Abstract
- Extracorporeal membrane oxygenation support can extend the duration of cardiopulmonary resuscitation, but neurologic complications may develop. Cardiac arrest is a fairly common complication following severe intracranial hemorrhage; this complication is encountered both out-of-hospital and in-hospital with variable frequency. To prevent cerebral complications, to detect the cause of cardiac arrest, and to guide further treatment, early neuroimaging study is needed. Herein, we report a case of intracranial hemorrhage identified after extracorporeal cardiopulmonary resuscitation, in which the cause of the hemorrhage was not clear.
-
Keywords: cardiopulmonary resuscitation; extracorporeal membrane oxygenation; intracranial hemorrhage
Despite recent advances in cardiopulmonary resuscitation (CPR), few patients could achieve return of spontaneous circulation (ROSC) with conventional CPR, and the outcome after cardiac arrest (CA) is still poor. Pretto et al.[1] and Safar et al.[2] reported the effectiveness of emergency cardiopulmonary bypass for CPR in an animal model, and discussed the possibility of employing cardiopulmonary bypass as a CPR method. Martin et al.[3] reported that percutaneous cardiopulmonary bypass could be initiated in the emergency department. Since then, veno-arterial extracorporeal membrane oxygenation (ECMO) has been performed in emergency departments (ED), and can be used for performing extracorporeal cardiopulmonary resuscitation (ECPR) in patients with out-of-hospital cardiac arrest (OHCA).[3,4] Recently, Chen et al.[5] reported that ECPR is superior to conventional CPR in in-hospital cardiac arrest patients. Although there is no sufficient evidence to support the efficacy of ECPR in OHCA patients, encouraging results have been obtained in several small case series.[4,6] However, there have been a few reports of complications in patients who underwent ECMO. Moreover, it is not clear when intracranial hemorrhage will occur after applying ECMO. We report a case of intracranial hemorrhage identified in the early stage after applying ECMO to support CPR. In this case, it was unclear whether this was the cause of cardiac arrest, or an ECMO-related neurologic complication.
Case Report
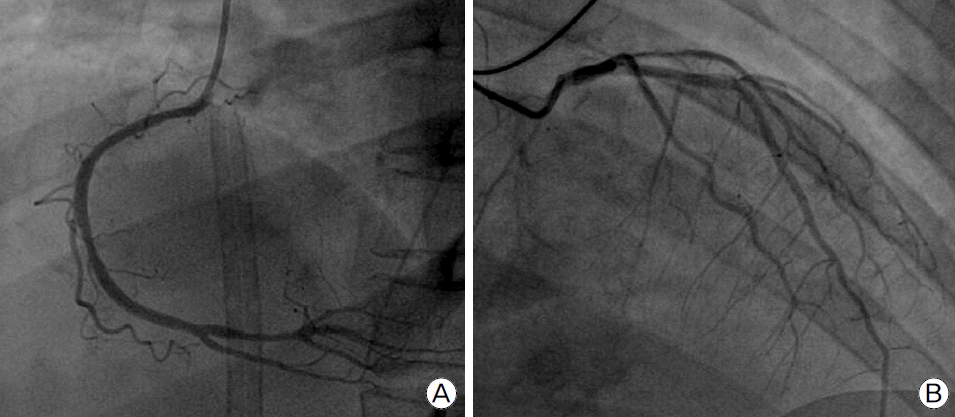
A 49-year-woman had been in her usual health, with hypertension, until approximately 6:30 on the morning of admission. When her husband found her collapsed, he called emergency medical services (EMS). Bystander CPR was performed by her husband as per EMS pre-arrival instructions. On examination by EMS personnel at 6:40 a.m., she was unresponsive. An automated external defibrillator (AED) monitor revealed ventricular fibrillation (VF). CPR was begun, shock was administered by an AED, and the patient was moved to an ambulance. During transfer to our ED, resuscitation efforts were continued without interruption. The ambulance arrived at the hospital at 7:05 a.m. On arrival, the patient was in cardiac arrest with refractory VF, and was pulseless. Her pupils were fully dilated (at about 7 mm) and unresponsive to light. CPR was performed immediately. During 15 minutes of resuscitation, the trachea was intubated. Amiodarone was administered intravenously, and seven direct-current countershocks were administered. Resolution of the VF failed to be achieved, so the decision was made to use extracorporeal membrane oxygenation (ECMO) for cardiopulmonary life support in our ED at 7:20 a.m. VA ECMO catheters were inserted into both femoral vessels for resuscitation at 7:48 a.m. The pump flow was set to 3.0 L/min, with a fraction of inspired oxygen of 1.0. Sustained ROSC was dramatically achieved at 7:50 a.m. Unfractionated heparin (3000 IU) as a bolus dose and 800 IU per hour by continuous infusion was used for anticoagulation. A 12-lead ECG showed sinus rhythm at 88 beats per minute; ST-segment depression of 2 to 3 mm in leads V1 through V4. The results of a complete blood count showed a WBC count of 19,200/μl, hemoglobin of 13.4 g/dl and platelet count of 223,000/μl. Her activated partial thromboplastin time (aPTT) was 48.8 sec (referece range, 23 to 41), and prothrombin time (PT) were within the normal limits. Arterial blood gas analysis (ABGA) in room air showed severe hypoxia, hypercapnia and acidosis: pH of 6.80, oxygen partial pressure (PaO2) of 23 mmHg, and carbon dioxide partial pressure (PaCO2) of 112 mmHg. Troponin-I was 0.18 ng/ml (reference range, 0 to 0.04). Serum glucose was 419 mg/dl. Aspartate aminotransferase and alanine aminotransferase were 95 IU/L and 77 IU/L, respectively. Blood levels of electrolytes, calcium, magnesium, total protein, creatinine, and albumin were normal. Prompt percutaneous coronary intervention was performed for presumed acute myocardial infarction (AMI). Coronary angiography (CAG) was performed at 8:20 a.m., revealing 40% flow limiting coronary stenoses in the mid to distal left anterior descending artery, and diffuse spasm of all three coronary arteries (Fig. 1). In the angiography room, her pupil size had returned to 3 mm, and pupils were sluggishly responsive to light. Unfortunately, brain computed tomography (CT), checked at 10:10 a.m. before post-resuscitation treatment, showed an intraventricular hemorrhage in both temporal horns, hemorrhage in the medial side of both frontal lobes, and severe brain swelling (Fig. 2A). However, a brain CT angiogram showed nonspecific findings in both internal carotid arteries, anterior cerebral arteries, middle cerebral arteries, and posterior cerebral arteries (Fig. 2B and 2C). Heparin was replaced by nafamostat as anticoagulant, and an anti-platelet agent was not used. After the family agreed to a “do not resuscitate” order, the patient expired the next morning.
Discussion
The case of this patient highlights two ECPR related major issues. One is intracranial complications of ECMO, and the other is cardiac abnormalities as a result of intracranial hemorrhage.
Although ECMO can improve the survival of patients with several severe conditions, there is a significant level of morbidity associated with the performance of this intervention. Some small series testing ECMO have reported complications associated with the use of ECMO in cardiogenic shock and cardiac arrest. However, the exact rates of these complications are not well understood, as they vary in frequency between series, in part due to the small sample sizes.[7–10] Neurological injuries, mainly intracranial hemorrhage and cerebral infarction are feared complications of ECMO that result in increased patient morbidity and risk of death. This can be attributed both to pre-ECMO asphyxia or hypotension and to the ECMO procedure itself, coagulopathy, and systemic heparinization.[11] To prevent or reduce cerebral damage and to guide further treatment, early diagnosis is needed. However, neurological evaluation of the patients on ECMO is often difficult because of sedation and pharmacologic paralysis. For adults and children, the methods available for diagnosis of cerebral injury while under ECMO therapy are limited. In a previous study, intracranial hemorrhage occurred in 7.4% of all ECMO-treated patients, while 36% of those patients survived to hospital discharge.[12] However, few studies have been performed on the pediatric and adult populations, making the reported frequency of intracranial complications during ECMO likely to be less reliable.[13,14] In addition, the time at which the intracranial hemorrhage occurs is not well known.
Among OHCA patients, between 4–16.2% have an arrest related to subarachnoid hemorrhage (SAH).[15,16] Proposed mechanisms for cardiac arrest in SAH patients include cardiac arrhythmias, respiratory arrest and cerebral herniation due to raised intracranial pressure. Cardiac abnormalities as a result of intracranial hemorrhage are likely to be the consequence of an adrenergic storm in the setting of neurologic disorders. The initial rhythm observed in those patients was most commonly pulseless electrical activity (PEA), with proportions ranging from 43% to 63%, and asystole, ranging from 44% to 57% in OHCA.[15–17] However, VF occurred only in 0 to 18%.[15–17] Various neurological disorders, most importantly SAH, were reported to have an electrographic presentation resembling the characteristic features of AMI in the form of repolarization abnormalities (e.g. ST-segment elevation).[18] The incidence of SAH was 15% to 20% in patients without a vascular lesion on cerebral angiography, and up to 24% of all SAH patients with a negative on their initial angiography were found to have an aneurysm on a repeated angiography.[19] Although vascular cause was ruled out by the CT angiogram, we could not exclude a microaneurysm in this case.
Neurotoxicity of the contrast media (CM) can sometimes mimic SAH, i.e., by leakage of CM into the subarachnoid space.[20] Although SAH is one of the major bleeding events that can but rarely occur after CAG, extravasation of the CM by disruption of the blood-brain barrier after CAG can sometimes mimic acute SAH on a brain CT.
In this case, initial ventricular fibrillation rhythm, ST-segment depression on a postresuscitation ECG, and cardiac enzyme elevation did not show evidence of the presence of an acute culprit lesion for myocardial infarction on the coronary angiography. However, the results of the coronary angiography could not rule out coronary artery vasospasm as the cause of her collapse. Diffuse spasms of all three coronary arteries could be interpreted as a consequence of coronary vasospasms due to intracranial hemorrhage. A vascular lesion as the cause of intracranial hemorrhage was not found on the brain CT angiogram. It is not certain whether ECMO-related neurologic complications were the cause of the intracranial hemorrhage, because brain CT was not checked immediately after ECMO insertion, but was delayed by about two and a half hours.
In summary, we present a case where the pathophysiology of intracranial hemorrhage after ECPR was not clear. Therefore, in ECPR patients, several etiologies should be considered for finding the cause of intracranial hemorrhage, and a brain CT should be obtained as early as possible after ECMO insertion.
NOTES
-
No potential conflict of interest relevant to this article was reported.
Fig. 1.Angiographic findings of the coronary arteries. (A, B) 40% flow limiting coronary stenoses in the mid to distal left anterior descending artery, and diffuse spasm of all three coronary arteries.
Fig. 2.Brain CT image shows an intraventricular hemorrhage in both temporal horns, hemorrhage in the frontal lobe, and severe brain swelling (A). The brain CT angiogram reveals no evidence of aneurysm in the major intracranial vessels (B, C).

References
- 1). Pretto E, Safar P, Saito R, Stezoski W, Kelsey S. Cardiopulmonary bypass after prolonged cardiac arrest in dogs. Ann Emerg Med 1987;16:611-9.ArticlePubMed
- 2). Safar P. Cerebral resuscitation after cardiac arrest: a review. Circulation 1986;74(6 Pt 2):IV138-53.PubMed
- 3). Martin GB, Rivers EP, Paradis NA, Goetting MG, Morris DC, Nowak RM. Emergency department cardiopulmonary bypass in the treatment of human cardiac arrest. Chest 1998;113:743-51.ArticlePubMed
- 4). Nagao K, Hayashi N, Kanmatsuse K, Arima K, Ohtsuki J, Kikushima K, et al. Cardiopulmonary cerebral resuscitation using emergency cardiopulmonary bypass, coronary reperfusion therapy and mild hypothermia in patients with cardiac arrest outside the hospital. J Am Coll Cardiol 2000;36:776-83.ArticlePubMed
- 5). Chen YS, Chao A, Yu HY, Ko WJ, Wu IH, Chen RJ, et al. Analysis and results of prolonged resuscitation in cardiac arrest patients rescued by extracorporeal membrane oxygenation. J Am Coll Cardiol 2003;41:197-203.ArticlePubMed
- 6). Kagawa E, Inoue I, Kawagoe T, Ishihara M, Shimatani Y, Kurisu S, et al. Assessment of outcomes and differences between in- and out-of-hospital cardiac arrest patients treated with cardiopulmonary resuscitation using extracorporeal life support. Resuscitation 2010;81:968-73.ArticlePubMed
- 7). Bakhtiary F, Keller H, Dogan S, Dzemali O, Oezaslan F, Meininger D, et al. Venoarterial extracorporeal membrane oxygenation for treatment of cardiogenic shock: clinical experiences in 45 adult patients. J Thorac Cardiovasc Surg 2008;135:382-8.ArticlePubMed
- 8). Hsu PS, Chen JL, Hong GJ, Tsai YT, Lin CY, Lee CY, et al. Extracorporeal membrane oxygenation for refractory cardiogenic shock after cardiac surgery: predictors of early mortality and outcome from 51 adult patients. Eur J Cardiothorac Surg 2010;37:328-33.ArticlePubMed
- 9). Lamarche Y, Chow B, Bédard A, Johal N, Kaan A, Humphries KH, et al. Thromboembolic events in patients on extracorporeal membrane oxygenation without anticoagulation. Innovations (Phila) 2010;5:424-9.ArticlePubMed
- 10). Loforte A, Montalto A, Ranocchi F, Della Monica PL, Casali G, Lappa A, et al. Peripheral extracorporeal membrane oxygenation system as salvage treatment of patients with refractory cardiogenic shock: preliminary outcome evaluation. Artif Organs 2012;36:E53-61.ArticlePubMed
- 11). Lidegran MK, Mosskin M, Ringertz HG, Frenckner BP, Lindén VB. Cranial CT for diagnosis of intracranial complications in adult and pediatric patients during ECMO: Clinical benefits in diagnosis and treatment. Acad Radiol 2007;14:62-71.ArticlePubMed
- 12). Hervey-Jumper SL, Annich GM, Yancon AR, Garton HJ, Muraszko KM, Maher CO. Neurological complications of extracorporeal membrane oxygenation in children. J Neurosurg Pediatr 2011;7:338-44.ArticlePubMed
- 13). Dalton HJ, Day SE. Neurologic outcome following ECLS in pediatric patients with respiratory or cardiac failure. In: ECMO extracorporeal cardiopulmonary support in critical care. 3rd ed. In: Van Meurs K, , Lally KP, , Peek G, , . Ann Arbor (MI), Extracorporeal Life Support Organization. 2005, pp 383-92.
- 14). Amigoni A, Pettenazzo A, Biban P, Suppiej A, Freato F, Zaramella P, et al. Neurologic outcome in children after extracorporeal membrane oxygenation: prognostic value of diagnostic tests. Pediatr Neurol 2005;32:173-9.ArticlePubMed
- 15). Kürkciyan I, Meron G, Sterz F, Domanovits H, Tobler K, Laggner AN, et al. Spontaneous subarachnoid haemorrhage as a cause of out-of-hospital cardiac arrest. Resuscitation 2001;51:27-32.ArticlePubMed
- 16). Inamasu J, Miyatake S, Tomioka H, Suzuki M, Nakatsukasa M, Maeda N, et al. Subarachnoid haemorrhage as a cause of out-of-hospital cardiac arrest: a prospective computed tomography study. Resuscitation 2009;80:977-80.ArticlePubMed
- 17). Mitsuma W, Ito M, Kodama M, Takano H, Tomita M, Saito N, et al. Clinical and cardiac features of patients with subarachnoid haemorrhage presenting with out-of-hospital cardiac arrest. Resuscitation 2011;82:1294-7.ArticlePubMed
- 18). Huang CC, Huang CH, Kuo HY, Chan CM, Chen JH, Chen WL. The 12-lead electrocardiogram in patients with subarachnoid hemorrhage: early risk prognostication. Am J Emerg Med 2012;30:732-6.ArticlePubMed
- 19). Schwartz TH, Solomon RA. Perimesencephalic nonaneurysmal subarachnoid hemorrhage: review of the literature. Neurosurgery 1996;39:433-40.ArticlePubMed
- 20). Velden J, Milz P, Winkler F, Seelos K, Hamann GF. Nonionic contrast neurotoxicity after coronary angiography mimicking subarachnoid hemorrhage. Eur Neurol 2003;49:249-51.ArticlePubMed
Citations
Citations to this article as recorded by










 KSCCM
KSCCM
 PubReader
PubReader ePub Link
ePub Link Cite
Cite