Effectiveness of a daytime rapid response system in hospitalized surgical ward patients
Article information
Abstract
Background
Clinical deteriorations during hospitalization are often preventable with a rapid response system (RRS). We aimed to investigate the effectiveness of a daytime RRS for surgical hospitalized patients.
Methods
A retrospective cohort study was conducted in 20 general surgical wards at a 1,779-bed University hospital from August 2013 to July 2017 (August 2013 to July 2015, pre-RRS-period; August 2015 to July 2017, post-RRS-period). The primary outcome was incidence of cardiopulmonary arrest (CPA) when the RRS was operating. The secondary outcomes were the incidence of total and preventable cardiopulmonary arrest, in-hospital mortality, the percentage of “do not resuscitate” orders, and the survival of discharged CPA patients.
Results
The relative risk (RR) of CPA per 1,000 admissions during RRS operational hours (weekdays from 7 AM to 7 PM) in the post-RRS-period compared to the pre-RRS-period was 0.53 (95% confidence interval [CI], 0.25 to 1.13; P=0.099) and the RR of total CPA regardless of RRS operating hours was 0.76 (95% CI, 0.46 to 1.28; P=0.301). The preventable CPA after RRS implementation was significantly lower than that before RRS implementation (RR, 0.31; 95% CI, 0.11 to 0.88; P=0.028). There were no statistical differences in in-hospital mortality and the survival rate of patients with in-hospital cardiac arrest. Do-not-resuscitate decisions significantly increased during after RRS implementation periods compared to pre-RRS periods (RR, 1.91; 95% CI, 1.40 to 2.59; P<0.001).
Conclusions
The day-time implementation of the RRS did not significantly reduce the rate of CPA whereas the system effectively reduced the rate of preventable CPA during periods when the system was operating.
INTRODUCTION
The rapid response system (RRS) has matured since in the 1990s, especially after the Institute for Healthcare Improvement initiated the campaign “100,000 Lives” in 2004 [1]. The concept of RRS is a rapid response to patients who exhibit at least one abnormal clinical sign that is exhibited prior to an in-hospital cardiac arrest; responding adequately to such clinical signs may prevent adverse events and decrease in-hospital mortality or cardiopulmonary arrest (CPA) [2,3]. Given that many preventable events in hospital occur because of the mismatch between hospital resources and patient’s needs, the RRS has been widely operated [4].
Meta-analyses have demonstrated that RRS is associated with decreasing non-intensive care unit (ICU) CPA incidence and hospital mortality [5,6] although previous studies have reported conflicting results [7,8]. A recent study showed that the part-time RRS also reduced the CPA incidence during RRS operating time in general ward patients [9]. Furthermore, the 2015 American Heart Association-Cardiopulmonary Resuscitation (CPR) guidelines have included and emphasized the role of a rapid response team (RRT) or a medical emergency team to ensure appropriate surveillance of in-hospital cardiac arrest and improve patient survival [10].
RRS structure primarily consists of four components; the afferent limb, the efferent limb, patient safety and process improvement element, and the administrative component [3,4]. However, the system composition depends on several factors such as the availability of hospital resources, organizational support, cultural aspects, the target patient population, etc. Therefore, the consensus for an optimal structure of a RRS has not yet been established [11]. This study aimed to investigate the effectiveness of daytime RRS for surgical hospitalized patients with limited resources and administrative support.
MATERIALS AND METHODS
Study Design and Patients
This study was a single-center retrospective cohort study to investigate the effectiveness of day-time RRS. We conducted this study at Seoul National University Hospital, a 1,779-bed tertiary care teaching hospital in Korea. Data were compared 2 years before-RRS periods (August 2013 to July 2015) with 2 years after-RRS periods (August 2015 to July 2017). Surgical patients hospitalized in 20 surgical wards comprising 641 beds were included. Patients who are admitted to the ICU, emergency department, and internal medicine wards were excluded because there was limitation of human resources and the RRS alert electronic medical records (EMRs) system was not installed. Additionally, as a RRS was reported to be effective for decreasing CPA in surgical patients in a recent study [12], and since the number of doctors who can care for patients in the general wards was limited, we decided to focus on patients in surgical wards who needed cover during the day. After the patients were discharged from the ICU to surgical wards, we followed up on these patients and monitored them until they were fully recovered. This study was approved by Institutional Review Board of Seoul National University Hospital (IRB No. 1708-007-874) and informed consent was waived.
Data Collection and Endpoints
The primary endpoint was the incidence of non-ICU CPAs per 1,000 admissions before and after the RRS implementation during the RRS hours of operation (weekday, 7 AM to 7 PM). The secondary outcomes were the incidence of total and preventable CPA; in-hospital mortality, frequencies of invoking a do-not-resuscitate (DNR) order (including instances after RRS activation), and the survival-to-discharge rates of patients with CPA.
Two independent, trained researchers reviewed EMRs and collected data using formal data extraction forms. Patient characteristics such as age, sex, Charlson comorbidity score, Acute Physiology and Chronic Health Evaluation II (APACHE II) score, CPA etiology, presence of pre-CPA alarm signs, number of CPAs, number of ICU admissions, ICU lengths of stay, and hospital lengths of stay were recorded. The data were compared between the two reviewers for consensus, with a third reviewer arbitrating if any disagreements arose [13].
The monthly occurrences of RRS activation, admissions and the number of major surgeries were retrieved, as were the case-mix indices (CMIs). The CMI is the relative value assigned to the diagnostic group of patients in the medical environment as well as the resource allocation for care and treatment of patients in the group; it represents the diversity or complexity of the hospital [14].
Daytime RRS in Surgical Wards
In August 2015, our hospital launched an RRS for adult patients, called Seoul National University Hospital Alert Valid Responder, which constituted a multidisciplinary team comprising seven intensivists who had dual certification for both critical care medicine and their own specialty (i.e., pulmonology, anesthesiology, surgery including thoracic/cardiovascular, and neurology specialists) and two experienced critical care nurses. One nurse with a certification for advanced critical care nursing had 7 years of working experience in a surgical ICU and other nurse had 8 years’ experience in a medical ICU. The seven intensivists had over 5 years of experience in critical care medicine. All doctors had additional rotating RRS duties besides of their duties; while two nurses were dedicated to RRS duties. The system was partially introduced in 20 general surgical wards with 641 beds during the daytime (i.e., from 7 AM until 7 PM) on weekdays; the RRS did not operated outside of regular working hours due to a limited workforce and lack of administrative support. Our team planned to expand the system gradually after verifying its effectiveness, and also planned to change the organizational culture and orient it further towards patient safety. There were no administrative changes in the nursing program or ward staffing during the pre- and post-RRS implementation periods at the hospital.
Before RRS implementation, the team advised nurses, doctors, and other hospital staff members to contact the RRS when a patient’s condition deteriorated. After RRS deployment, weekly meetings for reviewing RRS activation events, CPA, and unplanned ICU admissions were held to improve the efficiency of the system. Through the weekly meetings, preventable CPA was defined as CPA that could be detected several hours before the adverse event using pre-alarm signs and avoidable through appropriate and prompt medical management. Our teams tracked the time and date of CPA events to determine any pre-alarm signs existing which may have existed within 48 hours of events and to judge whether the events were potentially avoidable or not. The pre-alarm signs were abnormal vital signs based on RRS trigger criteria (Supplementary Table 1), including altered mental status and abnormal laboratory results (i.e., severe abnormal hydrogen ions or electrolyte imbalances). Potentially preventable CPA includes (1) missed signs or symptoms of pending CPA, (2) delayed responses or inappropriate treatment, (3) medication-related CPAs, and (4) procedure-related complication.
RRS triggering criteria, pre-CPA alarm signs, and workflow
Our hospital’s RRS afferent limb consisted of bedside medical staff’s making a direct phone call and the RRS’ dedicated nurses selective proactive rounding as the EMR screening program had not been implemented at the early stage (i.e., from August 2015 to October 2016). The detailed criteria for triggering the RRS included consideration of patients’ symptoms (i.e., respiratory, cardiovascular, neurological, or other symptoms), abnormal vital signs (Supplementary Table 1), and any other concerns raised by the bedside medical staff regarding a patient’s deteriorating condition.
Proactive rounding was carried via review of medical records for high-risk patients, physical assessment, and connection with a critical value reporting system that automatically sends a message to RRS members’ phone regarding a patient’s abnormal clinical test results from diagnostic examinations. High-risk patients who required proactive rounding were selected by a consensus by the RRS members including ICU discharge within 48 hours, patients with tracheostomy, more than stage 4 patients through Korean Patient Classification System-1 [15], and gynecologic or urologic cancer patients who received chemotherapy in surgical wards [16]. RRS nurses made high-risk patients list for proactive rounding every day, then checked the patient’s vital signs, laboratory results, doctor’s progress notes, and nurse’s records to select the patients who needed further management. When RRS nurses received calls from the ward staff about deteriorating patients, it was defined as RRS activation. If RRS nurses decided to act for further patient management during proactive rounding, these cases were defined as RRS proactive activation. Both call and proactive rounding activation resulted in: (1) transferring patients to the ICU; (2) consultation by RRS members; (3) delivering an advanced intervention by the RRS members at wards; (4) education to the ward staff; (5) observation on the ward, and; (6) accompanying ill patients to another location from the wards. Additionally, the team developed an electrical medical records screening program for the efficient running of the system, and the screening program has been operating since November 2016. The program’s triggering variables were the abnormal respiratory rate, oxygen saturation, heart rate, systolic blood pressure, body temperature, and urinary output. Detailed criteria are in Supplementary Table 1. Once afferent limb triggers the RRS using the above method, the RRS nurses begin initial assessments and deliver primary interventions. If the nurses judge that the patient needed management by RRS doctors, they notify the patient’s status to RRS doctors and devise an additional management plan. The team fulfils patient’s unmet needs and provides resources to stabilize and also triage the location depending on patients’ clinical progress. After stabilization from the event or relocating the patients, the team follow-up the patients for a period to check the full recovery. The RRS team not only care the deteriorating patients but also educate the health care provider in general wards and also deliver the end-of-life counseling. The RRS team advise ward doctors to give palliative care or DNR order to the activated patients who are in irreversible courses of deterioration. Our CPR team operates separately from the RRS team. The CPR team consists of two residents from medical ICU, one resident of anesthesiology, and wards staffs. After launching of RRS, RRS nurses joined the team to improve the efficacy of CPR.
Statistical Analysis
Data were analyzed using IBM SPSS ver. 23.0 (IBM Corp., Armonk, NY, USA). A Mann-Whitney test was used for analyzing the continuous variable demographic characteristics of patients with an arrest. For categorical variables, the chi-square and Fisher-exact tests were used to compare the data between the two groups. The Student t-test was used for normally distributed continuous variables such as admissions per month and case-mix index. Continuous variables are presented as mean and standard deviation or median (interquartile range), and categorical variables are presented as frequency (percentage). We estimated the relative risk (RR) of CPAs, in-hospital mortality, and DNR documentation using Poisson regression analysis to compare between pre- and post-RRS implementation. The results are presented as the RR and 95% confidence intervals (CIs); a P-value <0.05 was regarded as statistically significant.
RESULTS
A total of 61,315 patients during the pre-RRS period and 75,119 patients during the post-RRS period were admitted to the surgical ward with a 23% increase in patients between the two periods. A total of 19,705 major surgeries were performed during the study period (before and after RRS implementation, 9,510 cases vs. 10,195 cases; P<0.001) (Supplementary Table 2). The CMI as represented by hospital complexity (1.10 and 1.09, respectively; P=0.950) (Table 1) and the ward medical staff such as nurses and residents were comparable between the two periods (Supplementary Tables 3 and 4).
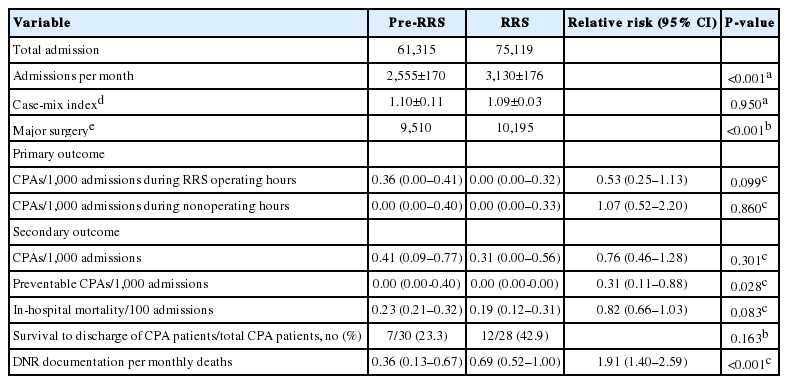
During the 2 years after RRS implementation, there were 1,580 RRS activations in 75,119 patients, for an activation rate of 21.0/1,000 admissions. The trends of RRS activation counts per month are represented in Figure 1. The rates of RRS call activation decreased from 35% to 12% during the 2 years of RRS implementation whereas rates of proactive rounding activation by RRS nurses and via EMR screening increased from 65% to 88%. The proactive rounding activation using the EMR screening program and high-risk patient list by RRS members was more frequent than the call by ward staff. Mean total RRS activation per month was 21.00±10.72 per 1,000 admissions. Mean RRS proactive rounding activation and call activation cases per 1,000 admissions were 15.90±9.99 and 5.10±1.82, respectively. The mean number of activation cases significantly increased after the EMR screening program operation (15.67± 5.68 vs. 29.89±11.47, P=0.001).
Monthly trends of rapid response system (RRS) activations per 1,000 admissions during the pre-RRS and RRS implementation periods. The blue bars represent the RRS call activation count per 1,000 admissions after RRS implementation (mean±standard deviation, 5.10±1.82). The orange bars represent the RRS proactive rounding (PR) activation count per month (15.90±9.99). The green circles and lines represent the total RRS activation counts per month (21.00±10.72). RRS activation cases included transferring a patient to the intensive care unit, RRS members delivering an advanced intervention, consultation, education, observation, or accompanying the transfer of ill patients to another location from the wards. EMR: electronic medical record.
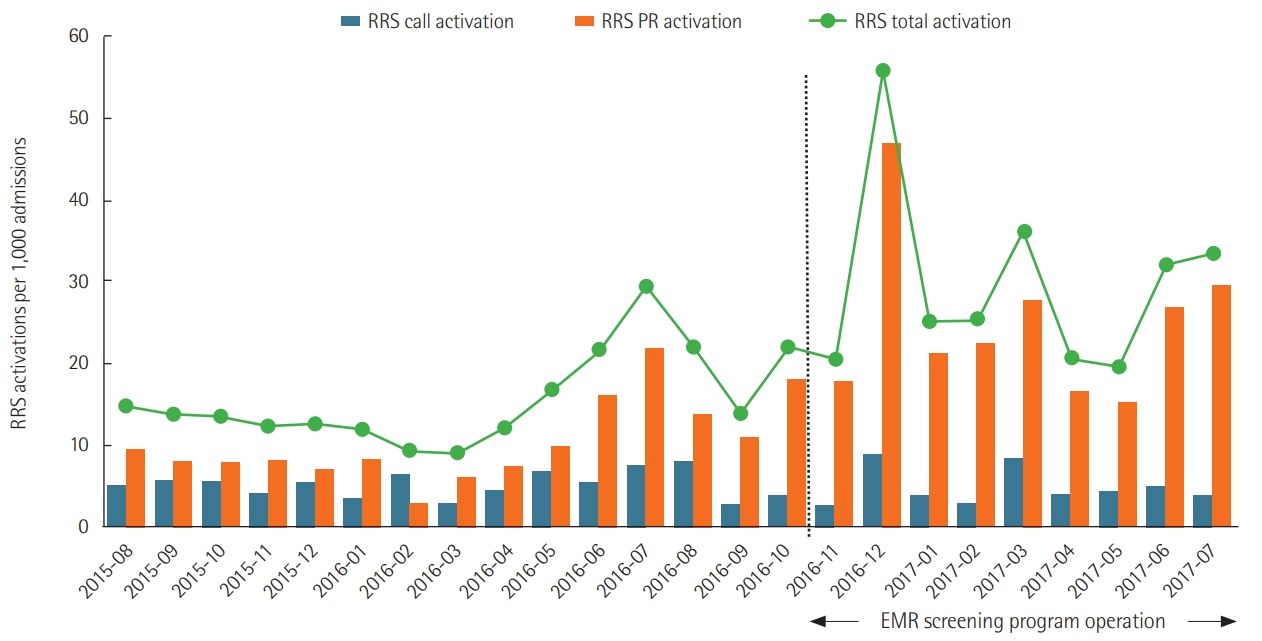
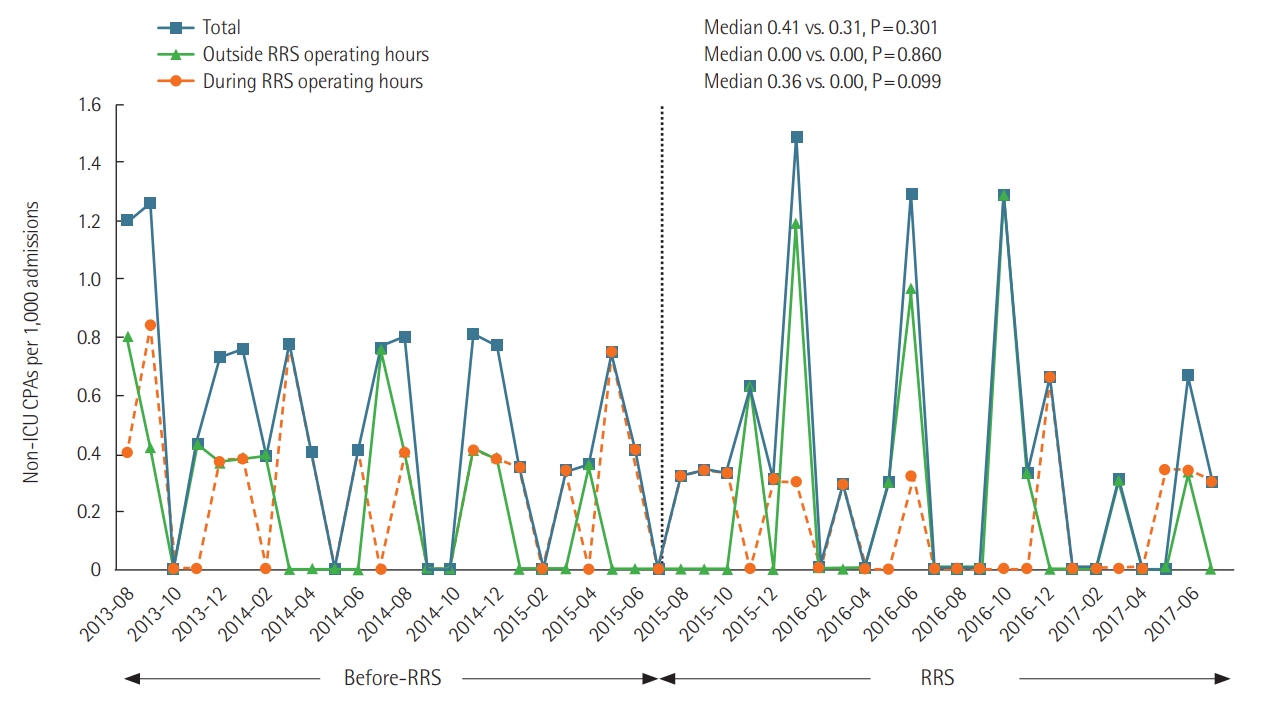
A total 58 non-ICU CPA occurred during study periods (30 during pre-RRS for 2 years vs. 28 during post-RRS for 2 years). The RR of CPA during RRS hours of operation in the RRS implementation period compared with the pre-RRS implementation period was 0.53 but this was not statistically significant (95% CI, 0.25 to 1.13; P=0.099) (Table 1, Figure 2). The total non-ICU CPA incidence per 1,000 admissions regardless of RRS operating hours did not significantly decrease after RRT implementation (RR, 0.76; 95% CI, 0.46 to 1.28, P=0.301) (Table 1, Figure 2). The RR of in-hospital mortality per 100 admissions in the RRS periods compared with the pre-RRS periods was 0.82 (95% CI, 0.66 to 1.03; P=0.083). The incidence of preventable CPA per 1,000 admissions after RRS implementation was 69% lower than before its implementation (RR, 0.31; 95% CI, 0.11 to 0.88; P=0.028) (Table 1, Figure 3). Moreover, the decision to implement a DNR order significantly increased during after RRS implementation period compared to the pre-RRS periods (RR, 1.91; 95% CI, 1.40 to 2.59; P<0.001) (Table 1).
Incidence of non-intensive care unit (ICU) cardiopulmonary arrests (CPAs) per 1,000 admissions. The blue squares and lines represent the total incidence of non-ICU CPAs per 1,000 admissions during the study periods regardless of rapid response system (RRS) operating hours (median, 0.41 vs. 0.31; P=0.301). The orange circles and dotted lines represent the incidence of non-ICU CPA per 1,000 admissions during RRS operating hours before and after implementation (median, 0.36 vs. 0.00; P=0.099). The green triangles and lines represent the incidence of non-ICU CPA per 1,000 admissions outside RRS operating hours before and after RRS implementation (median, 0.00 vs. 0.00; mean, 0.21 vs. 0.22; P=0.860).
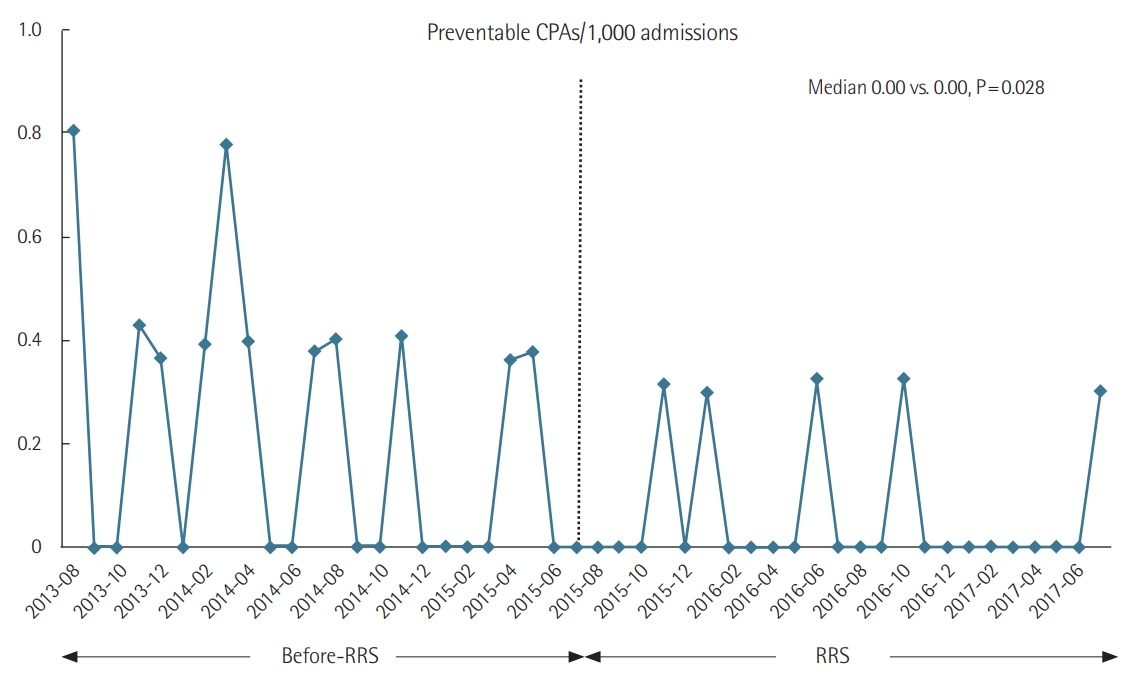
Incidence of preventable non-intensive care unit (ICU) cardiopulmonary arrests (CPAs) per 1,000 admissions. The diamonds and lines represent the incidence of preventable non-ICU CPA per 1,000 admissions before and after rapid response system (RRS) implementation (median, 0.00 vs. 0.00; mean, 0.21 vs. 0.07; P=0.028).
The characteristics of CPA cases are summarized in Table 2. The pre-CPAs alarm signs within 24 hours and 48 hours were significantly decreased post-RRS periods than in pre-RRS periods (P<0.001). The characteristics of CPA patients such as age, sex, Charlson comorbidity score, cause of CPAs, and number of resuscitations of spontaneous circulation were not significantly different between the pre-RRS and RRS periods (Table 2). In addition, ICU and hospital length of stay, APACHE II score of ICU-admitted patients, and in-hospital survival of those patients were also comparable (Table 2).
DISCUSSION
The main findings of our study are as follows: first, the daytime RRS did not significantly decrease the incidence of non-ICU CPA events during the RRS operating hours. Second, this system decreased the incidence of preventable CPA. Third, implementation of a daytime RRS was not associated with reduced in-hospital mortality in surgical ward patients; however, it was associated with increased frequency of invoking a DNR order.
The RRS has been widely implemented to improve patients’ clinical outcomes or safety [17] in various healthcare settings. However, the optimal RRS models and procedures remain unclear [18]. Hospital administrations are burdened by the cost and time required to adopt RRS procedures, although it is argued that adopting an RRS would pay for itself by reducing the rate of adverse events [19]. Moreover, there are many impediments to maintaining the system efficiently, including time constraints [3].
The total incidence of non-ICU CPA decreased 24%, in the first 2 years of RRS operation and the incidence of non-ICU CPAs during RRS operating hours decreased by 47% after RRS implementation but it was not statistically significant. A recent study showed that a part-time RRS effectively decreased the incidence of CPA in all hospitalized patients, with the decrease more prominent in non-internal medicine patients [9]. Another study found that employing RRS full-time in surgical patients decreased the incidence of CPA although not significantly so; this was consistent with our findings [20]. Possible reasons for such discrepancies are the different RRS activation processes or the ward staff’s lack of adherence to the RRS protocol [21]. Another possible reason could be that the baseline incidence of CPA in surgical patients was quite low in our hospital compared with previous studies [12,20]. These earlier studies which reported a significant reduction of CPA according to RRS adoption, had a higher baseline CPA incidence than reported in the present study [22]. Therefore, more time may be needed to prove the effectiveness of RRS by collecting sufficient samples. Another method to show improvement was to improve the ward staff’s awareness of RRS for the afferent limbs [21]. The total RRS activation and proactive rounding activation count was increased after use of EMR surveillance programs over time, whereas the call activation count was comparable. According to the reports, a direct call from bedside staff is important as afferent limb; however, the percentage of calls was low in the early roll-out of the RRS [23]. The call activation needs a sufficient period of time to mature for a RRS, especially in Korea’s hierarchical culture [24]. Therefore, proactive rounding with EMR screening programs or active manual surveillance with high-risk patients are needed to compensate for low doses of calls and delayed calls, and it could lead to successful implementation in the early stage of RRS.
When we reviewed the CPA cases during study periods, the preventable CPA incidence decreased by 69% after the RRS implementation. Moreover, the presence of pre-alarm signs within 24 and 48 hours before CPAs were significantly reduced. CPAs could potentially be prevented by avoiding the following: delays in the appropriate diagnosis and treatment, inadequate approaches to the patient’s status, incomplete treatment, inexperienced medical staff, and unclear medical decisions for interventions ranging from palliative care to active resuscitations while considering the patient’s medical history [25]. The decreased incidence of avoidable CPA at our hospital after RRS implementation, as well as the reduced pre-alarm signs before CPAs, indicated that our repeated education toward staff and active surveillance by RRS members would have reduced the preventable CPA through early recognition and appropriate management. For the afferent and efferent limbs to function adequately in a timely manner, continuing education and cultural change for safety are needed [26].
In-hospital mortality at our hospital was not associated with RRS implementation inconsistent with previous studies [27,28]. The advanced treatment for deteriorating patients and decision of transferring to ICUs, considering on mismatch patients demands and resources, could improve clinical outcomes. However, according to a prospective study, 2 years are required to verify that the incidence of CPA is decreasing owing to an RRS, and 4 years are required to statistically confirm a decreased in-hospital mortality rate [29]. Our pre- and post-RRS implementation study periods were relatively short for this study and the mortality data were not statistically adjusted according to other variables. To show more favorable outcomes post-RRS implementation in terms of lower rates of mortality, long term investigation is needed and statistically adjusting for other confounding factors is also recommended.
The ward staff was made more aware of DNR decisions through continuous education and recommendations by the RRS team members. As a result, the decision of DNR significantly increased after RRS implementation. Data from a previous study had suggested that the physicians’ confidence levels regarding conducting DNR discussions with patients are relatively low, and that additional efforts are required in this regard [30]. Hence, the RRS can provide patients an opportunity to improve their end-of-life standards by facilitating communication regarding DNR directives and changing the organizational culture [31].
Another consequence of implementing a daytime RRS is that the survival-to-discharge rate could be improved by including dedicated RRS nurses in traditional code teams. The survival-to-discharge rates of patients with CPA increased from 23% before RRS implementation to 43% after implementation although this change was not statistically significant. Our hospital’s code team runs on an ad hoc basis during crises; team members may never have worked together until a crisis occurred. Moreover, the code team members at our hospital did not include senior staff. For this reason, the trained RRS nurses could serve as good coordinators and promote communications during a crisis.
As mentioned above, several outcome measures were improved following the introduction of our daytime RRS, even though it was implemented only during a specific time and in certain departments. The daytime RRS model could be an alternative option in other hospitals that lack administrative support and/or have a shortage of human and financial resources.
Our study had some limitations. First, the retrospective design may have introduced bias regarding the interpretation of the data. Second, we conducted this study in surgical patients at a single hospital; therefore, our results might not be generalizable to other patients and settings such as medical patients or those in ICUs. Third, it is possible that other unexamined factors decreased the incidences of preventable CPA between the pre-RRS and RRS periods. Therefore, the results should be accepted with caution.
In conclusion, daytime RRS implementation did not reduced the incidence of CPA during operating times for surgical ward patients. However, the system reduced the incidence of preventable CPA and increased evoking DNR documentation. Therefore, daytime RRS may be helpful to decrease preventable CPA and avoid unnecessary resuscitation at the moment of the end of life.
KEY MESSAGES
▪ The rapid response system is associated with decreasing incidences of preventable non-intensive care unit cardiopulmonary arrest in surgical hospitalized patients despite limited resources and administrative support.
▪ Do-not-resuscitate documentation was needed to avoid unnecessary resuscitation for a quality of end-of-life increased after daytime rapid response system implementation.
Notes
CONFLICT OF INTEREST No potential conflict of interest relevant to this article was reported.
AUTHOR CONTRIBUTIONS
Conceptualization: HL, SML, SYO. Data curation: SK. Formal analysis: EY, HL. Methodology: SK, HL, JL. Writing–original draft: EY. Writing–review & editing: HL, SML, HGR, HJL, JL, SYO.
Supplementary Materials
Criteria for triggering the rapid response system for rapid response team activation
Number of major operations before and after rapid response system implementation
Number of nurses before and after rapid response system implementation
Number of residents before and after rapid response system implementation