Articles
- Page Path
- HOME > Acute Crit Care > Volume 36(3); 2021 > Article
-
Original Article
Pulmonary Impact of prone position on outcomes of COVID-19 patients with spontaneous breathing -
Mabrouk Bahloul1
 , Sana Kharrat1, Malek Hafdhi1, Anis Maalla2, Olfa Turki1, Kamilia Chtara1, Rania Ammar1, Basma Suissi2, Chokri Ben Hamida1, Hedi Chelly1, Khaiereddine Ben Mahfoudh2, Mounir Bouaziz1
, Sana Kharrat1, Malek Hafdhi1, Anis Maalla2, Olfa Turki1, Kamilia Chtara1, Rania Ammar1, Basma Suissi2, Chokri Ben Hamida1, Hedi Chelly1, Khaiereddine Ben Mahfoudh2, Mounir Bouaziz1 -
Acute and Critical Care 2021;36(3):208-214.
DOI: https://doi.org/10.4266/acc.2021.00500
Published online: August 12, 2021
1Department of Intensive Care, Habib Bourguiba University Hospital and Sfax University, Sfax, Tunisia
2Department of Radiology, Habib Bourguiba University Hospital and Sfax University, Sfax, Tunisia
- Corresponding author Mabrouk Bahloul Department of Intensive Care, Habib Bourguiba University Hospital, Sfax 3029, Tunisia Tel: +216-98-69-8267 Fax: +216-74-24-3427 E-mail: bahloulmab@yahoo.fr
Copyright © 2021 The Korean Society of Critical Care Medicine
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
-
Background
- In this study, we explored whether early application of the prone position (PP) can improve severe hypoxemia and respiratory failure in coronavirus disease 2019 (COVID-19) patients with spontaneous breathing.
-
Methods
- This is a prospective observational study of severe, critically ill adult COVID-19 patients admitted to the intensive care unit. All vital parameters were recorded in real time for all patients. Moreover, the results of chest computed tomography (CT), when available, were analyzed.
-
Results
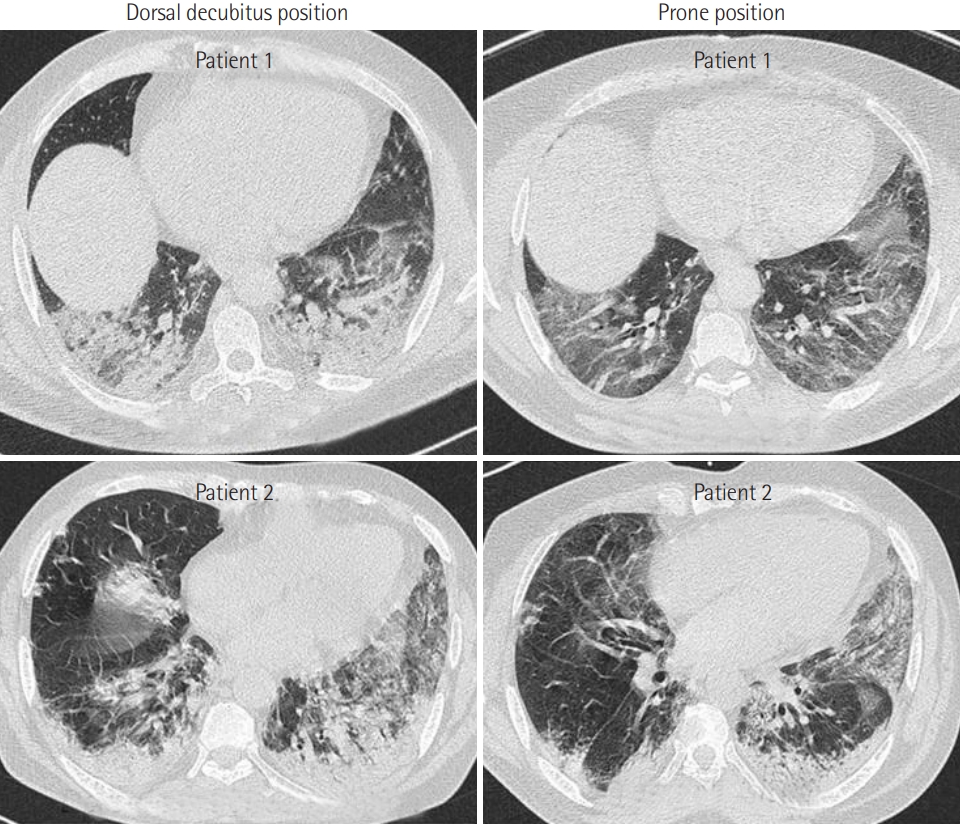
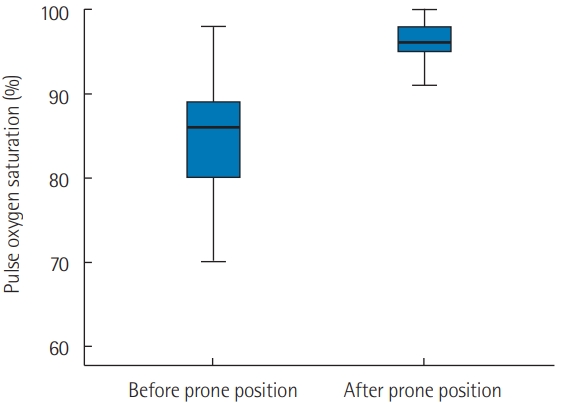
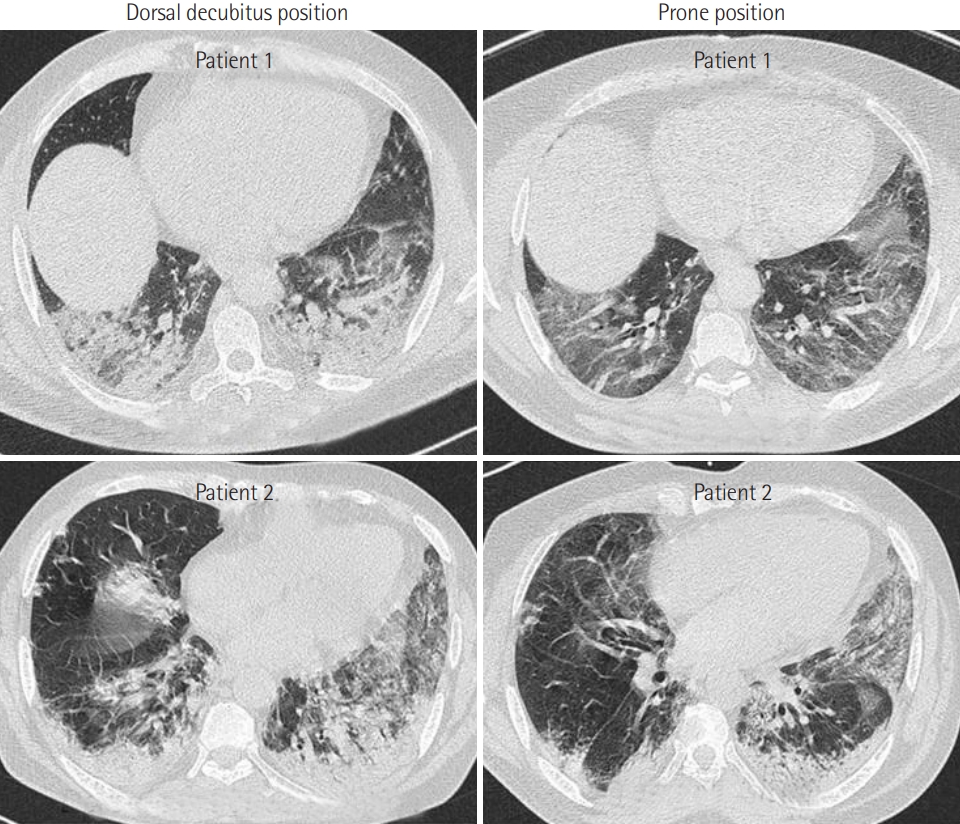
- PP was applied in 21 patients who were breathing spontaneously. The application of PP was associated with a significant increase in oxygen saturation measured by pulse oximetry (SpO2) from 82%±12% to 96%±3% (P<0.001) 1 hour later. Moreover, PP was associated with a significant reduction in respiratory rate from 31±10 to 21±4 breaths/min (P<0.001). Furthermore, the number of patients who exhibited signs of respiratory distress after PP was reduced from 10 (47%) to 3 (14%) (P=0.04). Early PP application also led to a clear improvement on CT imaging. It was not, however, associated with a reduction in mortality rate or in the use of invasive mechanical ventilation (P>0.05 for both).
-
Conclusions
- Our study confirmed that the early application of PP can improve hypoxemia and tachypnea in COVID-19 patients with spontaneous breathing. Randomized controlled trials are needed to confirm the beneficial effects of PP in COVID-19 patients with spontaneous breathing.
INTRODUCTION
MATERIALS AND METHODS
RESULTS
DISCUSSION
KEY MESSAGES
-
CONFLICT OF INTEREST No potential conflict of interest relevant to this article was reported.
-
AUTHOR CONTRIBUTIONS
Conceptualization: MB, SK, KBM, MB. Data curation: MB, SK, MH, OT, RA, BS, CBH, HC, KBM. Formal analysis: MB, SK, MH, AM, OT, KC, RA, BS, CBH. Methodology: MB, SK, AM, OT, KC, BS, CBH, HC, MB. Project administration: MB, BS, CBH. Visualization: MB, AM, HC, KBM. Writing–original draft: MB, MH, KC, CBH, HC, MB. Writing–review & editing: MB, SK, CBH, HC.
NOTES

| Variable | PP group (n=21) | PP-free group (n=17) | P-value |
|---|---|---|---|
| Demographic characteristics | |||
| Age (yr) | 61±10 | 60±12 | 0.68 |
| Obesity (BMI >30 kg/m2) | 10 | 4 | 0.18 |
| Diabetes mellitus | 7 | 7 | 0.74 |
| Arterial hypertension | 10 | 6 | 0.52 |
| COPD | 2 | 0 | 0.49 |
| Symptom on hospital admission | |||
| Dyspnea | 21 | 10 | 0.02 |
| Cough | 15 | 6 | 0.48 |
| Fever | 13 | 5 | 0.06 |
| Headache | 5 | 1 | 0.19 |
| Anosmia/ageusia | 3 | 2 | 0.60 |
| Vomiting | 3 | 2 | 0.60 |
| Abdominal pain | 5 | 0 | 0.05 |
| Diarrhea | 4 | 0 | 0.11 |
| Clinical findings on ICU admission | |||
| Respiratory rate/min | 31±10 | 27±7 | 0.16 |
| SpO2 under oxygen | 83±11 | 85±8 | 0.41 |
| Sign of respiratory fight | 10 | 5 | 0.32 |
| SAPS II score | 28.6±9.7 | 29.7±8.5 | 0.73 |
| SOFA score | 4.2±1.7 | 4.4±1.7 | 0.70 |
| pH | 7.44±0.03 | 7.43±0.03 | 0.45 |
| Blood bicarbonate (mmol/L) | 23.7±2.8 | 24.3±4.9 | 0.69 |
| PaO2/FiO2 ratio | 88±37 | 80±20 | 0.45 |
| GGOP >50% on chest CT scan | 15/17a | 8/8a | 0.45 |
| Treatment and outcome | |||
| High-flow nasal oxygen | 12 | 4 | 0.05 |
| Invasive mechanical ventilation | 9 | 4 | 0.30 |
| Corticosteroid | 21 | 17 | 1 |
| Diuretics | 19 | 10 | 0.05 |
| ICU stay (day) | 9.5±6.6 | 7.6±3.7 | 0.13 |
| Delay of invasive mechanical ventilation (day) | 8.1±5.8 | 6.0±3.2 | 0.17 |
| Survivor | 7 | 5 | 0.79 |
Values are presented as mean±standard deviation or number.
ICU: intensive care unit; PP: prone position; BMI: body mass index; COPD: chronic obstructive pulmonary disease; SpO2: oxygen saturation measured by pulse oximetry: SAPS: Simplified Acute Physiology Score; SOFA: Sequential Organ Failure Assessment; PaO2: arterial oxygen tension; FiO2: fractional inspired oxygen; GGOP: ground glass opacity portion; CT: computed tomography.
a Total number of patients with chest CT scan.
- 1. Worldometer. Coronavirus update [Internet]. Worldometer. 2020 [cited 2020 Jul 24]. Available from: https://www.worldometers.info/coronavirus/.
- 2. Vetter P, Vu DL, L’Huillier AG, Schibler M, Kaiser L, Jacquerioz F. Clinical features of covid-19. BMJ 2020;369:m1470. ArticlePubMed
- 3. Bahloul M, Ketata W, Lahyeni D, Mayoufi H, Kotti A, Smaoui F, et al. Pulmonary capillary leak syndrome following COVID-19 virus infection. J Med Virol 2021;93:94-6.ArticlePubMed
- 4. Ferrando C, Mellado-Artigas R, Gea A, Arruti E, Aldecoa C, Adalia R, et al. Awake prone positioning does not reduce the risk of intubation in COVID-19 treated with high-flow nasal oxygen therapy: a multicenter, adjusted cohort study. Crit Care 2020;24:597. ArticlePubMedPMC
- 5. Coppo A, Bellani G, Winterton D, Di Pierro M, Soria A, Faverio P, et al. Feasibility and physiological effects of prone positioning in non-intubated patients with acute respiratory failure due to COVID-19 (PRON-COVID): a prospective cohort study. Lancet Respir Med 2020;8:765-74.ArticlePubMedPMC
- 6. Caputo ND, Strayer RJ, Levitan R. Early self-proning in awake, non-intubated patients in the emergency department: a single ED’s experience during the COVID-19 pandemic. Acad Emerg Med 2020;27:375-8.ArticlePubMedPMC
- 7. Telias I, Katira BH, Brochard L. Is the prone position helpful during pontaneous breathing in patients with COVID-19? JAMA 2020;323:2265-7.ArticlePubMed
- 8. Le Gall JR, Lemeshow S, Saulnier F. A new Simplified Acute Physiology Score (SAPS II) based on a European/North American multicenter study. JAMA 1993;270:2957-63.ArticlePubMed
- 9. Ferreira FL, Bota DP, Bross A, Mélot C, Vincent JL. Serial evaluation of the SOFA score to predict outcome in critically ill patients. JAMA 2001;286:1754-8.ArticlePubMed
- 10. Hallifax RJ, Porter BM, Elder PJ, Evans SB, Turnbull CD, Hynes G, et al. Successful awake proning is associated with improved clinical outcomes in patients with COVID-19: single-centre high-dependency unit experience. BMJ Open Respir Res 2020;7:e000678. ArticlePubMed
- 11. Luks AM, Swenson ER. Pulse oximetry for monitoring patients with COVID-19 at home: potential pitfalls and practical guidance. Ann Am Thorac Soc 2020;17:1040-6.ArticlePubMedPMC
- 12. Venus K, Munshi L, Fralick M. Prone positioning for patients with hypoxic respiratory failure related to COVID-19. CMAJ 2020;192:E1532-7.ArticlePubMedPMC
- 13. Damarla M, Zaeh S, Niedermeyer S, Merck S, Niranjan-Azadi A, Broderick B, et al. Prone positioning of nonintubated patients with COVID-19. Am J Respir Crit Care Med 2020;202:604-6.ArticlePubMedPMC
- 14. Ponnapa Reddy M, Subramaniam A, Afroz A, Billah B, Lim ZJ, Zubarev A, et al. Prone positioning of nonintubated patients with coronavirus disease 2019: a systematic review and meta-analysis. Crit Care Med 2021;Apr 30; [Epub]. https://doi.org/10.1097/CCM.0000000000005086.Article
References
Figure & Data
References
Citations

- Awake prone positioning for patients with COVID-19-related respiratory failure: a systematic review and meta-analysis
Mara Graziani, Andrea Galeazzo Rigutini, Diletta Bartolini, Laura Traballi, Lorenzo Luzi, Rossana Regina, Francesco Bossi, Carla Caponi, Cecilia Becattini
Internal and Emergency Medicine.2024; 19(1): 147. CrossRef - Awake prone positioning for COVID-19 acute hypoxemic respiratory failure in Tunisia
Khaoula Ben Ismail, Fatma Essafi, Imen Talik, Najla Ben Slimene, Ines Sdiri, Boudour Ben Dhia, Takoua Merhbene
Acute and Critical Care.2023; 38(3): 271. CrossRef - Gravity-induced ischemia in the brain-and prone positioning for COVID-19 patients breathing spontaneously
J. Howard Jaster, Giulia Ottaviani
Acute and Critical Care.2022; 37(1): 131. CrossRef - Gravity-induced ischemia in the brain and prone positioning for COVID-19 patients breathing spontaneously: still far from the truth!
Mabrouk Bahloul, Sana Kharrat, Kamilia Chtara, Hedi Chelly, Chokri Ben Hamida, Mounir Bouaziz
Acute and Critical Care.2022; 37(1): 134. CrossRef - A Case of COVID-19 with Acute Exacerbation after Anti-Inflammatory Treatment
Yugo Ashino, Yoichi Shirato, Masahiro Yaegashiwa, Satoshi Yamanouchi, Noriko Miyakawa, Kokichi Ando, Yumiko Sakurada, Haorile Chagan Yasutan, Toshio Hattori
Reports.2022; 5(2): 24. CrossRef - Efficacy and safety of prone position in COVID-19 patients with respiratory failure: a systematic review and meta-analysis
Hyeon-Jeong Lee, Junghyun Kim, Miyoung Choi, Won-Il Choi, Joonsung Joh, Jungeun Park, Joohae Kim
European Journal of Medical Research.2022;[Epub] CrossRef
- Figure
- We recommend
- Related articles
-
- Factors related to lung function outcomes in critically ill COVID-19 patients in South Korea
- Gravity-induced ischemia in the brain and prone positioning for COVID-19 patients breathing spontaneously: still far from the truth!
- Gravity-induced ischemia in the brain-and prone positioning for COVID-19 patients breathing spontaneously

 KSCCM
KSCCM
 PubReader
PubReader ePub Link
ePub Link Cite
Cite