Abstract
- We experienced a case of venous vessel wall entrapment between the introducer needle and the guide wire during an attempt to perform right internal jugular vein (IJV) catheterization. The guide wire was introduced with no resistance but could not be withdrawn. We performed ultrasonography and C-arm fluoroscopy to confirm the entrapment location. We assumed the introducer needle penetrated the posterior vessel wall during the puncture and that only the guide wire entered the vein; an attempt to retract the wire pinched the vein wall between the needle tip and the guide wire. Careful examination with various diagnostic tools to determine the exact cause of entrapment is crucial for reducing catastrophic complications and achieving better outcomes during catheterization procedures.
-
Keywords: central venous catheter; complications; entrapment
Guidewire entrapment occurs rarely during central venous catheterization or during other procedures using the Seldinger technique. Entrapment can usually be resolved through manipulation of the wire, but sometimes surgical removal of the wire is required.[1,2] We experienced a rare case of guidewire entrapment during attempted central venous catheterization, and with the aid of various imaging tools, we were able to speculate on the location of the entrapment, form a hypothesis regarding its mechanism, and release the wire with no complications.
CASE REPORT
A fifty-four-year-old male patient, 161 cm in height and 62 kg in weight, was hospitalized for posterior thoracic spine fusion. He had been diagnosed with pulmonary tuberculosis nine years earlier and cured after treatment with medication for six months. His medical history was otherwise unremarkable. Preoperative laboratory tests, chest X-ray, and electrocardiography were within normal limits. Anesthesia was induced and maintained via total intravenous anesthesia, using propofol and remifentanil, to maintain the bispectral index (BIS; Aspect Medical Systems, Inc., Newton, MA, USA) in the range of 40–60. After the induction of anesthesia, the patient was placed in a 20° Trendelenburg position with the head turned to the left side for right internal jugular vein (IJV) cannulation (7 Fr Double-Lumen Central Venous Catheterization Set, Arrowguard Blue, Arrow International Inc., Reading, PA, USA). The patient’s right neck and shoulder were prepped with povidone-iodine. The finder needle (22-gauge syringe needle) was inserted at the apex of the triangle formed by the clavicular and sternal heads of the sternocleidomastoid muscle (SCM) and the clavicle, and regurgitation of venous blood was confirmed. The right IJV was punctured with the introducer needle at the same angle and depth (2.5 cm) as the finder needle. The syringe was disconnected and the guidewire was introduced with no resistance. When the 20 cm marker of the guidewire entered the introducer needle (postulated guidewire distance from the skin was about 17 cm, considering needle length), several premature atrial contractions (PACs) were observed on the electrocardiogram. We attempted to retract the guidewire a few centimeters in order to not provoke PACs and to see the marker to confirm the depth, but the wire was stuck and could not be withdrawn.
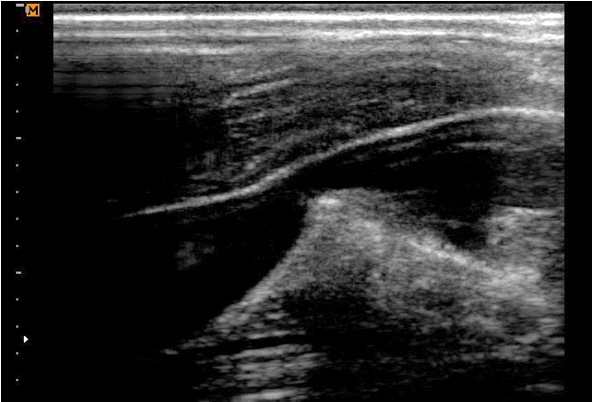
We then tried to remove the needle and guidewire altogether, but this was not possible. To confirm where the entrapment occurred, we scanned the patient’s neck area with ultrasonography (SonoAce R7, Samsung Medison Co., Ltd., Seoul, Korea), using an 8.0 MHz linear ultrasound probe (LN5-12, Samsung Medison Co., Ltd.) (Fig. 1). We were able to visualize that the guidewire and needle had gone through the posterior wall of the IJV, and the posterior vessel wall was being pulled with the wire and introducer needle, when we moved the guidewire and needle slightly back and forth.
We could not confirm whether the guidewire beyond that point was in the vessel or not, because the echo window was limited by the lung and clavicle. However, the length of the guidewire calculated to be in the body from the needle tip was longer than 13.6 cm (the 20 cm marker on the guidewire was just in the needle hub, and the length of the introducer was 6.35 cm), which was too long to be in the subcutaneous tissue or out of the vessel. Therefore, we decided to use a C-arm fluoroscope (OEC 9800 Mobile C-Arm, GE OEC Medical Systems, Inc., Salt Lake City, UT, USA) to confirm the location of the tip of the guidewire, and to confirm whether it was knotted or not (Fig. 2). Fluoroscopic imaging revealed that the guidewire tip was located almost in the center of the thorax, and it was not tangled.
The tip of the guidewire seemed to be located at the venoatrial junction or in the right heart. We initially suspected that the guidewire tip was somehow trapped in the tricuspid valve or the chordae, but in that case, the concurrent traction of the posterior vessel wall that was observed on the ultrasound imaging could not be explained. In addition, if that was the case, at least the introducer needle could have been removable. On fluoroscopic imaging, we confirmed that the guidewire was not fixed at the tip but floating freely, by slightly pulling and pushing the guide-wire and needle together. We speculated that the guidewire re-entered the vessel beyond the point that ultrasonography could visualize, and that the posterior vessel wall was stuck between the introducer needle and the guidewire. We subsequently manipulated the guidewire based on that hypothesis. Stabilizing the needle hub firmly, and holding the closest portion of the guidewire from the hub to maximize the transfer of the pushing force, we gently pushed the wire in, and it loosened. Then, we were able to remove the guidewire and needle. Using an 8.0 MHz linear ultrasound probe, we confirmed that there was no vascular injury except a small perivascular hematoma, and using transesophageal echocardiography (TEE), we also confirmed that there were no injuries to the tricuspid valve or chordae. No visible dents or bends were present in the guidewire. The patient was discharged after seven days, with no complications.
DISCUSSION
Guidewire entrapment occasionally occurs during central venous catheterization. Previous case reports have described several cases in which guidewires became entangled with inferior vena cava (IVC) filters, the tricuspid valve, or the right ventricle.[1,3–5] In some cases, the guidewire became knotted or tangled in subcutaneous tissue or the SCM.[2] In the case discussed here, the authors initially suspected knotting of the guidewire. Therefore, we used fluoroscopy to confirm the location of the tip of the guidewire, and found that it was located at the junction of the superior vena cava (SVC) and right atrium, and that it was not knotted. At that location, the wire might have been tangled with the chordae of the tricuspid valve or the valve itself, but as we moved it we could see the tip of the guidewire move freely about 0.5–1.0 cm on the fluoroscopic images. Additionally, if the guidewire entrapment occurred at the tricuspid valve or right ventricular level, it would pose no difficulty in withdrawing the introducer needle alone. Our case was not like that. If circumstances allowed, it would be ideal to perform TEE immediately after the tip of the wire was suspected to be located in the heart. The imaging tool for TEE is very expensive, and obtaining proper images and interpreting them requires substantial training. In addition, it may be difficult to introduce the TEE probe without contamination, when the neck area is prepped and the guidewire is inserted into the neck. Thus, it would not always be possible to perform TEE. In our case, we could rule out any valvular apparatus injury, and we confirmed that by performing TEE after retrieval of the guidewire.
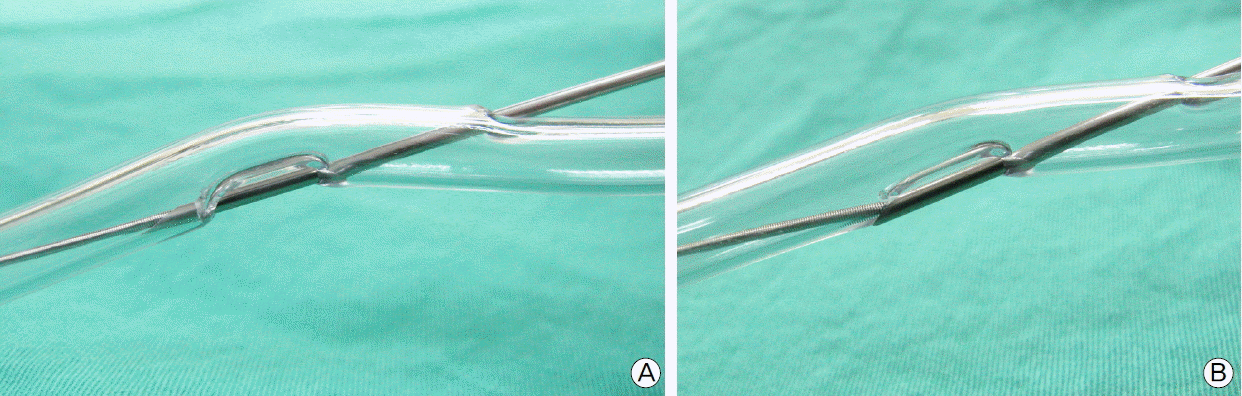
We postulated that the reason why retrieval of the guidewire was difficult in our case was because the venous vessel wall or perivascular tissue was stuck between the guidewire and the introducer needle. We assumed that the introducer needle somehow penetrated the posterior vessel wall during the puncture, and only the guidewire entered the vein. The needle tip was in the vein when we introduced the guidewire, but it seemed to be retracted several millimeters, with the needle tip contacting the vein wall at the external side. Then, as we tried to remove the guidewire the first time, the tip of the large bore introducer needle and the guidewire itself acted like a pincer, and the withdrawal force eventually bit in more tissue into a narrow space to make the situation much worse. We simulated the circumstances using an introducer needle, guidewire, and polyvinyl chloride (PVC) tubing (Fig. 3).
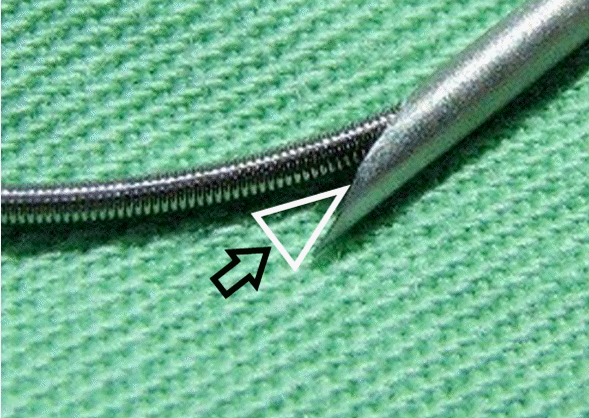
Our simulation allowed us to confirm our hypothesis, even though the thickness of the PVC tubing is much larger than the actual vein wall. Given that difference in thickness, it must be much easier for vein wall or perivascular tissue to become stuck between the needle and the guidewire (Fig. 4). With this hypothesis in mind, the authors tried to release the tissue by manipulating the guidewire, and after several attempts, it released. We removed the guidewire and needle, and evaluated the IJV and perivascular tissue with ultrasonography. There were no groove complications except a small perivascular hematoma.
There are some points to consider in order to reduce this type of complication. We lowered the patient’s head 20° to maximize the IJV cross-sectional area.[6] This maneuver may unintentionally increase the needle insertion angle against the vein, and it will make it easier for the needle to penetrate the posterior wall of the vessel. The bevel of the needle faced upward with the intention of facilitating blood regurgitation, and the angle of introducer needle was intentionally lowered after confirmation of the aspiration of blood, to facilitate the guidewire insertion. All of these maneuvers might have increased the possibility of the complication we experienced.
As we presented here, there is a rare possibility of guidewire entrapment caused by the mechanism described. Careful examination with various diagnostic tools to determine the exact cause of entrapment, and cautious consideration and ruling out of other possible causes of entrapment are crucial for reducing catastrophic complications and achieving better outcomes during catheterization procedures.
NOTES
-
Conflict of interest statement
We declare that we received no financial support and have no conflicts of interest regarding this case report.
Fig. 1.Ultrasonographic image shows posterior wall traction by pulling introducer needle and guidewire together.
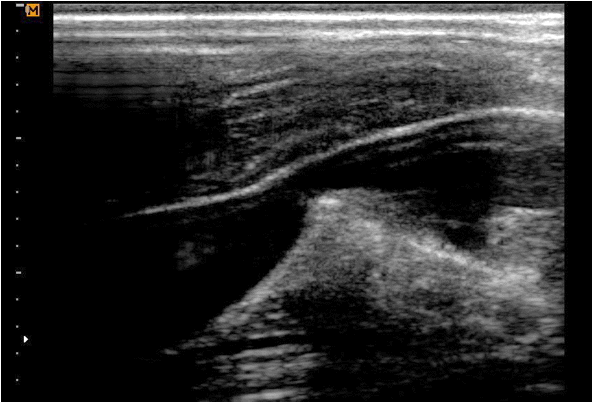
Fig. 2.Fluoroscopic image shows J-wire tip located in the thorax. Arrow: Tip of introducer needle.
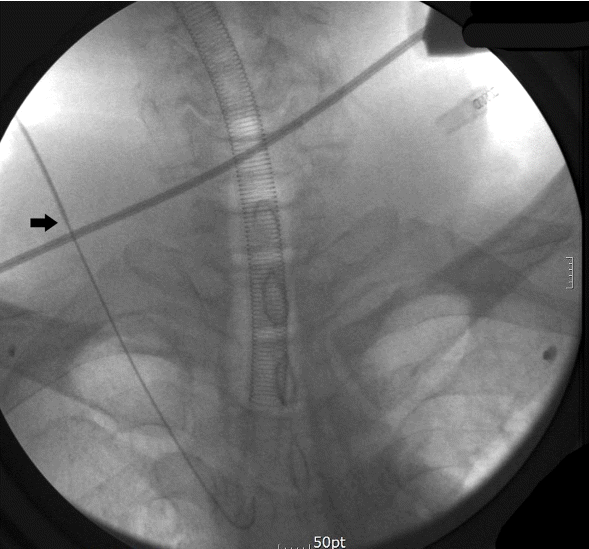
Fig. 3.Wall of PVC tubing stuck between introducer needle tip and guidewire. (A) shows possible initial location of needle tip, (B) shows needle tip pinching PVC tubing wall after retraction.

Fig. 4.Introducer needle and guidewire show pincer mechanism. Arrow indicates the area where the vessel wall may become stuck.

References
- 1). Koç F, Doğdu O, Sarli B, Kaya MG. [Tricuspid valve dysfunction associated with entrapment of the guide wire in the tricuspid valve during central venous catheterization]. Turk Kardiyol Dern Ars 2010;38:115-7.PubMed
- 2). Mastan M, Clothier PR, Ousta B, Deulkar U. Internal jugular venous cannulation complicated by J-tip guide wire entrapment. Br J Anaesth 2001;86:292-3.PubMed
- 3). Reddy S, Coba V, Horst M, Killu K. Ultrasound diagnosis of central line guidewire entrapment with an inferior vena cava filter. ICU Dir 2011;2:215-7.Article
- 4). Kang SH, Park WK, Do JY, Cho KH, Park JW, Yoon KW. J-tip guide wire entrapment within the heart during central venous catheterization. Hemodial Int 2012;16:438-40.ArticlePubMed
- 5). Hoda MQ, Das G, Mamsa KA, Salimullah H. Unusual site of guide-wire entrapment during central venous catheterization. J Pak Med Assoc 2006;56:139-41.PubMed
- 6). Marcus HE, Bonkat E, Dagtekin O, Schier R, Petzke F, Wippermann J, et al. The impact of Trendelenburg position and positive end-expiratory pressure on the internal jugular cross-sectional area. Anesth Analg 2010;111:432-6.ArticlePubMed
Citations
Citations to this article as recorded by











 KSCCM
KSCCM

 PubReader
PubReader ePub Link
ePub Link Cite
Cite