Abstract
- Kawasaki disease (KD) is an acute, systemic vasculitis of childhood. The early mortality of KD results from coronary complications, mainly aneurysmal thrombosis with myocardial infarction, and the subacute phase of KD has the highest risk of mortality. Although there have been reports of ischemic heart disease as late cardiologic sequelae of KD in young adults, acute myocardial infarction caused by coronary complications in the subacute phase of KD is rare. We experienced one pediatric patient who developed coronary artery aneurysm and acute myocardiac infarction (AMI) during the subacute phase of incomplete and intravenous immunoglobulin (IVIG)-nonresponsive KD. The patient was given a good prognosis due to close monitoring and early recognition of AMI. Physicians should carefully monitor KD patients who do not respond to initial IVIG therapy and who show progressive coronary artery dilatation. If such a patient complaints of chest pain and the ECG shows hyperacute T waves, the physician should suspect development of AMI.
-
Keywords: electrocardiogram; immunoglobulin; Kawasaki disease; myocardial infarction
Kawasaki disease (KD) is an acute, systemic vasculitis of childhood. Sometimes, patients’ clinical features cannot fulfill diagnostic criteria for KD, and it is categorized as incomplete KD. In such cases, diagnosis as KD may be delayed and they may have high risk of coronary artery complications.[1,2]
Intravenous immunoglobulin (IVIG), a potent anti-inflammatory agent, is an effective therapy in patients with KD. Ten to 15 percent of patients with KD who are initially treated with IVIG may be febrile until 36–48 hours after treatment.[3,4] Persistent fever is a sign of ongoing vasculitis and increased risk of coronary artery complication. Acute myocardiac infarction (AMI) mostly occurred within 1 year of illness and 35% of patients had AMI within the first 3 months after onset.[5] Unlike previous cases, we experienced one pediatric patient who developed AMI during the subacute phase of the incomplete and IVIG nonresponsive KD.
CASE REPORT
A five-year-old healthy male patient who had no specific medical history was hospitalized due to fever lasting one day, and painful mass palpation in the cervical area during a period of two days. On physical examination on the day of admission, his temperature, pulse, respiratory rate, and blood pressure were 38.6°C, 120 beats/minute (bpm), 24/minute and 90/60 mmHg, respectively. He showed abdominal pain, vomiting, nausea, periorbital edema and sublingual lymph node enlargement (right > left, 4 × 4 cm). However, no sign of a rash or change of hand, foot, lip or tongue was observed. Serologic laboratory findings showed white blood cell count (WBC) 11,250 mm3 (neutrophil 79.5%), C-reactive protein (CRP) 13.31 mg/dl, erythrocyte sedimentation rate (ESR) 27 mm/hr, N-terminal pro-brain natriuretic peptide (NT-pro-BNP) 36.0 pg/ml and elevated aspartate aminotransferase (AST)/alanine aminotransferase (ALT) 122/147 U/L. Urine examination showed pyuria. Several serologic viral antibody tests including Epstein-Barr virus, hepatitis, herpes simplex, cytomegalovirus and rubella showed negative findings. Abdominal ultrasonogram (USG) showed pericholecystic edema and diffuse wall thickening of the terminal ileum with multiple mesenteric lymph node enlargement. In addition, neck USG showed cervical lymph node enlargement with reactive hyperplasia.
Antibiotic therapy was started promptly. In spite of antibiotic therapy for two days, high fever continued and cervical lymph node enlargement did not improve. In addition, he showed conjunctival injection without discharge. WBC (13,030 mm3), CRP (14.29 mg/dl), and NT-pro BNP (5,298 pg/ml) were elevated. From urine culture, sterile pyuria was confirmed. Kawasaki disease was suspected by consideration of patient’s clinical symptoms and laboratory findings and IVIG 2 g/kg infusion and high dose aspirin (100 mg/kg) medication were started. However, fever persisted 48 hours after administration of IVIG therapy, and a second IVIG infusion and methylprednisolone pulse therapy (30 mg/kg) were administered. Echocardiography on day 5 of admission showed dilatation in the proximal segment of both coronary arteries (right coronary artery [RCA] 5.4 mm, left main coronary artery [LM] 3.9 mm, and left anterior descending artery [LAD] 4.1 mm). On day 8 of admission, the patient became afebrile. On echocardiography on day 12 of admission, the aneurysms increased in size (RCA 7.5 mm, LM 4.3 mm, and LAD 4.1 mm) and clopidogrel (1 mg/kg) was added.
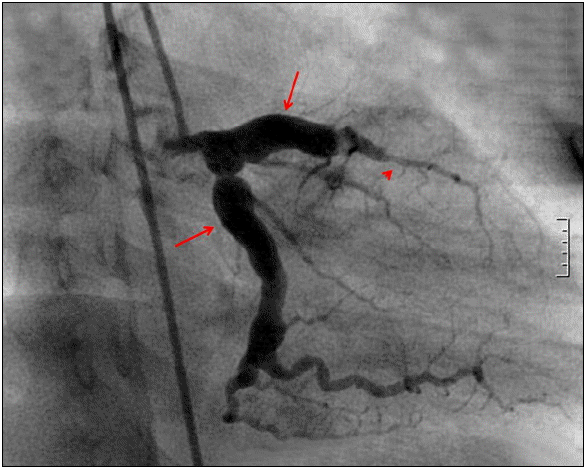
On day 14 of admission, the patient complained of sudden onset of chest pain. An Electrocardiogram (ECG) showed normal sinus rhythm with normal intervals but hyperacute T waves in the anteroseptal precordial leads (Fig. 1). The patient was transferred to the cardiology intensive care unit and monitored continuously. Follow-up ECG showed the improvement of tall T, followed by ST-segment elevation, loss of the R waves, and abnormal Q waves in V2-4 (Fig. 2). Echocardiography showed the left ventricle with depressed global contractility with diffuse hypokinesia in addition to the known coronary artery aneurysms and dilatation. Results of blood tests were: platelets count 1,044,000 mm3, serum potassium 6.2 mEq/L, creatinine kinase- MB 142.1 ng/ml, troponin I 14.81 ng/ml, and NT-pro BNP 4477.2 pg/ml. For correction of hyperkalemia, intravenous bicarbonate and calcium gluconate were administered. Coronary angiography showed signs of LAD total occlusion (Fig. 3), and percutaneous transluminal coronary angioplasty (PTCA) using a balloon was performed. After successful intervention, LAD was recannalized.
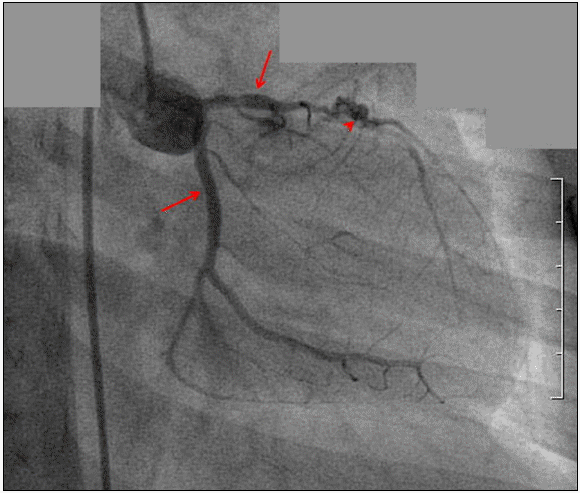
On day 20 of admission, the boy was discharged from the hospital and has taken medication, including low dose aspirin, warfarin, angiotensin-converting-enzyme inhibitor (ACEI), beta-blocker, and diuretics. After the coronary intervention, the patient was stable without chest pain under oral medication. In the follow up study six months later, left ventricular function was normalized and ECG showed reappearance of R wave in V2-4. A follow up coronary angiography showed patent LAD and aneurysmal change on the previous ballooning site, but the previous aneurysms in the proximal coronary artery were improved (Fig. 4).
DISCUSSION
The prevalence of coronary artery complications that occurred in patients with incomplete KD has shown contradictory results. One study showed that incidence of coronary artery complications in complete KD was larger than that in incomplete KD.[6] Another study reported that the prevalence of coronary artery complications in incomplete KD was higher than in complete KD and showed inverse correlation with the number of current symptoms.[1] They reported that delayed diagnosis of incomplete KD caused coronary artery complications. In a recent study, no significant difference in the prevalence of coronary artery complications was observed between complete and incomplete KD.[7]
IVIG has a potent anti-inflammatory effect in KD although the mechanisms of action are unknown.[3] The current recommended dose for IVIG in children with KD is a single infusion of 2 g/kg over a period of 8–2 hours. IVIG decreases the incidence of coronary artery complications.[8] Previous studies compared the incidence of refractory KD between incomplete KD and complete KD. One study reported that the proportions of refractory KD were not significantly different between complete and incomplete KD.[9] Results of another study showed that the proportions of patients with incomplete KD receiving additional IVIG therapy were significantly smaller than in patients with complete KD.[6] Among refractory KD patients who received a second high-dose of IVIG, 20 to 48 percent of patients developed coronary artery complications.[10,11]
Our patient showed only two symptoms, cervical lymph node enlargement and conjunctival injection without discharge. On day 3 of admission, because he had persistent fever and increased levels of CRP and NT pro BNP in repeated blood tests, incomplete KD was suspected and IVIG therapy was started. When fever did not subside after initial IVIG therapy, additional IVIG and steroid therapy were promptly administered for prevention of coronary artery complications. Although the therapeutic effect of steroid therapy for refractory KD is controversial, several studies showed that methylprednisolone pulse therapy (MPT) combined with IVIG reduced incidence of coronary artery lesions and is effective on initial IVIG unresponsive KD.[12–14] In this case, initiation of treatment was not delayed. Nevertheless, persistent fever during a period of eight days, which reflects ongoing vascular inflammation, resulted in formation of coronary artery aneurysms.
Patients with coronary aneurysms, especially giant (> 8 mm internal diameter) and rapidly expanding aneurysms, can experience thrombotic complications. In spite of active antithrombotic treatment, thrombus results in AMI and death.[3,5] AMI from KD mostly occurs in the first year after KD.[3,5] The previous study of 195 children with AMI secondary to KD reports that 73% of patients had AMI within 1 year of KD onset, and 50% of them had AMI within the first 3 months.[5] They also reported that mortality in patients with symptomatic AMI is higher than in the patient with asymptomatic AMI. In Korea, there have been reports of ischemic heart disease as late cardiologic sequelae of KD.[15–17] In our case, AMI occurred at the subacute phase of KD just after subsiding fever. In fact, the subacute phase is the period of highest risk of sudden death in KD patients,[18] and the early mortality results from coronary complications, mainly aneurysmal thrombosis with myocardial infarction. [5,17] Therefore, early recognition and treatment for AMI are critical. KD patients with giant or rapidly expanding aneurysms must hospitalize into intensive care unit (ICU) and physicians should carefully monitor them. If patient complaints of chest pain and ECG shows hyperacute T wave, physicians should suspect the development of AMI. Close monitoring, early recognition, and aggressive anticoagulant therapy can result in good prognosis.
Like adult patients, percutaneous balloon angioplasty, rotational ablation, directional coronary artherectomy, stent insertion and coronary artery bypass graft can be used to treat pediatric patients with coronary artery stenosis or occlusion. Experts reported the guidelines for thrombolytic therapy and catheter intervention according to patient’s age, conditions and lesion sites in coronary artery lesion in KD.[19,20] If patient is a young child and has stenotic lesions with mild or no calcifications within 6 years of KD onset, PTCA should be the choice of treatment.[20]
In conclusion, physicians should carefully monitor KD patients who do not respond to initial IVIG therapy and show progressive coronary artery dilatation. In addition, physicians should suspect development of AMI if patients complain of chest pain and ECG shows hyperacute T wave.
Fig. 1.Electrocardiogram on admission day 14 showing hyper-acute T waves in the anteroseptal precordial leads (arrow).
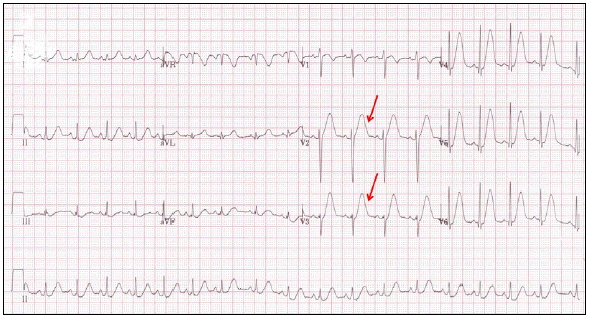
Fig. 2.Electrocardiogram showing the improvement of tall T, followed by ST-segment elevation (arrow), loss of the R waves, and abnormal Q waves (arrow head) in V2-4.
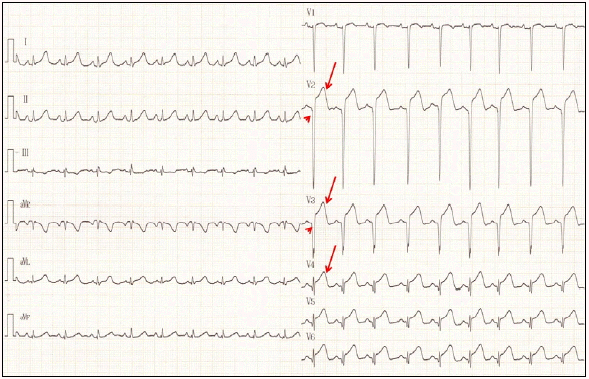
Fig. 3.Coronary angiography showed signs of total occlusion of left anterior descending artery (arrow head) and aneurismal changes of coronary arteries (arrow).

Fig. 4.A follow up coronary angiography showed patent left anterior descending artery and aneurysmal change around the previous ballooning site (arrow head). The previous aneurysms in the proximal coronary arteries were improved (arrow).
References
- 1). Sonobe T, Kiyosawa N, Tsuchiya K, Aso S, Imada Y, Imai Y, et al. Prevalence of coronary artery abnormality in incomplete Kawasaki disease. Pediatr Int 2007;49:421-6.ArticlePubMed
- 2). Manlhiot C, Yeung RS, Clarizia NA, Chahal N, McCrindle BW. Kawasaki disease at the extremes of the age spectrum. Pediatrics 2009;124:e410-5.ArticlePubMed
- 3). Newburger JW, Takahashi M, Gerber MA, Gewitz MH, Tani LY, Burns JC, et al. Diagnosis, treatment, and long-term management of Kawasaki disease: a statement for health professionals from the Committee on Rheumatic Fever, Endo- carditis, and Kawasaki Disease, Council on Cardiovascular Disease in the Young, American Heart Association. Pediatrics 2004;114:1708-33.ArticlePubMed
- 4). Burns JC, Glodé MP. Kawasaki syndrome. Lancet 2004;364:533-44.ArticlePubMed
- 5). Kato H, Ichinose E, Kawasaki T. Myocardial infarction in Kawasaki disease: clinical analyses in 195 cases. J Pediatr 1986;108:923-7.ArticlePubMed
- 6). Sudo D, Monobe Y, Yashiro M, Mieno MN, Uehara R, Tsuchiya K, et al. Coronary artery lesions of incomplete Kawasaki disease: a nationwide survey in Japan. Eur J Pediatr 2012;171:651-6.ArticlePubMed
- 7). Giannouli G, Tzoumaka-Bakoula C, Kopsidas I, Papadogeorgou P, Chrousos GP, Michos A. Epidemiology and risk factors for coronary artery abnormalities in children with complete and incomplete kawasaki disease during a 10-year period. Pediatr Cardiol 2013;34:1476-81.ArticlePubMed
- 8). Terai M, Shulman ST. Prevalence of coronary artery abnormalities in Kawasaki disease is highly dependent on gamma globulin dose but independent of salicylate dose. J Pediatr 1997;131:888-93.ArticlePubMed
- 9). Ghelani SJ, Sable C, Wiedermann BL, Spurney CF. Increased incidence of incomplete Kawasaki disease at a pediatric hospital after publication of the 2004 American Heart Association guidelines. Pediatr Cardiol 2012;33:1097-103.ArticlePubMed
- 10). Burns JC, Capparelli EV, Brown JA, Newburger JW, Glode MP. Intravenous gamma globulin treatment and retreatment in Kawasaki disease. US/ Canadian Kawasaki Syndrome Study Group. Pediatr Infect Dis J 1998;17:1144-8.ArticlePubMed
- 11). Hashino K, Ishii M, Iemura M, Akagi T, Kato H. Re-treatment for immune globulin-resistant Kawasaki disease: a comparative study of additional immune globulin and steroid pulse therapy. Pediatr Int 2001;43:211-7.ArticlePubMed
- 12). Ogata S, Bando Y, Kimura S, Ando H, Nakahata Y, Ogihara Y, et al. The strategy of immune globulin resistant Kawasaki disease: a comparative study of additional immuneglobulin and steroid pulse therapy. J Cardiol 2009;53:15-9.ArticlePubMed
- 13). Teraguchi M, Ogino H, Yoshimura K, Taniuchi S, Kino M, Okazaki H, et al. Steroid pulse therapy for children with intravenous immunoglobulin therapy-resistant Kawasaki disease: a prospective study. Pediatr Cardiol 2013;34:959-63.ArticlePubMed
- 14). Jibiki T, Kato I, Shiohama T, Abe K, Anzai S, Takeda N, et al. Intravenous immune globulin plus corticosteroids in refractory Kawasaki disease. Pediatr Int 2011;53:729-35.ArticlePubMed
- 15). Lee SY, Park JE. Acute myocardial infarction in young patient probably due to Kawasaki disease. Korean Circ J 2001;31:119-24.Article
- 16). Kim WJ, Song JG, Yoo BW, Kim Y, Choi JY, Sul JH, et al. Giant coronary aneurysm with stenosis and thrombus formation due to Kawasaki Disease: Treatment with graft stent. J Korean Pediatr Cardiol Soc 2005;9:394-9.
- 17). Choi YS, Kwon T, Lee H, Lee SY, Lee WT. Medicolegal Investigation of Kawasaki Disease: Three cases of sudden death by coronary artery lesions. Korean J Leg Med 2003;27:39-44.
- 18). Kliegman RM, Stanton BF, Geme JW, Schor NF, Behrman RE. Nelson textbook of pediatrics. 19th ed.Philadelphia, Saunders. 2011, pp 862-7.
- 19). Paredes N, Mondal T, Brandão LR, Chan AK. Management of myocardial infarction in children with Kawasaki disease. Blood Coagul Fibrinolysis 2010;21:620-31.ArticlePubMed
- 20). Ishii M, Ueno T, Akagi T, Baba K, Harada K, Hamaoka K, et al. Guidelines for catheter intervention in coronary artery lesion in Kawasaki disease. Pediatrics Int 2001;43:558-62.Article
Citations
Citations to this article as recorded by















 KSCCM
KSCCM
 PubReader
PubReader ePub Link
ePub Link Cite
Cite