Abstract
- The incidence of acute leukemia during pregnancy is extremely rare, and often it is not easy to differentiate it from other diseases associated with pregnancy such as sepsis or hemorrhage. Pregnancy itself is not known to affect the natural course of leukemia; however, complications of leukemia like anemia, infections, and coagulopathy can adversely influence both the fetus and the mother. In this case, a pregnant patient misdiagnosed with septic shock and severe leukocytosis was correctly diagnosed with acute myeloid leukemia after surgical delivery.
-
Keywords: acute myeloid leukemia; intensive care unit; pregnancy
The occurrence of leukemia during pregnancy is very rare with a frequency of one in 75,000–100,000 pregnancies.[1,2] Being diagnosed with a cancer like leukemia pose tough challenge for pregnant women. And the diagnosis itself is a tough task for physicians because diagnostic practices become much complicated for blood cancer, compared with mainstream practices performed for solid cancers.[3] Leukemia during pregnance is manifested by ambiguous symptoms, including fatigue, systematic purpura, hemorrhage, shortness of breath, myeloproliferative disorder, unexplained anemia and platelet decrease.[4] Leukemia is therefore difficult to correctly diagnose in normal pregnancy follow-up. Nonetheless, diagnostic technology is evolving fast, contributing to the discovery of a new type of myeloproliferative cancer associated with pregnancy and its increasing trend.[5] Also, pregnant patient and her unborn baby can be successfully managed with antitumor therapy and induction birth, depending on gestational age.
As pregnancy does not affect the outcome of leukemia, termination of pregnancy is not medically required.[6,7] However, decreased platelets, anemia and low white blood cells (WBC) may lead to hemorrhage, infection and disturbances of oxygen and nutrition supply to the fetus, increasing the risks of miscarriage, fetal mortality, preterm labor and low birth weight.[8] Immediate chemotherapy and induction are therefore performed for the management of leukemia during pregnancy. Because belated treatment would result in a short survival time of less than 3 months, there is a need to prioritize timely treatment,[9] and a prompt diagnosis is crucial.
While acute leukemia diagnosed during pregnancy is rare, a case of acute leukemia diagnosed in pregnant woman just after delivery seems rarer. We present a case of acute myeloid leukemia (AML) diagnosed in a pregnant intensive care unit (ICU) patient who had undergone an emergency cesarean delivery (ECD) due to suspected sepsis and acute pulmonary embolism (PE) and review the relevant literature.
CASE REPORT
A 33-year-old pregnant woman with gestational age of 32 weeks and 5 days admitted to the hospital because of a 2-week history of heat sensation, shortness of breath and chest pain. Chest discomfort became severe a day before admission with a decrease in consciousness, forcing the patient to vist the closest hospital where the patient’s hemoglobin was 6.2 g/dl, compared with the previous value of 11.9 g/dl (at gestational age of 8 weeks 2 days). The patient was then transferred to out hospital. The patient had a history of appendectomy and three previous vaginal births with no other personal or family history. On admission, the patient showed height of 160 cm, weight of 70 kg, blood pressure of 133/82 mmHg, pulse rate of 126 bpm, respiration rate of 30 bpm, temperature of 38.6°C and SpO2 of 82%. The patient received oxygen via nasal prong at 5 L/min. In addition to acute distress, shortness of breath and heat sensation, the patient complained pressured-induced pain in the costovertebral angle.
After admission, the examination of the peripheral blood showed WBC of 13.74 × 103/mm3, hemoglobin of 6.4 g/dl, platelets of 120 × 103/μl, sodium of 138 mEq/L and potassium of 4.0 mEq/L. Biochemical examination showed total protein of 6.7 g/dl, albumin of 3.1 g/dl, AST/ALT of 14/10 IU/L, ALP of 103 IU/L, serum BUN/creatinine of 4.6/0.48 mg/dl, lactic acid of 1.2 mmol/L, fibrinogen of 427 mg/dl, FDP of 35.88 μg/ml, D-dimer of 986 mg/dl and NT-proBNP of 764 pg/ml. Blood clotting test showed no abnormalities and arterial blood gas (ABG) analysis showed pH 7.462, pCO2 24.2 mmHg and pO2 57.0 mmHg. Proteinuria was also found in urine test. Chest radiography obtained in the emergency room showed a large cardiac silhouette, pleural effusion and atelectasis (Fig. 1. With exacerbating breathing difficulties and chest discomfort, a loss of consciousness lasted, all of which raised a strong suspicion of PE and sepsis. As we decided that the fetus is supplied with insufficient oxygen in the patient’s condition, we arranged an ECD for the patient in the Obstetric Unit. After propofol and uxamethonium chloride were infused, anesthesia was induced using rapid sequence induction, followed by rocuronium induction and sevoflurane-remifentanil induction. Central venous catheter and radial artery cannulation were inserted to facilitate fluid and blood pressure monitoring and blood infusion during surgery. As the use of carbetocin and oxytocin failed to cause uterus contraction, uterine tamponade was created using the Bakri balloon. The patient gave birth to a baby boy weighing 1775.0 gm with Apgar scores of 1.3 and 5.4. After being intubated, the baby was transferred to the Newborn ICU. During surgery, the patient was given phyiological saline of 850 ml, hydroxyethyl starch (Volulyte 6%™, Fresenius-kabi Korea) of 1,000 ml and concentrated red blood cells of 750 ml and maintained urine output at 100 ml/h and blood loss of 800 ml. Total surgery time was 95 min and total anesthesia time was 120 min. After surgery, the patient was transferred to the ICU for further diagnosis, postoperative management and hemodynamic stabilization.
Because of the patient’s condition on admission, characterized by fever (> 38.0°C), increasing WBC (13.74 × 103/mm3), hypotension (average arterial pressure < 65 mmHg), tachycardia (heart rate > 120 bpm), tachypnea (respiration rate > 30 bpm), sepsis was suspected. We therefore decided to perform blood culture and used vephalosporin, ampicillin sodium/sulbactam sodium and aminoglycoside antibiotics. A possibility of developing PE after cesarean delivery along with suspected pulmonary edema due to deteriorating cardiac function called for the applications of contrast-enhancing CT for PE and transthoracic echocardiography. In addition, the appearance of pulmonary edema and pleural effusion on chest radiograph and hypoxia suggested by the results of ABG analysis (7.32–44 mmHg-124 mmHg-98.5%) after induction of anesthesia (FiO2% 0.6) prompted us to perform lung protective ventilation strategies in which ideal body weight ×6 and positive end-expiratory pressure (PEEP) of 5–10 were applied to maintain tidal volume of PO2 > 90 mmHg. After 4 days of artificial respiration, the patient’s FiO2 was gradually decreased and PO2 was 200 mmHg on a FiO2 of 0.4. Her chest radiography also showed a gradual clearing of pulmonary edema and pleural effusion. During artificial respiration in the ICU, the patient maintained propofol and remifentanil sedation for 36 hours after surgery. Norepinephrine was continuously infused at a rate of 0.2 ug/kg/min to maintain an average arterial pressure of 65 mmHg or higher. Although contrast-enhanced CT scan obtained after ICU admission revealed atelectasis in both lungs along with pleural effusion in small amounts, PE was not deteted (Fig 2). Transthoracic echocardiography showed no abnormal findings. We also reduced the dose of norepinephrine and discontinued it 3 days after the initial infusion, and the patient maintained normal blood pressure. However, postoperative laboratory tests revealed abnormal results: WBC of 17.46 × 103/mm3, 12% lymphocytes, 5% bone marrow cells, 8% meta-myelocytes, 22% mononuclear cells and 21% neutrophil. Peripheral blood smear test performed next day also revealed 3% blast cells. Moreover, despite the use of propacetamol-based fever reducer and a wide range of antibiotics such as piperacillin sodium/tazobactam and teicoplanin, the patient’s temperature went over 38.5°C during ICU stay. We suspected a blood cancer and conducted collaborative practice with the Division of Hematology. Next day, WBC increased to 2.26 × 103/mm3 or more than twice the previous day’s level and further abnormality of WBC differential distribution was suggested by 3% blast cells, 12% immature cells, 44% mononuclear cells and 16% neutrophil, calling for bone marrow biopsy and genetic analysis. Abdominal radiography, taken because of continued abdominal bloating and pain after surgery, showed that small intestine is completely blocked (Fig. 3a). Contrast-enhanced CT scanning of the abdomen/pelvis was also performed but no blocked intestine was detected although paralytic intestinal obstruction was suspected (Fig. 3b). We administered erythromycin lactobionate and metoclopramide HCL for the management of paralytic intestinal obstruction and arranged the patient to resume her diet. However, her had watery diarrhea, making us to suspect pseudo-membranous gastroenteritis and perform a difficile toxin test. The test result was however negative.
After 3 days in the ICU, the patient underwent bone marrow test. By confirming fully recovered consciousness and spontaneous breathing, we performed extubation. Further tests were performed to evaluate stem cells, histopathological factors, molecular genetics of cancer and parenchyma cells via bone marrow puncture and biopsy. While the suspension of AML was largely supported by stem cell evaluation results, the marrow, which was tightly packed by a proliferation of several meta-myelocytes and immature blasts on pathological examination, also matched with AML. The patient was NPM1 mutation positive in molecular genetics test and showed a positive reaction for CD13 (My7), HLA-DR, CD33 (My9), CD14, CD45, CD38 and anti MPO among the parenchyma cells.
After 4 days in the ICU, the patient’s vital signs were stable with no other symptoms except for diarrhea, which was associated with paralytic intestinal obstruction. She was therefore transferred to the Hematology ward. At 12 days after surgery, the patient began receiving chemotherapy starting with cytarabine 165 mg per day for 7 days, followed by idarubicin 19.8 mg per day for 3 days. In the general ward, chest radiography showed fluid collecting around the lung (Fig. 4.) We performed thoracentesis to remove the fluid from the lung and chemical analysis of collected fluid. The patient’s newborn baby was breathing spontaneously and slowly increasing her daily portion while being looked after in the neonatal ICU to meet the required goal weight of 2 kg. Blood culture results, which came out 5 days later after the testing at time of ICU admission, showed no sign of bacteria.
DISCUSSION
During pregnancy, plasma volume increases by 50% where as red blood cell numbers increase by 20% to 50%, leading to the onset of nomocylic anemia in pregnant women whose normal hemoglobin values range from 10 to 12 g/dl. Nomocylic anemia in pregnant women can get wrose if they do not have enough iron and folate which are crucial for hematopoiesis and fetal growth. Elevated WBC is considered typical during pregnancy, while it is usually caused by the increase of endogenous steroid hormone levels. WBC count can reach 12,000/µl during the third trimester of pregnancy and go up to 20,000–30,000/µl at parity. The number of platelets does not change much in normal pregnancy, but it can drop to less than 50,000/µl, albeit rarely, as a symptom of thrombocytopenia in young pregnant women causing idiopathic thrombocytopenic purpura.[4] The symptoms of leukemia are unspecific in pregnant women, but fatigue, shortness of breath, heat sensation, chills and muscle pain can appear with hematopoiesis-related symptoms such as hemorrhage, infection and bruise However, leukemia is difficult to diagnose in pregnant women based on presenting symptoms because those symptoms are not easily distinguishable from hematological changes associated with pregnancy. In case of suspected leukaemia, bone marrow examination is required. Bone marrow evaluation can be conducted safely regardless of gestational age.[4] However, reports in which a pregnant woman was admitted to the ICU due to acute leukemia have been extremely rare. The leading causes of mortality or ICU admission during pregnancy include sepsis, PE, amniotic fluid embolism, preeclampsia, obstetrical hemorrhage and heart disease.[10] The possibility that leukemia symptoms overlap with the symptoms of pulmonary complications cannot be ruled out, careful differential diagnosis is therefor required.
Sepsis is the most common cause of maternal mortality and morbidity in the world, accounting for approximately 15% of total maternal deaths, despite increasing effective hygiene and healthcare system and appropriate use of antibiotics around the world. The actual contribution of sepsis to maternal mortality would be higher if unreported cases were included.[11] Blanco et al.[12] reported the occurrence of bacteremia was 7.5 per 1,000 pregnant women and the incidence of sepsis in patients with bacteremia to be 10%. In the early stages, ambiguous signs and symptoms of sepsis can be mistaken for cardiopulmonary and immunodetection changes associated pregnancy, particularly in the early stages of sepsis. Generally, sepsis cause flu-like symptoms with fever as the major symptom in many cases, although symptoms can be varied by affected organs. Women with sepsis tend to develop hypothermia. Because sepsis affects unborn baby as well, prompt diagnosis is important. [11,12] In addition to sepsis, pulmonary complications are considered difficult to diagnose during pregnancy., demanding a thorough understanding of physiological changes of respiratory system and multifaceted approaches in order to achieve successful maternal-fetal outcome.[13] During pregnancy, breathing capacity and tidal volume significantly increase by 30% to 50% and total forced vital capacity, preliminary forced expiratory volume and residual volume decreases. But respiration rate does not increase. The residual volume significantly decreases by 20% during the third trimester of pregnancy, which is one of the most significant physiological changes of respiratory system caused by the rising diaphragm due to expansion of the uterus.[14,15] PE, one of pulmonary complications, is defined as an obstruction of more than one artery and caused by thrombus or blood clot deeply embedded in the veins in the lower limbs. PE can develop in the third trimester of pregnancy and more frequently develops after delivery. Common symptoms of PE include sudden extreme shortness of breath, arterial hypoxia, cyanosis, hypotension and dilated external jugular vein. For initial management of PE, securing airway and air supply are necessary. However, prevention is more important. PE can be diagnosed with a blood test like the D-dimer test or ultrasound scan. However, CT or scanning is used to confirm the initial diagnosis.[16] Pulmonary edema means an abnormal collection of fluid volume outside the blood vessels in the lungs,[17,18] which disturbs the diffusion of oxygen and carbon dioxide. When oxygen levels drop, hypoxia rapidly occurs, exposing the mother and baby to a lack of oxygen and causing adverse effects.[13]
In this paper, AML was diagnosed in a pregnant patient who had a preterm birth via ECD in the third trimester of pregnancy. The patient showed no abnormal findings in routine prenatal screenings. On admission, her symptoms and test results suggested respiratory complications such as sepsis, PE and pulmonary edema, making the diagnosis of leukemia difficult. Patients with acute leukemia commonly complain continued fatigue, nose bleeding, gum bleeding, weight loss, fever, brain pain and abdominal pain. They also may show anemia or bruise on the skin in physical examination and gastrointestinal hemorrhage. Often myeloproliferative disorder or lymphadenopathy occurs and fever and respiratory infection progress to sepsis. The impact of leukemia on the nervous system is rare, but headache, nausea and vomiting can occur. In blood test, the diagnosis of leukemia is based on symptoms of anemia, low platelet counts and a decrease or increase in the number of WBC. The number of WBC can remain unchanged. However, blasts are present in all patients with leukemia.[4] In this paper, abdominal discomfort caused by paralytic internal obstruction was one of the biggest complaints of the patient after ICU admission. The occurrence of intestinal obstruction (IO) is generally estimated at 1 per 1,500–66,431 pregnancies. This condition is commonly diagnosed in the 2nd or 3rd trimester of pregnancy and sometimes caused by a C-section. The most common symptom of IO is abdominal pain (98%), followed by vomiting and constipation. Pressure-induced pain occurred in 71% of patients with IO. when their stomach was touched. Abnormal peristalsis movement was observed in 55% of patients with IO, and abdominal radiography is widely used to diagnose IO.[19] In this paper, because the patient suffered from paralytic IO during the entire hospitalization period, we saw it as a complication of surgery or leukemia.
Caligiuri et al.[8] reviewed 350 case reports describing leukemia during pregnancy and reported that 72 cases out of 350 were diagnosed during the first pregnancy, and 44 of these cases had AML. Reynoso et al.[20] reported 58 cases of leukemia diagnosed during pregnancy for 10 years. Of 58 cases, 75% developed leukemia in the 2nd or 3rd trimester of pregnancy, and about half of them was diagnosed with AML. The earlier diagnosis of leukemia during pregnancy leads to the worse prognosis in the perinated period. That is because leukemia in the early stage of pregnancy causes anemia in the mother, disseminated intravascular coagulopathy and changes in WBCs, which in turn result in disorder in blood flow and exchange of oxygen and nutrients as well.[7] In addition, the combined use of chemotherapy drugs reportedly increases fetal growth disorder, fetal mortality, perinatal mortality and congenital malformed baby. However, the relationship between chemotherapy and these risk factors is not clear.[6]
Even though the frequency of AML during pregnancy is very low, its effects on the maternal and fetal conditions are critical when treatment is delayed or inappropriate, enabling the mother to lose a chance to treat leukemia or reach remission. Timely treatment is therefore vital. To decide timing for treatment, diagnosis is prerequisite; however differential diagnosis is very unpleasant process for pregnant women. Once blood test results show abnormal findings suggesting leukemia in pregnant women, blood cancer examination and diagnostic approach should be not ignored in any circumstances. With technological advances in management of perinatal pregnancy and immature babies, the maternal and fetal outcomes in pregnant patients diagnosed with leukemia are expected to improve.
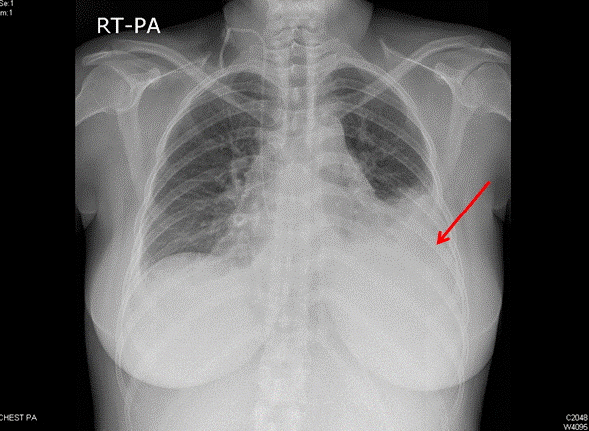
Fig. 1.Chest anterior-posterior X-ray shows suspicious cardiomegaly, pleural effusion and atelectasis at left lower lung field (at emergency room).

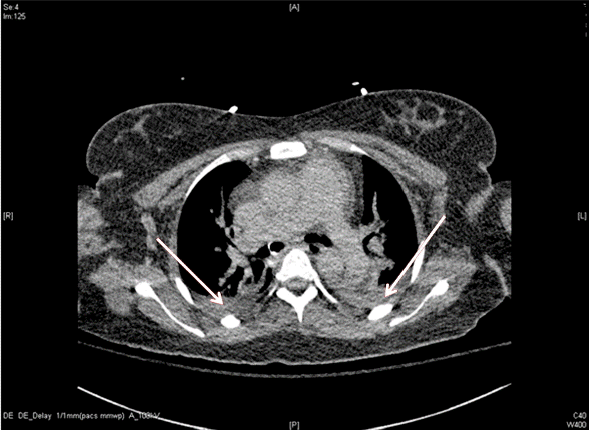
Fig. 2.Pulmonary embolism contrast-enhanced computed tomography (CT) shows diffuse dependent atelectasis of the both lungs and minimal amount of bilateral pleural effusion. No evidence of visible thromboembolism in the pulmonary artery in this CT (ICU admission # 1 day). ICU: intensive care unit.

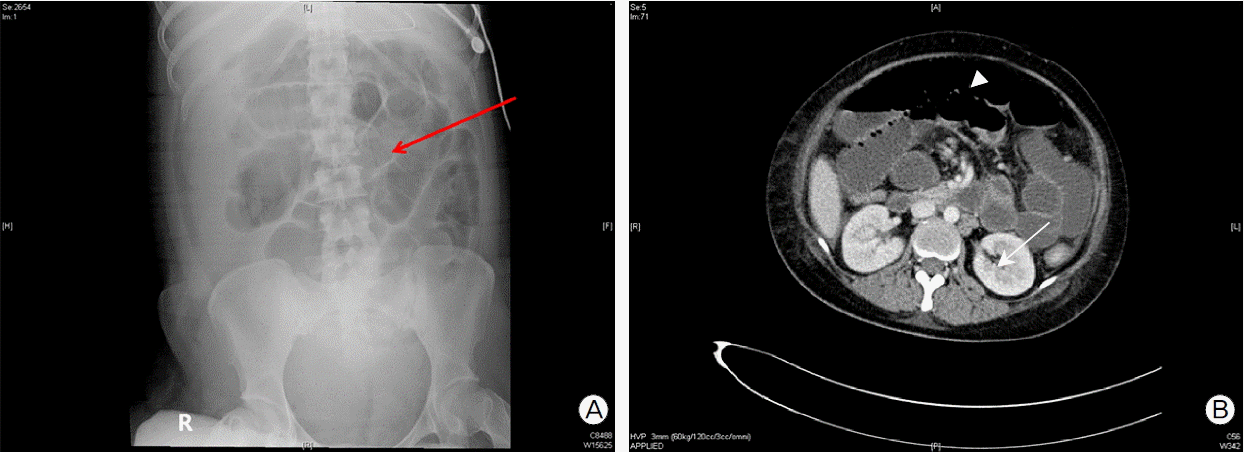
Fig. 3.(A) Abdominal flat X-ray shows complete obstruction of small bowel (jejunum or ileum). (ICU admission # 2 day) (B) Abdominal pelvis contrast-enhanced computed tomography (CT) shows, (ICU admission # 2 day) 1. Diffuse dilatation of small bowel loops (is seen more likely paralytic ileus. arrowhead) 2. Lt. kidney cortex focal perfusion defect (arrow). ICU: intensive care unit.

Fig. 4.Chest X-ray shows increased left pleural effusion (ICU discharge # 10 day). ICU: intensive care unit.
References
- 1). Pavlidis NA. Coexistence of pregnancy and malignancy. Oncologist 2002;7:279-87.ArticlePubMed
- 2). Haas JF. Pregnancy in association with a newly diagnosed cancer: a population-based epidemiologic assessment. Int J Cancer 1984;34:229-35.ArticlePubMed
- 3). Hurley TJ, McKinnell JV, Irani MS. Hematologic malignancies in pregnancy. Obstet Gynecol Clin North Am 2005;32:595-614.ArticlePubMed
- 4). Pejovic T, Schwartz PE. Leukemias. Clin Obstet Gynecol 2002;45:866-78.ArticlePubMed
- 5). Harrison C. Pregnancy and its management in the Philadelphia negative myeloproliferative diseases. Br J Haematol 2005;129:293-306.ArticlePubMed
- 6). Ali R, Ozkalemkaş F, Ozçelik T, Ozkocaman V, Ozan U, Kimya Y, et al. Maternal and fetal outcomes in pregnancy complicated with acuteleukemia: a single institutional experience with 10 pregnancies at 16 years. Leuk Res 2003;27:381-5.ArticlePubMed
- 7). Cardonick E, Iacobucci A. Use of chemotherapy during human pregnancy. Lancet Oncol 2004;5:283-91.ArticlePubMed
- 8). Caligiuri MA, Mayer RJ. Pregnancy and leukemia. Semin Oncol 1989;16:388-96.PubMed
- 9). Peleg D, Ben-Ami M. Lymphoma and leukemia complicating pregnancy. Obstet Gynecol Clin North Am 1998;25:365-83.ArticlePubMed
- 10). Clark SL, Belfort MA, Dildy GA, Herbst MA, Meyers JA, Hankins GD. Maternal death in the 21st century: causes, prevention, and relationship to cesarean delivery. Am J Obstet Gynecol 2008;199:36.e1. -5. discussion 91–2..ArticlePubMed
- 11). Bamfo JE. Managing the risks of sepsis in pregnancy. Best Pract Res Clin Obstet Gynaecol 2013;27:583-95.ArticlePubMed
- 12). Blanco JD, Gibbs RS, Castaneda YS. Bacteremia in obstetrics: clinical course. Obstet Gynecol 1981;58:621-5.PubMed
- 13). Frye D, Clark SL, Piacenza D, Shay-Zapien G. Pulmonary complications in pregnancy: considerations for care. J Perinat Neonatal Nurs 2011;25:235-44.ArticlePubMed
- 14). Contreras G, Gutiérrez M, Beroíza T, Fantín A, Oddó H, Villarroel L, et al. Ventilatory drive and respiratory muscle function in pregnancy. Am Rev Respir Dis 1991;144:837-41.ArticlePubMed
- 15). Crapo RO. Normal cardiopulmonary physiology during pregnancy. Clin Obstet Gynecol 1996;39:3-16.ArticlePubMed
- 16). Brown HL, Hiett AK. Deep vein thrombosis and pulmonary embolism in pregnancy: diagnosis, complications, and management. Clin Obstet Gynecol 2010;53:345-59.ArticlePubMed
- 17). Sciscione AC, Ivester T, Largoza M, Manley J, Shlossman P, Colmorgen GH. Acute pulmonary edema in pregnancy. Obstet Gynecol 2003;101:511-5.ArticlePubMed
- 18). Poole JH, Spreen DT. Acute pulmonary edema in pregnancy. J Perinat Neonatal Nurs 2005;19:316-31.ArticlePubMed
- 19). Stukan M1, Kruszewski Wiesław J, Dudziak M, Kopiejć A, Preis K. [Intestinal obstruction during pregnancy]. Ginekol Pol 2013;84:137-41.PubMed
- 20). Reynoso EE, Shepherd FA, Messner HA, Farquharson HA, Garvey MB, Baker MA. Acute leukemia during pregnancy: the TorontotoLeukemia Study Group experience with long-term follow-up of children exposed in utero to chemotherapeutic agents. J Clin Oncol 1987;5:1098-106.ArticlePubMed
Citations
Citations to this article as recorded by














 KSCCM
KSCCM
 PubReader
PubReader ePub Link
ePub Link Cite
Cite