.8) | 18 (18.8) | 9 (13.8) | 0.520 |
| Chronic kidney disease | 22 (13.7) | 14 (14.6) | 8 (12.3) | 0.816 |
| Chronic lung diseaseb | 17 (10.6) | 11 (11.5) | 6 (9.2) | 0.796 |
| Chronic liver disease | 14 (8.7) | 9 (9.4) | 5 (7.7) | 0.783 |
| Neuromuscular disease | 13 (8.1) | 8 (8.3) | 5 (7.7) | >0.999 |
| Source of infectionc | ||||
| Respiratory | 76 (47.2) | 48 (50.0) | 28 (43.1) | 0.424 |
| Catheter-related | 43 (26.7) | 35 (36.5) | 8 (12.3) | 0.001 |
| Musculoskeletal and soft tissue | 26 (16.1) | 20 (20.8) | 6 (9.2) | 0.053 |
| Intra-abdomen | 12 (7.5) | 6 (6.3) | 6 (9.2) | 0.548 |
| Urinary tract | 5 (3.1) | 3 (3.1) | 2 (3.1) | >0.999 |
| ICU type | ||||
| Medical | 65 (40.4) | 34 (35.4) | 31 (47.7) | 0.142 |
| Surgical | 17 (10.6) | 10 (10.4) | 7 (10.8) | >0.999 |
| Emergency | 40 (24.8) | 23 (24.0) | 17 (26.2) | 0.853 |
| Neurosurgical | 18 (11.2) | 11 (11.5) | 7 (10.8) | >0.999 |
| Cardio-stroke | 8 (5.0) | 5 (5.2) | 3 (4.6) | >0.999 |
| Trauma | 13 (8.1) | 13 (13.5) | 0 | 0.001 |
| Bacteria | ||||
| Methicillin-resistant Staphylococcus aureus | 59 (36.6) | 36 (37.5) | 23 (35.4) | 0.868 |
| Carbapenem-resistant Acinetobacter baumannii | 59 (36.6) | 37 (38.5) | 22 (33.8) | 0.618 |
| Vancomycin-resistant Enterococcus faecium | 20 (12.4) | 11 (11.5) | 9 (13.8) | 0.808 |
| ESBL(+) Klebsiella pneumoniae | 18 (11.2) | 11 (11.5) | 7 (10.8) | >0.999 |
| ESBL(+) Escherichia coli | 9 (5.6) | 5 (5.2) | 4 (6.2) | >0.999 |
| Carbapenem-resistant Pseudomonas aeruginosa | 5 (3.1) | 2 (2.1) | 3 (4.6) | 0.394 |
| Period from ICU admission to bacteremia (day) | 9 (0–413) | 10 (0–413) | 7 (0–366) | 0.554 |
| Administration of the first recommended antibioticsd | 120 (74.5) | 85 (88.5) | 35 (53.8) | <0.001 |
| Period of administration of the first recommended antibiotics (day)d | ||||
| Before microorganisms identification | 0 (0–55) | 0 (0–25) | 0 (0–55) | 0.979 |
| After microorganisms identification | 7 (0–116) | 14 (0–116) | 0 (0–33) | <0.001 |
| Requirement for vasopressora | 82 (50.9) | 39 (40.6) | 43 (66.2) | 0.002 |
| Requirement of hemodialysisa | 44 (27.3) | 20 (20.8) | 24 (36.9) | 0.031 |
| Requirement for neuromuscular blocking agenta | 39 (24.2) | 19 (19.8) | 20 (30.8) | 0.134 |
| 28-Day mortality | 84 (52.2) | 37 (38.5) | 47 (72.3) | <0.001 |
Values are presented as median (range) for continuous variables or number (%) for categorical variables.
IDC: infectious disease consultation; APACHE: Acute Physiology and Chronic Health Evaluation; SOFA: Sequential Organ Failure Assessment; ICU: intensive care unit; LOS: length of stay; MV: mechanical ventilation; ESBL: extended-spectrum beta-lactamases.
a All clinical data was calculated or checked from medical records on blood collection day;
b Chronic lung diseases included chronic obstructive pulmonary disease, asthma and bronchiectasis;
c Of total patients, 24 patients had more than two sources of infection;
d The first recommended antibiotics were known as “drug of choice” and were summarized as follows: methicillin-resistant Staphylococcus aureus, vancomycin; Carbapenem-resistant Acinetobacter baumannii and carbapenem-resistant Pseudomonas aeruginosa, colistin; Vancomycin-resistant Enterococcus faecium, Linezolid; Carbapenem, ESBL(+) Klebsiella pneumonia and ESBL(+) Escherichia coli.
| Organism |
ESBL (n = 27) |
CRPA & CRAB (n = 64) |
MRSA (n = 59) |
VRE (n = 20) |
||||
|---|---|---|---|---|---|---|---|---|
| IDC (n = 16) | No IDC (n = 11) | IDC (n = 39) | No IDC (n = 25) | IDC (n = 36) | No IDC (n = 23) | IDC (n = 11) | No IDC (n = 9) | |
| Glycopeptide | 6 (37.5) | 4 (36.4) | 13 (33.3) | 10 (40.0) | 30 (83.3) | 19 (82.6) | 1 (9.1)a | 5 (55.6)a |
| Carbapenem | 16 (100)a | 5 (45.5)a | 13 (33.3) | 12 (48.0) | 9 (25.0) | 6 (26.1) | 1 (9.1) | 7 (77.8) |
| Fluoroquinolone | 1 (6.3) | 4 (36.4) | 1 (2.6)a | 8 (32.0)a | 5 (13.9) | 4 (17.4) | 1 (9.1) | 1 (11.1) |
| Third-generation cephalosporins | 0 | 11 (18.2) | 1 (2.6)a | 7 (28.0)a | 5 (13.9) | 3 (13.0) | 0 | 0 |
| Anti-pseudomonal penicillin | 0 | 2 (18.2) | 3 (7.7) | 2 (8.0) | 4 (11.1) | 3 (13.0) | 0 | 0 |
| Fourth generation cephalosporins | 0 | 0 | 2 (5.1) | 2 (8.0) | 10 (27.8) | 4 (17.4) | 1 (9.1) | 0 |
| Colistin | 5 (31.3) | 1 (9.1) | 32 (82.1)a | 13 (52.0)a | 8 (22.2) | 2 (8.7) | 2 (18.2) | 1 (11.1) |
| Aminoglycoside | 0 | 0 | 1 (2.6) | 1 (4.0) | 1 (2.8) | 0 | 0 | 0 |
| Minocycline | 5 (31.3) | 0 | 15 (38.5)a | 2 (8.0)a | 1 (2.8) | 2 (8.7) | 0 | 1 (11.1) |
| Macrolide | 0 | 0 | 3 (7.7) | 2 (8.0) | 1 (2.8) | 0 | 0 | 0 |
| Second generation cephalosporins | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Trimethoprim-sulfamethoxazole | 0 | 0 | 1 (2.6) | 1 (4.0) | 0 | 0 | 0 | 1 (11.1) |
| Linezolid | 1 (6.3) | 1 (9.1) | 0 | 1 (4.0) | 3 (8.3) | 2 (8.7) | 7 (63.6) | 3 (33.3) |
| Tigecycline | 0 | 1 (9.1) | 3 (7.7) | 0 | 0 | 1 (4.3) | 1 (9.1) | 0 |
Values are presented as number (%).
IDC: infectious disease consultation; ESBL: extended-spectrum β-lactamase; CRPA: carbapenem-resistant Pseudomonas aeruginosa; CRAB: carbapenem-resistant Acinetobacter baumannii; MRSA: methicillin-resistant Staphylococcus aureus; VRE: vancomycin-resistant Enterococcus faecium.
a In the chi-square or Fisher exact tests (for small numbers), significance was defined as P < 0.05.
| Factor |
Univariate analysis |
Multivariate analysis |
||
|---|---|---|---|---|
| HR (95% CI) | P-value | HR (95% CI) | P-value | |
| Total patients | ||||
| Infectious disease consultation | 0.325 (0.210–0.502) | <0.001 | 0.353 (0.226–0.552) | <0.001 |
| Requirement for vasopressorsa | 3.154 (1.980–5.025) | <0.001 | 2.409 (1.483–3.914) | <0.001 |
| Requirement for HDa | 2.599 (1.672–4.043) | <0.001 | 1.640 (1.037–2.593) | 0.034 |
| Requirement for NMBAsa | 1.978 (1.248–3.314) | 0.004 | 2.357 (1.446–3.843) | 0.001 |
| Thrombocytopeniaa | 2.298 (1.436–3.675) | 0.001 | 1.747 (1.078–2.830) | 0.023 |
| Hemato-oncologic diseases as comorbidities | 2.187 (1.373–3.483) | 0.001 | - | - |
| Age ≥65 yr | 1.591 (1.028–2.460) | 0.037 | - | - |
| In patients with SOFA ≥9a | ||||
| Infectious disease consultation | 0.313 (0.177–0.554) | <0.001 | 0.298 (0.167–0.532) | <0.001 |
| Requirement for NMBAsa | 1.681 (0.959–2.949) | 0.070 | 1.834 (1.033–3.256) | 0.038 |
| Requirement for vasopressorsa | 1.968 (0.958–4.041) | 0.065 | - | - |
| Requirement for HDa | 1.837 (1.065–3.169) | 0.029 | - | - |
| In patients with gram-negative bacteremia | ||||
| Infectious disease consultation | 0.203 (0.114–0.361) | <0.001 | 0.180 (0.097–0.333) | <0.001 |
| Requirement for vasopressorsa | 3.178 (1.733–5.831) | <0.001 | 3.168 (1.670–6.010) | <0.001 |
| Requirement for HDa | 3.047 (1.753–5.298) | <0.001 | 2.607 (1.473–4.614) | 0.001 |
| Requirement for NMBAsa | 2.226 (1.249–3.969) | 0.007 | 1.879 (1.025–3.446) | 0.041 |
| Thrombocytopeniaa | 1.722 (0.932–3.183) | 0.083 | - | - |
| Hemato-oncologic diseases as comorbidities | 1.648 (0.929–2.922) | 0.087 | - | - |
Statistical significance was tested by univariate Cox proportional hazards models and multivariate Cox hazards models.
HR: hazard ratio; CI: confidence interval; HD: hemodialysis; NMBA: neuromuscular blocking agents; SOFA: Sequential Organ Failure Assessment.
a All clinical data was calculated or checked from medical records on blood collection day.
- 1. Brusselaers N, Vogelaers D, Blot S. The rising problem of antimicrobial resistance in the intensive care unit. Ann Intensive Care 2011;1:47. ArticlePubMedPMC
- 2. Magiorakos AP, Srinivasan A, Carey RB, Carmeli Y, Falagas ME, Giske CG, et al. Multidrug-resistant, extensively drug-resistant and pandrug-resistant bacteria: an international expert proposal for interim standard definitions for acquired resistance. Clin Microbiol Infect 2012;18:268-81.ArticlePubMed
- 3. Vardakas KZ, Rafailidis PI, Konstantelias AA, Falagas ME. Predictors of mortality in patients with infections due to multi-drug resistant Gram negative bacteria: the study, the patient, the bug or the drug? J Infect 2013;66:401-14.ArticlePubMed
- 4. Magill SS, Edwards JR, Bamberg W, Beldavs ZG, Dumyati G, Kainer MA, et al. Multistate point-prevalence survey of health care-associated infections. N Engl J Med 2014;370:1198-208.ArticlePubMedPMC
- 5. Levy Hara G, Kanj SS, Pagani L, Abbo L, Endimiani A, Wertheim HF, et al. Ten key points for the appropriate use of antibiotics in hospitalised patients: a consensus from the Antimicrobial Stewardship and Resistance Working Groups of the International Society of Chemotherapy. Int J Antimicrob Agents 2016;48:239-46.ArticlePubMed
- 6. Levy-Hara G, Amábile-Cuevas CF, Gould I, Hutchinson J, Abbo L, Saxynger L, et al. “Ten commandments” for the appropriate use of antibiotics by the practicing physician in an outpatient setting. Front Microbiol 2011;2:230. PubMedPMC
- 7. Pulcini C, Defres S, Aggarwal I, Nathwani D, Davey P. Design of a ‘day 3 bundle’ to improve the reassessment of inpatient empirical antibiotic prescriptions. J Antimicrob Chemother 2008;61:1384-8.ArticlePubMedPDF
- 8. Pulcini C, Botelho-Nevers E, Dyar OJ, Harbarth S. The impact of infectious disease specialists on antibiotic prescribing in hospitals. Clin Microbiol Infect 2014;20:963-72.ArticlePubMed
- 9. Schmitt S, McQuillen DP, Nahass R, Martinelli L, Rubin M, Schwebke K, et al. Infectious diseases specialty intervention is associated with decreased mortality and lower healthcare costs. Clin Infect Dis 2014;58:22-8.ArticlePubMedPDF
- 10. Kim JH, Hong SK, Kim KC, Lee MG, Lee KM, Jung SS, et al. Influence of full-time intensivist and the nurse-to-patient ratio on the implementation of severe sepsis bundles in Korean intensive care units. J Crit Care 2012;27:414. e11-21.Article
- 11. Kwak SH, Jeong CW, Lee SH, Lee HJ, Koh Y. Current status of intensive care units registered as critical care subspecialty training hospitals in Korea. J Korean Med Sci 2014;29:431-7.ArticlePubMedPMC
- 12. Dellinger RP, Levy MM, Rhodes A, Annane D, Gerlach H, Opal SM, et al. Surviving Sepsis Campaign: international guidelines for management of severe sepsis and septic shock, 2012. Intensive Care Med 2013;39:165-228.ArticlePubMedPMC
- 13. Acute Respiratory Distress Syndrome Network; Brower RG, Matthay MA, Morris A, Schoenfeld D, Thompson BT, et al. Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. N Engl J Med 2000;342:1301-8.ArticlePubMed
- 14. Knaus WA, Draper EA, Wagner DP, Zimmerman JE. APACHE II: a severity of disease classification system. Crit Care Med 1985;13:818-29.ArticlePubMed
- 15. Vincent JL, Moreno R, Takala J, Willatts S, De Mendonça A, Bruining H, et al. The SOFA (Sepsis-related Organ Failure Assessment) score to describe organ dysfunction/failure. On behalf of the Working Group on Sepsis-Related Problems of the European Society of Intensive Care Medicine. Intensive Care Med 1996;22:707-10.ArticlePubMed
- 16. Perkins NJ, Schisterman EF. The inconsistency of “optimal” cutpoints obtained using two criteria based on the receiver operating characteristic curve. Am J Epidemiol 2006;163:670-5.ArticlePubMedPMCPDF
- 17. Turner RB, Valcarlos E, Won R, Chang E, Schwartz J. Impact of infectious diseases consultation on clinical outcomes of patients with Staphylococcus aureus bacteremia in a community health system. Antimicrob Agents Chemother 2016;60:5682-7.ArticlePubMedPMC
- 18. Bai AD, Showler A, Burry L, Steinberg M, Ricciuto DR, Fernandes T, et al. Impact of infectious disease consultation on quality of care, mortality, and length of stay in Staphylococcus aureus bacteremia: results from a large multicenter cohort study. Clin Infect Dis 2015;60:1451-61.ArticlePubMedPDF
- 19. Tedeschi S, Trapani F, Giannella M, Cristini F, Tumietto F, Bartoletti M, et al. An antimicrobial stewardship program based on systematic infectious disease consultation in a rehabilitation facility. Infect Control Hosp Epidemiol 2017;38:76-82.ArticlePubMed
- 20. Hamandi B, Husain S, Humar A, Papadimitropoulos EA. Impact of infectious disease consultation on the clinical and economic outcomes of solid organ transplant recipients admitted for infectious complications. Clin Infect Dis 2014;59:1074-82.ArticlePubMedPDF
- 21. Choi SH, Cho SY, Park JH, Chung JW. Impact of infectious-disease specialist consultations on outcomes of Staphylococcus aureus bacteremia in a hospital with a low volume of patients with S. aureus bacteremia. J Infect 2011;62:181-5.ArticlePubMed
- 22. Howard P, Pulcini C, Levy Hara G, West RM, Gould IM, Harbarth S, et al. An international cross-sectional survey of antimicrobial stewardship programmes in hospitals. J Antimicrob Chemother 2015;70:1245-55.ArticlePubMedPDF
- 23. Holloway KA, Rosella L, Henry D. The Impact of WHO essential medicines policies on inappropriate use of antibiotics. PLoS One 2016;11:e0152020.ArticlePubMedPMC
- 24. Saunderson RB, Gouliouris T, Nickerson EK, Cartwright EJ, Kidney A, Aliyu SH, et al. Impact of routine bedside infectious disease consultation on clinical management and outcome of Staphylococcus aureus bacteraemia in adults. Clin Microbiol Infect 2015;21:779-85.ArticlePubMedPMC
- 25. Robinson JO, Pozzi-Langhi S, Phillips M, Pearson JC, Christiansen KJ, Coombs GW, et al. Formal infectious diseases consultation is associated with decreased mortality in Staphylococcus aureus bacteraemia. Eur J Clin Microbiol Infect Dis 2012;31:2421-8.ArticlePubMed
- 26. Fries BL, Licitra C, Crespo A, Akhter K, Busowski MT, Salazar D, et al. Infectious diseases consultation and the management of Staphylococcus aureus bacteremia. Clin Infect Dis 2014;58:598-9.ArticlePubMedPDF
References
Figure & Data
References
Citations

- Impact of a Pro-Active Infectious Disease Consultation on the Management of a Multidrug-Resistant Organisms Outbreak in a COVID-19 Hospital: A Three-Months Quasi-Experimental Study
Davide Fiore Bavaro, Nicolò De Gennaro, Alessandra Belati, Lucia Diella, Roberta Papagni, Luisa Frallonardo, Michele Camporeale, Giacomo Guido, Carmen Pellegrino, Maricla Marrone, Alessandro Dell’Erba, Loreto Gesualdo, Nicola Brienza, Salvatore Grasso, Gi
Antibiotics.2023; 12(4): 712. CrossRef - The role of infectious disease consultations in the management of patients with fever in a long-term care facility
Soo-youn Moon, Kyoung Ree Lim, Jun Seong Son, Ali Amanati
PLOS ONE.2023; 18(9): e0291421. CrossRef - Impact of infectious diseases consultation among patients with infections caused by gram-negative rod bacteria: a systematic literature review and meta-analysis
Shinya Hasegawa, Satoshi Kakiuchi, Joseph Tholany, Takaaki Kobayashi, Alexandre R. Marra, Marin L. Schweizer, Riley J. Samuelson, Hiroyuki Suzuki
Infectious Diseases.2022; 54(8): 618. CrossRef - Infectious Disease Consults of Pseudomonas aeruginosa Bloodstream Infection and Impact on Health Outcomes
Swetha Ramanathan, Fritzie S Albarillo, Margaret A Fitzpatrick, Katie J Suda, Linda Poggensee, Amanda Vivo, Martin E Evans, Makoto Jones, Nasia Safdar, Chris Pfeiffer, Bridget Smith, Geneva Wilson, Charlesnika T Evans
Open Forum Infectious Diseases.2022;[Epub] CrossRef - Timeliness of Infectious Diseases Referral and Inappropriate Antibiotic Usage Post-Referral in an Asian Tertiary Hospital
Liang En Wee, Aidan Lyanzhiang Tan, Limin Wijaya, Maciej Piotr Chlebicki, Julian Thumboo, Ban Hock Tan
Tropical Medicine and Infectious Disease.2019; 4(4): 137. CrossRef

 KSCCM
KSCCM
 PubReader
PubReader ePub Link
ePub Link Cite
Cite