Abstract
-
Background
- Endotracheal suctioning is associated with complications that include bleeding, infection, hypoxemia, cardiovascular instability, and tracheal mucosal injury. Recently, a closed-suction catheter with a pressure valve (Acetrachcare, AceMedical Co., Republic of Korea) was developed. We hypothesized that this new catheter might reduce tracheal mucosal injury compared to a conventional closed-suction catheter (Trachcare, Kimberly-balla RD, USA).
-
Methods
- This prospective, randomized study enrolled medical and surgical patients who required mechanical ventilation for more than 48 hours. Patients were randomized into two groups: one group was suctioned with the conventional closed-suction catheter (CCC) and the other group was suctioned with the closed-suction catheter with pressure valve (CCPV). Bronchoscopy was performed 48 hours later, and the severity of tracheal mucosal injury was graded on a 5-point scale, as follows: 0 = normal; 1 = erythema or edema; 2 = erosion; 3 = hemorrhage; and 4 = ulceration or necrosis.
-
Results
- A total of 76 patients (37 with CCPV and 39 with CCC) were included. There were no significant differences between the groups regarding demographic characteristics, changes in hemodynamic parameters during suction, incidence of pneumonia, length of intensive care unit (ICU) stay, or ICU mortality. On bronchoscopic evaluation, the use of the CCPV led to a significant decrease in tracheal mucosal injury (median tracheal mucosal injury grade 1 [IQR 0–1] vs. 2 [IQR 1–3], p = 0.001).
-
Conclusions
- We conclude that the novel closed-suction catheter with pressure valve may reduce tracheal mucosal injury compared to conventional catheters.
-
Keywords: closed-suction catheter; endotracheal suction; tracheal mucosal injury
INTRODUCTION
Endotracheal suctioning is an essential procedure for the management of intubated patients to prevent airway obstruction and atelectasis. Although open tracheal suction is commonly used in intubated patients, this procedure may increase the risk of developing atelectasis and hypoxia because oxygen and positive end-expiratory pressure are discontinued during suctioning.[1–4] Moreover, open suction can cause cardiovascular complications such as arrhythmia and hypotension by stimulating the vagus nerve.[2–5] Infection, increased intracranial pressure and tracheal mucosal damage can also result from open suctioning.[2–5] Therefore, closed tracheal suctioning emerged as a safer option as it does not require the removal of mechanical ventilation during suctioning. Closed suctioning, backed by continuous supply of oxygen and positive end-expiratory pressure, is said to provide physiological stability and reduce the risk of nosocomial infection by keeping ventilator circuit closed.[6–12] However, its efficacy in preventing infections is still debated.[13,14]
Newly developed closed-suction catheter (AceTrachcare, AceMedical Co., Republic of Korea) has dial-type pressure valve, located on the suction port, enables physicians to vary the level of suction pressure depending on the amount of secretions (Fig. 1), making it more effective in reducing tracheal mucosal injury, compared to conventional closed-suction catheters. This study aimed to compare tracheal mucosal injuries from this new suction catheter (AceTrachcare) and one of the most commonly used closed-suction catheters (Trachcare, Kimberly-balla RD, USA). We also compared the efficacy of these two closed- suction systems on hypoxia, hemodynamic factors, infection, intensive care unit (ICU) length of stay and ICU mortality.
MATERIALS AND METHODS
This study was conducted at Seoul National University Bundang Hospital from March to November 2013. This study protocol was approved by a local Clinical Research Ethics Board Committees (IRB No. E-1210/174-002). Patients aged 20 years or older were admitted to medical or surgical ICU and required mechanical ventilation for at least 48 hours after intubation, meeting the selection criteria. Patients were excluded if: they underwent tracheostomy prior to ICU admission, they were intubated at a previous hospital or operating room, they had a chest tube, they could not receive sedatives or muscle relaxants, they showed unstable vital signs for six hours prior to catheter application, they could not undergo bronchoscopy, they had tracheal mucosal damage according to the previous bronchoscopic examination or they did not agree to give informed consent. Eligible patients were randomly assigned to the experimental (AceTrachcare) group and the control (Trachcare) group.
Two different sizes of suction catheter were used depending on the internal diameter of endotracheal tubes: 12 Fr (4 mm) for 7.5 mm tube and 14 Fr (4.7 mm) for 8.0 mm tube. Each endotracheal suctioning was performed at an interval of three hours, and ICU nurses adjusted the time intervals based on the needs of individual patients. Inspired oxygen concentration was increased to 100% 15 min before suctioning and brought back down again 30 min after suctioning. For each patient, we inserted a suction catheter through the endotracheal tube until resistance was met, slowly pulled back the catheter about 2–3 cm, and then suctioned gently for no more than 15 seconds. The pressure of wall suction was set at 100 mmHg for both the experimental and control groups. However, in the experimental group, suction pressure was adjusted by an intensivist using the pressure valve. Suction pressure was adjusted with three levels, depending on the amount of secretions: Low, medium and high. Suction catheters were cleaned after each suctioning using normal saline and replaced every 48 hours.
Pressure-controlled ventilation was performed with low tidal volume of 6 ml per kilogram of predicted body weight as part of lung protective strategy, and adjusted based on arterial blood gas (ABG) results. All intensive cares were provided based on the practice guidelines for ICU patients under the supervision of intensivist.
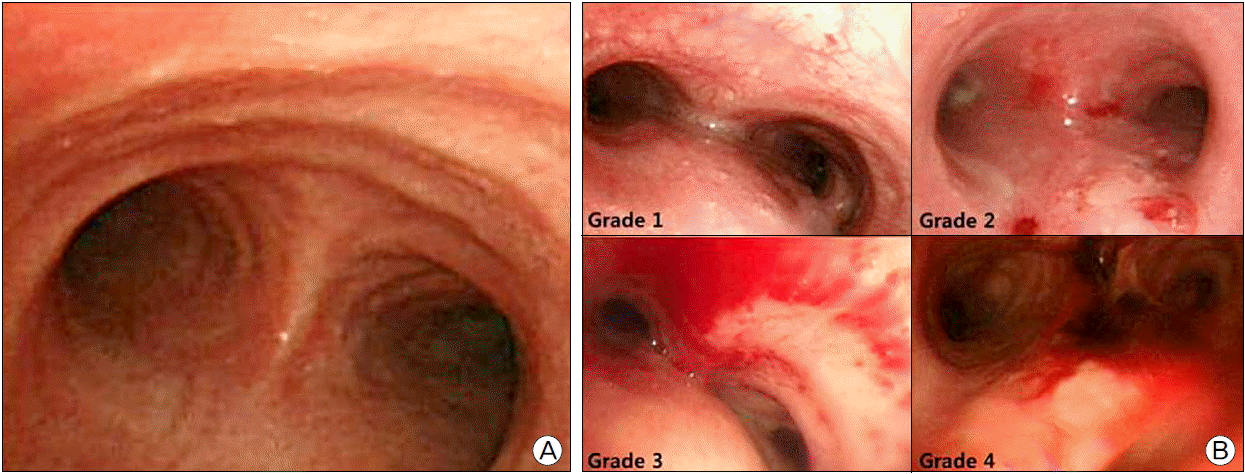
To measure the damage of tracheal mucosa, bronchoscopy was performed 48 hours after catheter application, and the degrees of tracheal mucosal damage were divided into five levels (Fig. 2). (0 = normal, 1 = erythema or edema, 2 = erosion, 3 = hemorrhage, 4 = ulceration or necrosis). The recordings of mucosal state were taken during bronchoscopy, and the degree of tracheal mucosal damage was reviewed by two intensivists.
We also monitored changes in heart rate, hemodynamics status such as mean arterial pressure and new onset of arrhythmia, color and amount of secretions and the number of suctioning between the baseline and 48 hours after catheter application. Hypoxia was observed using pulse oximetry during suctioning, and the partial pressures of oxygen and carbon dioxide were measured using ABG analysis immediately after suctioning. In addition, medical records of nosocomial infection, ICU length of stay and ICU mortality were collected.
In this study, we assigned at least 28 patients to each group under the assumption that the use of the newly developed suction catheter would lead to a 20% decrease in tracheal mucosal damage.[15] Statistical analysis was performed using SPSS 18.0 (Statistical Package for Social Sciences, Chicago, IL, USA). Continuous variables were analyzed using Student t test or Mann-Whitney U test. Categorical variables were analyzed using chi-square test. Results were expressed as mean ± SD, median ± quartiles or frequency (%), and p < 0.05 were considered statistically significant.
RESULTS
A total of 462 patients met inclusion criteria for this study among 855 patients who were admitted to either the medical ICU or the surgical ICU at Seoul National University Bundang Hospital from March through November 2013. Only 76 patients participated in this study as the majority of candidates did not consent to data collection after assessment for eligibility. Of 76 patients, 37 were assigned to the experimental group and 39 to the control group. However, the number of patients who completed the clinical trial was 29 in the experimental group and 31 in the control as eight patients did not undergo bronchoscopy in each group due to death or early extubation. There were no significant differences in general characteristics of the two groups, including age, gender, causes of admission, bleeding tendency and severity of disease (APACHE II score) (Table 1). The two groups showed a similar pattern in distribution of color and amount of secretions, which has a significant impact on endotracheal suctioning. No change was discovered in color and amount of secretions after catheter application in both groups. The total number of suctioning performed for 48 hours was not significantly different between the two groups (Table 2).
When the tracheal mucosa was examined with a bronchoscope 48 hours after catheter application, the experimental (AceTrachcare) group consisted of 9 patients with no injury, 15 with erythema or edema, and 5 with mucosal erosion. No patients displayed either mucosal hemorrhage or mucosal ulcer/necrosis. The control (Trachcare) group comprised 2 patients with no injury, 9 with erythema or edema, 8 with mucosal erosion, 9 with mucosal hemorrhage, and 3 with mucosal ulcer or necrosis (Table 3). When tracheal mucosal damages were classified into five levels, the median level was 1.00 [IQR 0–1.00] in the experimental group, demonstrating significantly less damage, compared to 2.00 [IQR 1.00–3.00] in the control group (p = 0.001).
Changes in hemodynamics from the baseline (24 hours before catheter application) and 48 hours after catheter application were used to identify physiological stability during suctioning. 3 patients in the experimental group and 1 in the control group showed increased heart rate of more than 20% after catheter application. Arrhythmia was detected in 8 and 9 patients, respectively, in the experimental and control groups during suctioning, but all patients already had history of arrhythmia before catheter application. There was no significant difference in maximum heart rate and mean arterial pressure between the two groups during suctioning. In summary, there were no suctioning-induced hemodynamic complications apart from arrhythmia (Table 2).
When monitored with pulse oximetry during suctioning, a significant decrease (more than 20%) in oxygen saturation did not occur in both groups. And ABG test performed immediately after catheter application showed that no patient demonstrated either more than 20% decrease in partial pressure of oxygen or more than 20% increase in partial pressure of carbon dioxide compared to previous test. Patients were stable without exhibiting hypoxia during suctioning (Table 2).
ICU outcomes were also evaluated in terms of nosocomial infection, ICU length of stay and ICU mortality. To evaluate nosocomial infection in an indirect manner, sputum and blood culture results were analyzed. Patients with positive sputum culture were 22 and 23, respectively, in the experimental and control groups, while patients with positive blood culture results were 4 and 2, respectively, in the two groups. The mean ICU length of stay was 17.1 days and 21.6 days in the experimental and control groups, and ICU mortality cases were 12 in each group. Therefore, there were no significant differences between the two groups with respect to ICU outcomes (Table 3).
DISCUSSION
The purpose of this study was to evaluate the efficacy of newly developed closed suction catheter containing a pressure valve. Newly developed closed-suction catheter demonstrated better outcomes in tracheal mucosal injury without increasing complications due to its pressure valve, which allows adjustment of pressure according to the amount of secretion. The mucosal damage can be caused by either mechanical trauma of catheter or negative pressure applied during suctioning. In this study, less tracheal mucosal damage is likely attributable to controlled negative pressure rather than mechanical factors. Since wall suction pressure was set to the same level for both groups during suctioning, the efficacy of pressure valve is evident in this study. Clinical practice guidelines for endotracheal suctioning recommends to minimize suction pressure for the removal of secretion and to keep suction pressure of less than 150 mmHg for adult patients.[4,16] However mid-level pressure is commonly applied when suctioning in real practices. New catheter used in this study led to less damage to tracheal mucosa as it contains a dial-type pressure valve for easy pressure control.
Some previous studies described closed-suction catheters as the cause of tracheal mucosal damage as they are harder and less convenient to handle than open-suction catheters[17] and need longer suctioning time with low efficiency.[18,19] The experimental catheter used in this study is considered capable of overcoming limitations of conventional close suction systems. We did not evaluate directly suctioning efficiency of the new catheter in this study. However, the total number of suctioning performed with the experimental catheter and the control catheter for 48 hours was not significantly different. We therefore postulate that suctioning efficiency of the two catheters was similar.
Closed-suction catheter is widely used in the ICU due to favorable outcomes, including improved oxygenation, reduced hypoxia, positive end-expiratory pressure maintenance and subsequent decrease in atelectasis and decreased risk of nosocomial infection.[6–12] Closed suction systems may be more effective for the ICU environment where ICU bed density is too high. In fact, some Korean studies showed closed suction system outperforms open suctioning in reducing infections.[9–11] In this study, there were no differences between the experimental and control catheters with respect to the incidence of hypoxia, hemodynamics, infection and ICU length of stay probably because both were closed-suction catheters. Further studies are needed to identify the difference between closed and open-suction catheters with respect to abovementioned outcomes.
Suctioning-induced damage to tracheal mucosa was evaluated using bronchoscopy 48 hours after catheter application. The images of tracheal mucosa were reviewed by two intensivists to increase interrater reliability. To determine the degree of damage, we modified a Damage index developed in a previous study of catheter[15] and classified the damage into five levels based on severity. Thus analysis results became more specific, compared to a previous study investigating the presence or absence of tracheal mucosal damage.[20] Injuries to the tracheal mucosa can be caused by a variety of factors during suctioning, including suctioning method, suction frequency and pressure, catheter size and catheter insertion depth. Participants were randomly assigned to the experimental and control groups to minimize effects of other variables than suction pressure. Nonetheless, variables could not be completely controlled when suctioning was performed by many nurses. That is one of the limitations of this study.
This study demonstrated that the experimental catheter containing a pressure valve was more effective at reducing damage to the tracheal mucosa than one of conventional closed-suction catheters. This new catheter can be safely and effectively used for intubated patients in ICU. Its application will also prove useful in patients with a high risk of tracheal mucosal damage due to various reasons such as bleeding.
Fig. 1.(A) The closed-suction catheter with pressure valve. (B) A dial valve can adjust the level of suction pressure according to sputum amount.
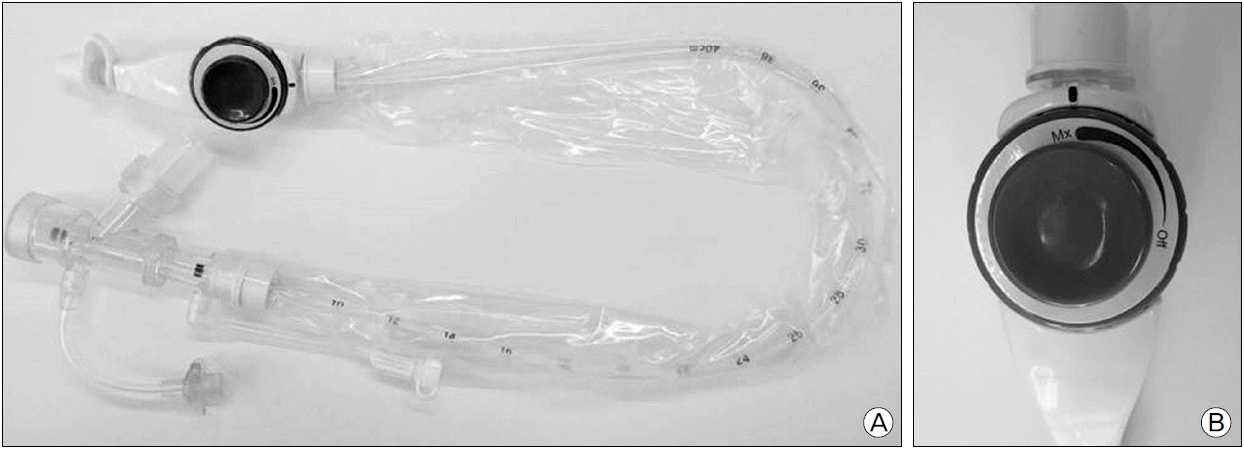
Fig. 2.(A) Image of normal tracheal mucosa at the carina level. (B) Images for the degree of tracheal mucosal injury. Grade 1 = erythema or edema, Grade 2 = erosion, Grade 3 = hemorrhage, Grade 4 = ulceration or necrosis.
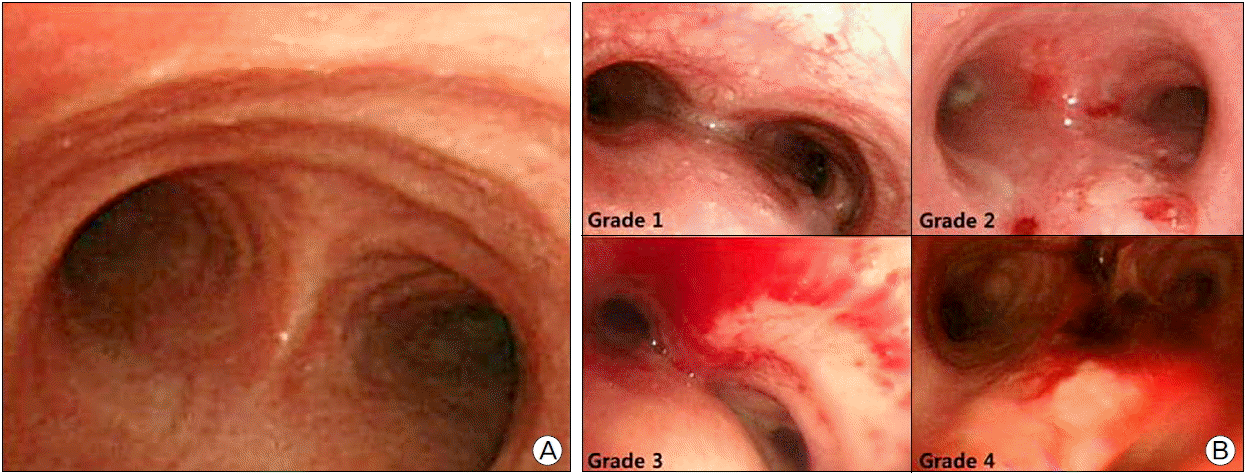
Table 1.Demographic data of patients
|
Closed-suction catheter with pressure valve |
Conventional closed-suction catheter |
p value |
|
Age (yr) |
69.7 ± 15.1 |
74.2 ± 9.7 |
0.131 |
|
Gender (Male), n (%) |
24 (64.9) |
27 (69.2) |
0.686 |
|
Admission cause, n (%) |
|
|
0.766 |
|
Pneumonia |
15 (40.5) |
19 (48.7) |
|
|
PostOP |
7 (18.9) |
4 (10.3) |
|
|
Neurologic disease |
3 (8.1) |
4 (10.3) |
|
|
Sepsis, Septic shock |
2 (5.4) |
4 (10.3) |
|
|
ARDS |
3 (8.1) |
2 (5.1) |
|
|
HF, MI |
3 (8.1) |
2 (5.1) |
|
|
Multiple trauma |
2 (5.4) |
2 (5.1) |
|
|
Others |
2 (5.4) |
2 (5.1) |
|
|
Coagulation lab abnormality, n (%) |
14 (37.8) |
12 (30.8) |
0.516 |
|
APACHE II score |
22.7 ± 7.2 |
24.6 ± 6.6 |
0.221 |
Table 2.Variables about suctioning and hemodynamic factors
|
Closed-suction catheter with pressure valve |
Conventional closed-suction catheter |
p value |
|
Sputum appearance, n (%) |
|
|
0.924 |
|
Yellowish |
18 (48.6) |
20 (51.3) |
|
|
Whitish |
10 (27.0) |
9 (23.1) |
|
|
Blood tinged |
9 (24.3) |
10 (25.6) |
|
|
Sputum amount, n (%) |
|
|
0.541 |
|
Small |
12 (32.4) |
9 (23.1) |
|
|
Moderate |
23 (62.2) |
26 (66.7) |
|
|
Large |
2 (5.4) |
4 (10.3) |
|
|
Number of suction for 48 hr |
29.2 ± 6.3 |
29.1 ± 5.4 |
0.932 |
|
Maximal heart rate |
118.5 ± 21.5 |
120.5 ± 23.6 |
0.693 |
|
Occurence of arrhythmia, n (%) |
8 (21.6) |
9 (23.1) |
0.879 |
|
Mean arterial pressure (mmHg) |
102.1 ± 15.6 |
100.2 ± 16.0 |
0.592 |
|
Minimal O2 saturation (%) |
92.6 ± 6.6 |
92.9 ± 5.3 |
0.856 |
|
Mean partial pressure O2 (mmHg) |
115.0 ± 53.2 |
98.9 ± 55.9 |
0.203 |
|
Mean partial pressure CO2 (mmHg) |
38.7 ± 13.5 |
36.2 ± 6.8 |
0.328 |
|
Maximal inspiratory pressure (cmH2O) |
15.0 ± 6.0 |
14.4 ± 5.7 |
0.649 |
Table 3.Tracheal mucosal injury and ICU outcomes
|
Closed-suction catheter with pressure valve |
Conventional closed-suction catheter |
p value |
|
Tracheal mucosa injury, n (%) |
|
|
0.001 |
|
Gr 0 |
9 (31.0) |
2 (6.5) |
|
|
Gr 1 |
15 (51.7) |
9 (29.0) |
|
|
Gr 2 |
5 (17.2) |
8 (25.8) |
|
|
Gr 3 |
0 |
9 (29.0) |
|
|
Gr 4 |
0 |
3 (9.7) |
|
|
Sputum culture positive, n (%) |
22 (59.5) |
23 (59.0) |
0.966 |
|
Blood culture positive, n (%) |
4 (10.8) |
2 (5.1) |
0.358 |
|
Total ICU stay (d) |
13.0 (6.5–24.0) |
12.0 (7.0–32.0) |
0.877 |
|
ICU mortality, n (%) |
12 (32.4) |
12 (30.8) |
0.876 |
References
- 1). Brandstater B, Muallem M. Atelectasis following tracheal suction in infants. Anesthesiology 1969;31:468-73.ArticlePubMed
- 2). Cabal L, Devaskar S, Siassi B, Plajstek C, Waffarn F, Blanco C, et al. New endotracheal tube adaptor reducing cardiopulmonary effects of suctioning. Crit Care Med 1979;7:552-5.ArticlePubMed
- 3). Simbruner G, Coradello H, Fodor M, Havelec L, Lubec G, Pollak A. Effect of tracheal suction on oxygenation, circulation, and lung mechanics in newborn infants. Arch Dis Chil 1981;56:326-30.Article
- 4). American Association for Respiratory Care. AARC clinical practice guidelines. Endotracheal suctioning of mechanically ventilated patients with artificial airways 2010. Respir Care 2010;55:758-64.PubMed
- 5). Shim C, Fine N, Fernandez R, Williams MH Jr. Cardiac arrhythmias resulting from tracheal suctioning. Ann Intern Med 1969;71:1149-53.ArticlePubMed
- 6). Johnson KL, Kearney PA, Johnson SB, Niblett JB, MacMillan NL, McClain RE. Closed versus open endotracheal suctioning: Costs and physiologic consequences. Crit Care Med 1994;22:658-66.ArticlePubMed
- 7). Cereda M, Villa F, Colombo E, Greco G, Nacoti M, Pesenti A. Closed system endotracheal suctioning maintains lung volume during volume-controlled mechanical ventilation. Intensive Care Med 2001;27:648-54.ArticlePubMed
- 8). Maggiore SM, Lellouche F, Pigeot J, Taille S, Deye N, Durrmeyer X, et al. Prevention of endotracheal suctioning-induced alveolar derecruitment in acute lung injury. Am J Respir Crit Care Med 2003;167:1215-24.ArticlePubMed
- 9). Lee ES, Kim SH, Kim JS. [Effects of a closed endotracheal suction system on oxygen saturation, ventilator-associated pneumonia, and nursing efficacy]. Taehan Kanho Hakhoe Chi 2004;34:1315-25.ArticlePubMed
- 10). Cha KS, Park HR. Endotracheal colonization and ventilator-associated pneumonia in mechanically ventilated patients according to type of endotracheal suction system. J Korean Acad Nurs 2011;41:175-81.ArticlePubMed
- 11). Oh J, Whang K, Jung H, Park J. Comparison of the pattern in semi-quantitative sputum cultures based on different endotracheal suction techniques. Korean J Crit Care Med 2012;27:70-4.Article
- 12). Jongerden IP, Kesecioglu J, Speelberg B, Buiting AG, Leverstein-van Hall MA, Bonten MJ. Changes in heart rate, mean arterial pressure, and oxygen saturation after open and closed endotracheal suctioning: A prospective observational study. J Crit Care 2012;27:647-54.ArticlePubMed
- 13). Lorente L, Lecuona M, Martín MM, García C, Mora ML, Sierra A. Ventilator-associated pneumonia using a closed versus an open tracheal suction system. Crit Care Med 2005;33:115-9.ArticlePubMed
- 14). Siempos II, Vardakas KZ, Falagas ME. Closed tracheal suction systems for prevention of ventilator-associated pneumonia. Br J Anaesth 2008;100:299-306.ArticlePubMed
- 15). Link WJ, Spaeth EE, Wahle WM, Penny W, Glover JL. The influence of suction catheter tip design on tracheobronchial trauma and fluid aspiration efficiency. Anesth Analg 1976;55:290-7.ArticlePubMed
- 16). Burton G, Hodgkin J, Ward J. Respiratory care: a guideline to clinical practice. New York, Lippincott Williams & Wilkins. 1997, pp 555-609.
- 17). Blackwood B. The practice and perception of intensive care staff using the closed suctioning system. J Adv Nurs 1998;28:1020-9.ArticlePubMed
- 18). Lindgren S, Almgren B, Högman M, Lethvall S, Houltz E, Lundin S, et al. Effectiveness and side effects of closed and open suctioning: An experimental evaluation. Intensive Care Med 2004;30:1630-7.ArticlePubMed
- 19). Lasocki S, Lu Q, Sartorius A, Fouillat D, Remerand F, Rouby JJ. Open and closed-circuit endotracheal suctioning in acute lung injury: Efficiency and effects on gas exchange. Anesthesiology 2006;104:39-47.ArticlePubMed
- 20). Ramanathan S, Sinha K, Arismendy J, Chalon J, Turndorf H. Humidification and airway pressures during high-frequency jet ventilation delivered through the suction-biopsy channel of a flexible bronchofiberscope. Crit Care Med 1984;12:820-3.ArticlePubMed
Citations
Citations to this article as recorded by

- “Study on Device System to Reduce Tracheal Mucosal Injury in Intubation Patients” [ASME Journal of Medical Devices, 2022, 16(3), p. 031006; DOI: 10.1115/1.4054334]
Journal of Medical Devices.2022;[Epub] CrossRef - Efficacy of the Closed Suction Applied To Patients In Intensive Care Units with Different Techniques: A Nonrandomized Controlled Trial
Zuhal GÜLSOY, Şerife KARAGÖZOĞLU
Cumhuriyet Medical Journal.2020;[Epub] CrossRef










 KSCCM
KSCCM
 PubReader
PubReader ePub Link
ePub Link Cite
Cite