Abstract
- We incidentally discovered a case of deep neck infection during the treatment of a patient who presented with complaints of decreased consciousness, abdominal distension, and electrolyte imbalance. The patient had neither clinical symptoms nor radiologic findings indicative of deep neck infection; rather, the findings indicated intra-abdominal sepsis and adrenal crisis, for which the symptomatic treatment was provided. When the expected improvement was not observed, we retrospectively reviewed the patient’s test records and discovered deep neck infection. Empiric parenteral antibiotic therapy and infection source control, as appropriate for this patient, were administered within 72 hours of hospital admission, without which the condition may have proved fatal. In this report we discuss the best approach for the management of unresolved infectious disease and review the clinical features of deep neck infection.
-
Keywords: deep neck infection; sepsis
With recent advancements in antibiotic treatment and improvements in dental care, the incidence of deep neck infection is decreasing. However, deep neck infection can lead to fatal complications, such as upper airway obstruction, descending mediastinitis, and pleural empyema, with some studies reporting mortality rates of 50% in the presence of complications.[1] Quick diagnosis, appropriate antibiotic treatment, airway maintenance, and pus drainage are essential to prevent complications and save lives. We experienced a case of a patient with sepsis who was admitted for decreased consciousness, abdominal distension, and electrolyte imbalance; the patient was then incidentally diagnosed with deep neck infection and was successfully treated. This paper reviews the literature on the etiology, clinical presentation, treatment, and prognosis of deep neck infection and discusses the appropriate diagnosis and treatment for patients with unresolved sepsis.
CASE REPORT
A 77-year-old woman was admitted to the emergency department with the complaints of decreased consciousness and abdominal distension after undergoing brain magnetic resonance imaging (MRI) at another hospital. Brain MRI confirmed the absence of any specific disease that might have caused her decreased consciousness. The patient was receiving long-term treatment (through a private clinic) for hypertension and arthritis, although the exact regimen at the time of admission could not be verified. There were no other specific underlying diseases. The patient showed signs of moon face and was obese; at the time of admission, her vital signs were stable and there were no signs of fever. However, her pulse rate was increased to 110 beats per minute, and she showed signs of drowsy mental status, severe abdominal distension, diffuse abdominal tenderness, and rebound tenderness. There were no abnormal findings on neurological examination, but the patient complained of general weakness and dyspnea. Oxygen saturation measured using bedside pulse oximetry was 88%. Under the suspicion of a surgical emergency, an on-call surgeon performed an emergency examination. Laboratory and radiologic examinations as well as microbial cultures from blood and urine were processed. Laboratory findings included the following: hemoglobin, 12.3 g/dl; white blood cell (WBC) count, 24,850/ml (95.6% neutrophils), blood urea nitrogen, 43.7 mg/dl; creatinine, 0.96 mg/dl; sodium, 116 mEq/L; chloride, 84 mEq/L; potassium, 5.1 mEq/L; and C-reactive protein (CRP), 312.8 mg/L. The urine analysis result revealed no abnormalities, and there were no evidences suggesting urinary tract infection. Arterial blood gas analysis measurements taken after supplying oxygen through a nasal cannula revealed no abnormalities in the partial pressure of oxygen and carbon dioxide. Because her dyspnea improved, tests were performed without the administration of any additional respiratory treatments such as intubation. Chest radiography showed sub-segmental atelectasis in the right middle lung field (Fig. 1) but there was no evidence of heart murmur or lung crackle on auscultation and the breathing sound was clear. Abdominal radiography showed severe distension in the small and large intestines (Fig. 2) and abdominal computed tomography (CT) scan revealed severe distension of the intestine as well as constipation, although the exact location of the bowel obstruction could not be confirmed. The patient‘s medication regimen was confirmed to be a steroid, so a standard high-dose adrenocorticotropic hormone (ACTH) stimulation test was conducted, immediately followed by empirical intravenous injection of hydrocortisone. Afterward, the results of the ACTH stimulation test revealed baseline ACTH and cortisol levels of 19.68 pg/dl and 0.49 µg/dl, respectively; cortisol levels measured 20 and 40 minutes after injection of synacthen were 37.03 µg/dl and 43.62 µg/dl, respectively. These results allowed the possibility of primary adrenal insufficiency to be eliminated, while indicating secondary adrenal insufficiency; however, additional tests were not performed in consideration of the patient’s hospital course. Based on these results, the following approach was taken. First, because of the presence of abdominal distension and an elevated WBC count, we could not rule out the possibility of an intestinal obstruction. Therefore, the patient was fasted, a nasogastric tube and a rectal tube were placed, and a third-generation cephalosporin was given empirically. The patient was also placed under close surgical observation. Second, acute adrenal crisis was determined as the cause for the patient’s general weakness, decreased consciousness, and hyponatremia, for which intravenous hydrocortisone was injected and sodium replacement was performed.
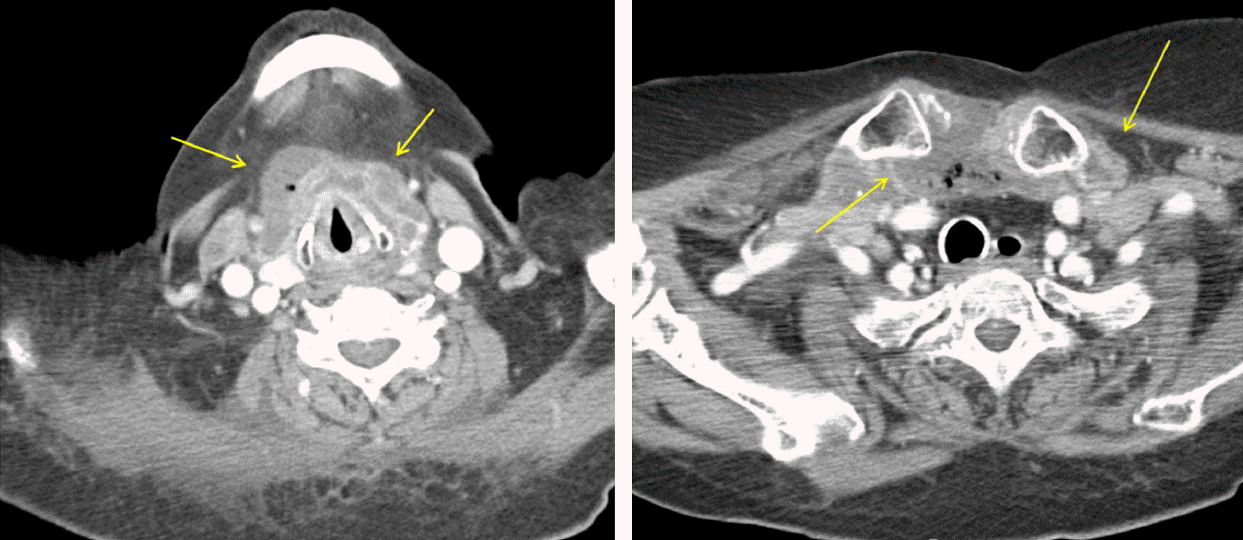
On the second day of hospitalization, large quantities of gas and stool were excreted through the rectal tube; signs of abdominal distension improved. On the third day of admission, signs of abdominal pain and distension were absent and vital signs were stable without fever. Therefore, the possibility of intestinal obstruction was completely ruled out, and the patient resumed oral intake. Laboratory test results revealed the following: hemoglobin, 9.6 g/dl; WBC count, 18,950/ml (86% neutrophils); sodium, 127 mEq/L; and CRP, 134 mg/L. Although the physical examination of the abdomen had improved, CRP level and WBC count were still elevated, and the patient’s mental status had not improved. Therefore, previous studies were re-examined; that day, the microorganism from the blood microbial culture performed at the time of admission was proven to be methicillin-susceptible Staphylococcus aureus (MSSA). So the antibiotic was switched from the existing third-generation cephalosporin to cefazolin. The abdominal CT scan taken at the time of admission was re-examined. As a result, an abscess, which had not been identified initially because our focus had been on the patient’s abdominal distension and pain, was discovered near the right sternoclavicular joint. The view that revealed the abscess had not originally been available on the abdominal CT scan, but the abscess could be incidentally discovered because the on-call resident had observed atelectasis in the right lung on the chest radiograph and requested that the abdominal CT scan continue up to the chest (Fig. 3). Deep neck infection was suspected, and clindamycin was added to the antibiotic regimen. A neck CT was immediately performed (Fig. 4) and revealed deep neck infection involving multiple neck spaces.
On the fifth day of admission, surgery for the deep neck infection was performed and irrigation and debridement of the involved deep neck spaces were performed. After surgery, the patient was admitted to the intensive care unit and received postoperative care. The vital signs immediately after surgery were stable, with good respiration. However, the patient’s WBC count increased to 50,700/ml (93% neutrophils), so the patient was closely observed for septic shock or sepsis-induced hypo-tension during treatment. Cefazolin and clindamycin were maintained, and care was taken to avoid pulmonary edema by retaining an appropriate amount of urine through the use of fluids and diuretics. On the first postoperative day, the patient remained drowsy. The results of laboratory tests showed the following: hemoglobin, 10.6 g/dl; WBC count, 32,520/ml (97% neutrophils); sodium, 138 mEq/L; chloride, 102 mEq/L; and potassium, 2.8 mEq/L; the WBC count was lower than that on the previous day. On the third postoperative day, the result of the pus culture that had been performed on the day of the surgery was proven to be MSSA strain previously detected in blood. On the fourth postoperative day, the patient’s WBC count was 12,750/ml (92.4% neutrophils) and CRP level was 34.14 mg/L. The patient became alert and oral intake was started. On the ninth postoperative day, the patient was moved to a general ward and frequent wound dressing was performed. The administration of antibiotics and hydrocortisone was continued using fluid and diuretics, and care was taken to avoid a positive fluid balance. Wound and blood microbial culture tests were negative for bacteria; however, the wound had substantial dead space and healing was slow. Frequent wet dressing and conservative treatment was provided. On the 48th postoperative day, the patient was discharged after wound healing had been complete, and she did not experience specific complications.
DISCUSSION
Deep neck infection refers to an abscess or cellulitis occurring in the potential space formed by the neck fascial planes. Infections mostly occur in the parapharyngeal, submandibular, lateral pharyngeal, and retropharyngeal spaces, and Ludwig‘s abscess is observed relatively frequently.[2–4] In cases of infection that invades multiple deep neck spaces, as in this case, mediastinal extension has been frequently reported.[5] Pain is the most common clinical symptom with deep neck infection, followed by odynophagia, neck swelling, and fever.[6] Before the development of antibiotics, infections of the pharynx and tonsils were the primary causes of deep neck infections. In the era of antibiotic therapy, the causes are increasingly being replaced by regional trauma following violence or dental infection.[2,6] The development of antibiotics and improvements in dental care have resulted in a reduced incidence of deep neck infections; however, these infection are now frequently observed in elderly individuals with systemic diseases.[3] The results of a recently conducted retrospective study have shown that Streptococcus viridians accounted for the bulk of associated pathogens [1,3,4]; a high proportion of Klebsiella pneumoniae was detected among Gram-negative bacteria,[1,3] while high proportions of Staphylococcus epidermidis and S. aureus were detected among Gram-positive bacteria.[4] For all patients with suspected deep neck infection, empirical antibiotics must be administered without waiting for culture results. Antibiotics effective against β-lactamase-resistant pathogens, such as penicillin plus a β-lactamase inhibitor and cefoxitin, cefuroxime, imipenem, and meropenem, are recommended. In addition, clindamycin or metronidazole should also be used in combination with one of the above agents, to cover for anaerobic bacteria, while gentamycin is recommended as cover for Klebsiella species in diabetic patients. Vancomycin is recommended as the initial empirical antibiotic for intravenous drug users and patients with immune dysfunction.[7] Treatment depends on quick suspicion and diagnosis, early surgical drainage, empirical antibiotic administration, and ensuring an airway. In particular, in the case of Ludwig’s angina, which occurs in the oral cavity and rapidly spreads to the supramylohyoid and inframylohyoid compartments, airway obstruction is frequently observed and a tracheostomy must be performed at an early stage.[2,4,7]
Our case showed clear signs of abdominal distension and pain, decreased consciousness, and an electrolyte imbalance at the time of admission. These signs and symptoms led the diagnosis toward a life-threatening surgical emergency and endocrine emergency; deep neck infection was not suspected. Moreover, specific findings such as erythema and swelling in the neck and sternum were not observed during visual inspection, while neck and sternum pain was not specifically checked because of the patient’s drowsy mental status and generalized pain. After admission and treatment according to the initial diagnosis, the expected outcome (reduction in WBC count and improvement in the level of consciousness) was not observed. Meanwhile, MSSA, which is almost never observed in blood culture tests in patients with intra-abdominal sepsis, was identified. At this point, the initial diagnosis was suspected to have been wrong, and the patient was re-examined. Subsequent physical examination revealed tenderness in the sternoclavicular joint. Had the resident who provided the initial care at the time of admission not requested that the abdominal CT continue all the way to the chest, the diagnosis of deep neck infection would have been delayed and the patient‘s clinical course would have been fatal. This patient had tachycardia, dyspnea, leukocytosis, and altered mental status at the time of admission, and therefore met the sepsis diagnostic criteria of the Surviving Sepsis Campaign guidelines published in 2012.[8] According to these guidelines, appropriate empirical antibiotics must be administered for patients with sepsis within 1 hour, and source control must be achieved within 12 hours. However, in this case, appropriate antibiotics were administered > 72 hours after admission, and control of the infection source was achieved 4 days later.
In conclusion, basic principles are important in assessing a patient, and a thorough physical examination in the emergency room, while considering all possibilities, is believed to be critical. In particular, in septic patients who do not show the expected outcome from the recommended treatment, it is important to set aside the initial diagnosis and explore other possibilities. Similarly, the suspicion of deep neck infection could be helpful for the treatment of elderly patients with systemic diseases.
Fig. 1.Chest radiography showed sub-segmental atelectasis in the right middle lung field.
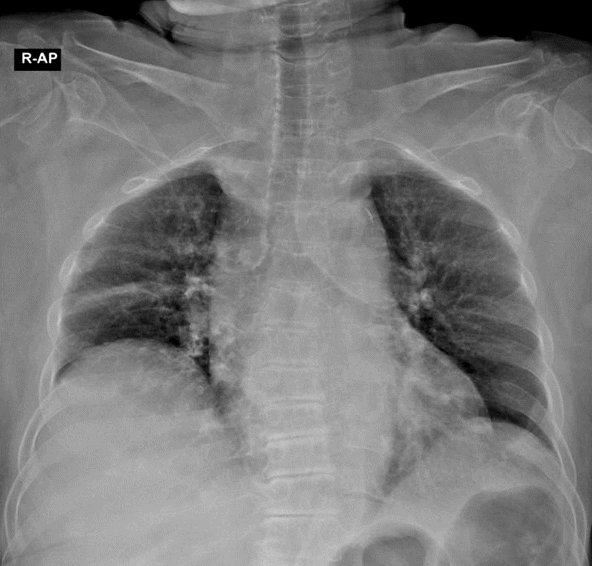
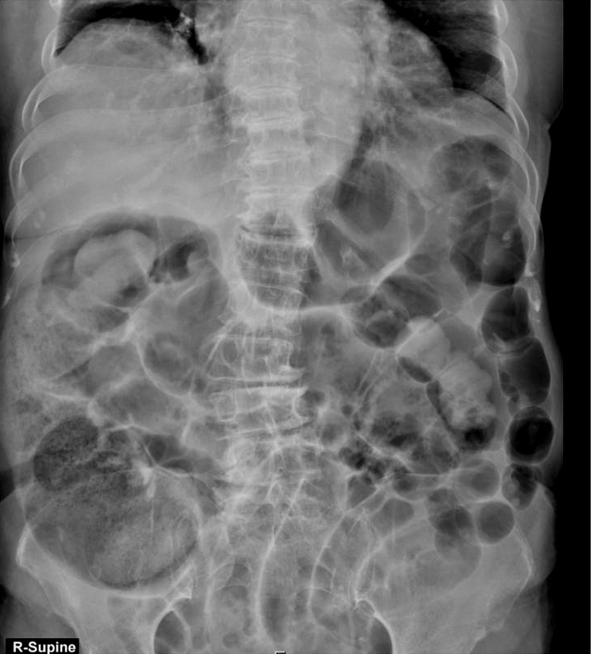
Fig. 2.Abdomen radiography showed severe distension in the small and large intestines.
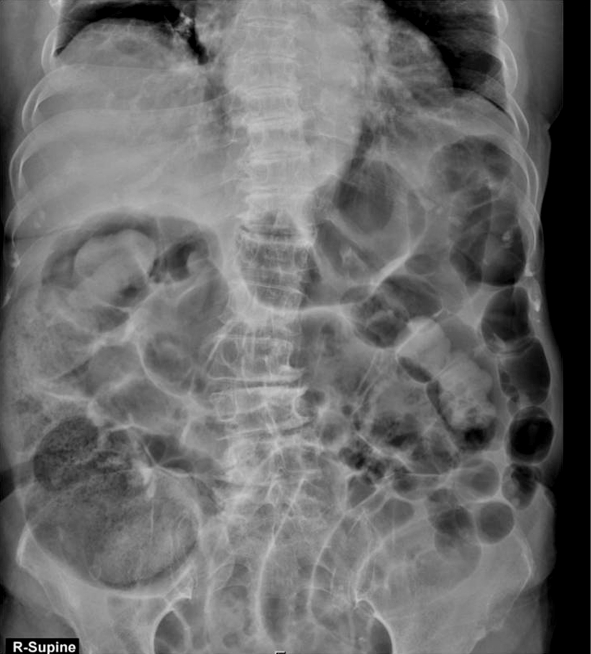
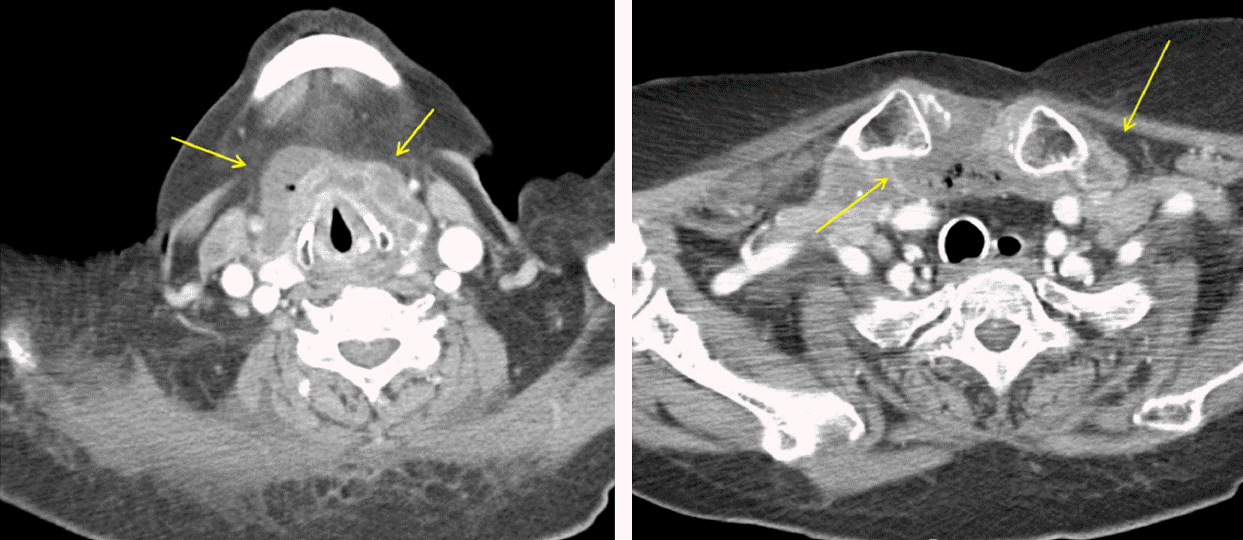
Fig. 3.Retrospective review of the abdominal CT scan revealed an abscess formation around the right sternoclavicular joint region.
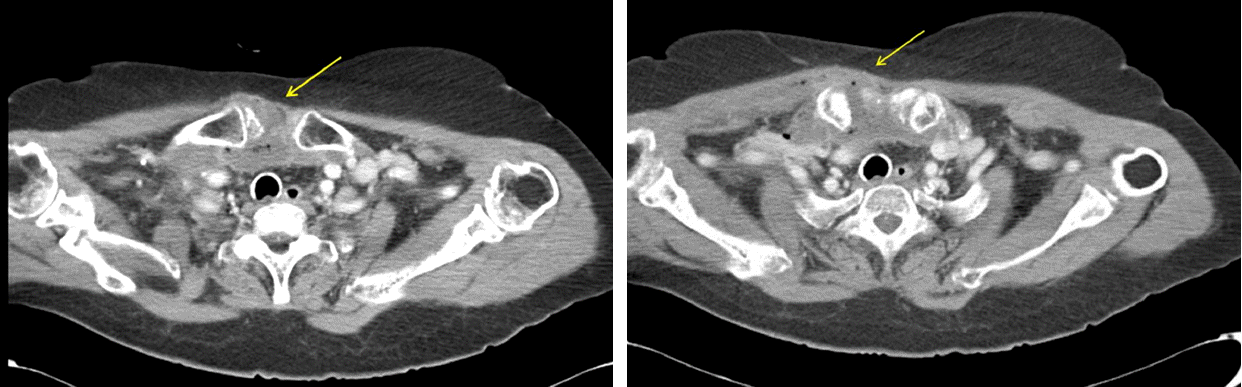
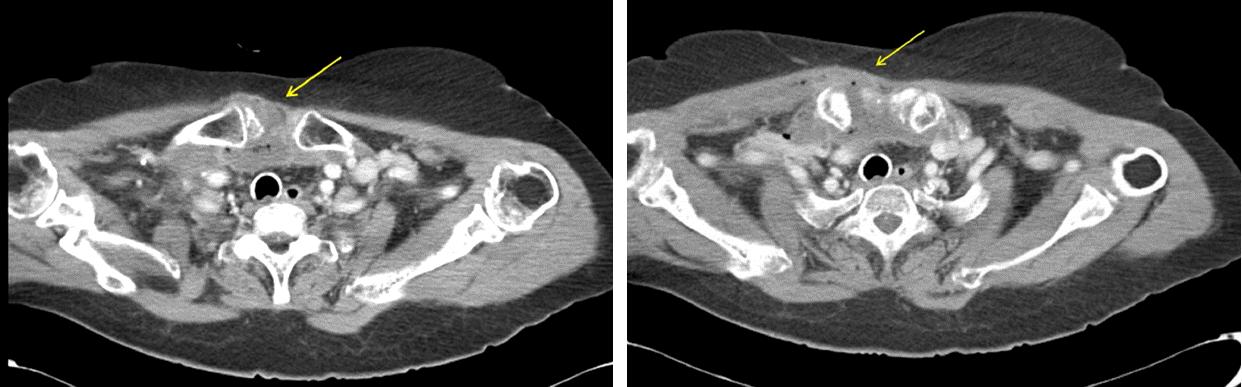
Fig. 4.Neck CT scan showed deep neck infection involving anterior neck, right sternoclavicular joint region, right supraclavicular fossa, left submandibular region, and left parapharyngeal space at the suprahyoid bone level.
References
- 1). Wang LF, Kuo WR, Tsai SM, Huang KJ. Characterizations of Life-Threatening Deep Cervical Space Infections: A Review of One Hundred Ninety-Six Cases. Am J Otolaryngol 2003;24:111-7.ArticlePubMed
- 2). Har-El G, Arowsty JH, Shaha A, Lucente FE. Changing trends in deep neck abscess. Oral Surg Oral Med Oral Pathol 1994;77:446-50.ArticlePubMed
- 3). Huang TT, Liu TC, Chen PR, Tseng FY, Yeh TH, Chen YS. Deep neck infection: analysis of 185 cases. Head Neck 2004;26:854-60.ArticlePubMed
- 4). Parhiscar A, Har-El G. Deep neck abscess: a retrospective review of 210 cases. Ann Otol Rhinol Laryngol 2001;110:1051-4.ArticlePubMed
- 5). Kang SK, Lee S, Oh HK, Kang MW, Na MH, Yu JH, et al. Clinical features of deep necck infections and predisposing factors for mediastinal extension. Korean J Thorac Cardiovasc Surg 2012;45:171-6.ArticlePubMedPMC
- 6). Sethi DS, Stanley RE. Deep neck abscesses-changing trends. J Laryngol Otol 1994;108:138-43.ArticlePubMed
- 7). Vieira F1, Allen SM, Stocks RM, Thompson JW. Thompson: Deep Neck Infections. Otolaryngol Clin N Am 2008;41:459-83. vii. Article
- 8). Dellinger RP, Levy MM, Rhodes A, Annane D, Gerlach H, Opal SM, et al. Surviving Sepsis Campaign: International Guidelines for Management of Severe Sepsis and Septic Shock: 2012. Intensive Care Med 2013;39:165-228.ArticlePubMedPMC
Citations
Citations to this article as recorded by









 KSCCM
KSCCM


 PubReader
PubReader ePub Link
ePub Link Cite
Cite