The effects of direct hemoperfusion with polymyxin B-immobilized fiber in patients with acute exacerbation of interstitial lung disease
Article information
Abstract
Background
Acute exacerbation of interstitial lung disease (AE-ILD) causes clinically significant deterioration and has an extremely poor prognosis with high mortality. Recently, several studies reported the effectiveness of direct hemoperfusion with a polymyxin B-immobilized fiber column (PMX-DHP) in patients with AE-ILD as a potential therapy. This study describes the clinical effectiveness and safety of PMX-DHP in patients with AE-ILD.
Methods
We retrospectively reviewed the medical records of 10 patients (11 episodes) with AE-ILD treated with PMX-DHP from January 2018 to June 2019. We compared laboratory and physiologic data of the ratio of partial pressure arterial oxygen to fraction of inspired oxygen (P/F ratio) and level of inflammatory markers before and after implementation of PMX-DHP.
Results
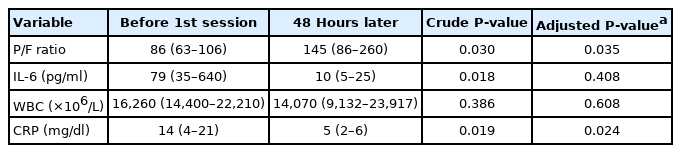
Ten patients were included according to the 2016 revised definition of acute exacerbation of idiopathic pulmonary fibrosis (IPF). Nine patients had IPF and one patient had fibrotic nonspecific interstitial pneumonia. Most patients (90.9%) were treated with a steroid pulse, and four patients (36.4%) were treated with an immunosuppressant. The median number of PMX-DHP cycles was 2, and the median duration of each cycle was 6 hours. After PMX-DHP, the mean P/F ratio improved (86 [range, 63–106] vs. 145 [86–260], P=0.030) and interleukin-6 and c-reactive protein decreased (79 [35–640] vs. 10 [5–25], P=0.018 and 14 [4–21] vs. 5 [2–6], P=0.019, respectively). The 30-day mortality rate was 27.3% and the 90-day mortality rate was 72.7%.
Conclusions
PMX-DHP treatment improved P/F ratio and reduced inflammatory markers in AE-ILD patients.
INTRODUCTION
Acute exacerbation of interstitial lung disease (AE-ILD) is a life-threatening event with very poor prognosis [1]. The mortality for idiopathic pulmonary fibrosis (IPF) is estimated at greater than 50%, with a median survival of only 1–4 months [2-5]. Despite increasing recognition and intervention efforts for AE-ILD, no treatment has demonstrated clinical benefit. Corticosteroids and immunosuppressive drugs have been used to treat AE-ILD patients, but new therapies with high efficacy are needed.
Polymyxin B-immobilized fiber column (PMX-DHP) was originally developed to adsorb endotoxins released by Gramnegative bacteria during septic shock [6]. Other studies have found that PMX-DHP might be useful for patients with acute respiratory distress syndrome and acute lung injury, which are pathologically characterized by diffuse alveolar damage [7,8]. The actual mechanism of PMX-DHP in AE-ILD patients is not understood well. However, an in vitro study recently suggested that absorption of various cytokines involved in inflammation, fibrosis, and vascular permeability might be associated with the positive impacts of PMX-DHP treatments [9]. Several recent reports have shown that PMX-DHP treatment has beneficial effects on oxygenation, inflammatorymarker removal, and long-term outcomes in AE-ILD patients [10-12]. However, the clinical outcomes and survival benefits have not been accurately evaluated. In addition, most previous studies were performed in Japan. Therefore, the aim of this study was to evaluate the clinical effects and safety of PMX-DHP in Korean patients with AE-ILD.
MATERIALS AND METHODS
This study was approved by the Institutional Review Board of Haeundae Paik Hospital (IRB No. 2019-08-021), and the requirement for written informed consent was waived due to the retrospective nature.
Study Subjects
Clinical data from 10 AE-ILD patients (11 AE episodes) who were treated with PMX-DHP from January 2018 to June 2019 at Haeundae Paik Hospital, Busan, Republic of Korea, were retrospectively analyzed. Diagnosis of ILD was based on international guidelines [13,14]. The diagnostic criteria for AE-ILD based on Collard et al. [15] were as follows: (1) previous or concurrent IPF diagnosis; (2) acute worsening or development of dyspnea typically within the past month; (3) high-resolution computed tomography (HRCT) with new bilateral ground-glass opacity and/or consolidation superimposed on a background pattern consistent with a typical interstitial pneumonia pattern; and (4) deterioration not fully explained by cardiac failure or fluid overload [15].
Clinical Information
Baseline characteristics and survival data were obtained from medical records. Spirometry, diffusing capacity of the lung for carbon monoxide (DLCO), and forced vital capacity (FVC) were determined according to recommendations from the American Thoracic Society (ATS) and the European Respiratory Society and are reported as percentage of normal predicted value [16,17]. The 6-minute walk test (6MWT) was performed according to ATS guidelines [18]. IPF severity was classified according to gender-age-physiology (GAP) models, which were used to predict IPF progression [19].
PMX-DHP Treatment
After AE-ILD diagnosis, patients were treated with a corticosteroid pulse (500 mg or 1 g of methylprednisolone over 1 day) and antibiotics as conventional therapy. Simultaneously, PMX-DHP treatment was started after diagnosis in patients with severe AE-ILD. PMX-DHP treatment was performed for 6 hours per day over two consecutive days using a PMX column (Toraymyxin 20R; Toray Medical, Tokyo, Japan). Before use, a PMX-fiber cartridge was rinsed with 4 L physiological saline and primed with 2,000 U heparin sodium dissolved in 0.5 L saline. Blood access for direct hemoperfusion with PMX-DHP was obtained using a double-lumen catheter inserted into the jugular or femoral vein. The PMX-fiber cartridge was connected to a polymethyl methacrylate membrane (Hemofeel CH-1.0N, Toray Medical) that returned blood to the femoral vein. The hemoperfusion flow rate was 120–150 mL/min. Nafamostat mesilate (NM; Toril Pharmaceuticals, Tokyo, Japan) was administered into a continuous renal replacement therapy circuit as a regional anticoagulant.
Assessment of PMX-DHP Effects
Serum inflammatory markers of white blood cells (WBCs), c-reactive protein (CRP), interleukin (IL)-6, and the ratio of partial pressure arterial oxygen to fraction of inspired oxygen (P/F ratio) were measured before and 48 hours after PMX-DHP treatment. IL-6 level was measured by electrochemiluminescence immunoassay using the Cobas e411 analyzer (Hitachi High-Technologies, Tokyo, Japan). We compared the results before and 48 hours after PMX-DHP treatment.
Statistical Analysis
Data are presented as frequency with percentage for categorical variables and as median and interquartile range for continuous variables. Paired t-test or Wilcoxon’s signed-rank test was performed for comparison between two assessment time points. To determine normal distribution, we used the Shapiro-Wilk test. The generalized linear mixed model was employed for longitudinal data to adjust covariates. Cox’s multivariate regression models were fit to identify prognostic factors independently related to overall survival, which was estimated using the Kaplan-Meier curve with spaghetti plots for data visualization. All statistical analyses were performed using IBM SPSS ver. 24.0 (IBM Corp., Armonk, NY, USA), and P-values < 0.05 were considered statistically significant.
RESULTS
Baseline Clinical Characteristics
Ten patients (11 episodes) were included in this study. Nine patients (10 episodes) were clinically diagnosed with IPF and one patient was diagnosed with fibrotic nonspecific interstitial pneumonia by surgical lung biopsy. The median patient age was 66 years (range, 62–74 years), and seven patients (63.6%) were male. The median duration from diagnosis of ILD to acute exacerbation of ILD was 13 months (range, 3–32 months). Three patients (27.3%) had a history of lung cancer. Most patients showed moderately restrictive lung function (median FVC, 51% of predicted; range, 47%–66%) and low DLCO (median, 37% of predicted; range, 35%–46%) on pulmonary function testing. Functional capacity (total distance and de-saturation during 6MWT) was poor. According to GAP stage, IPF severity was classified as stage I, II, or III, with stage II being most common (54.5%). The median baseline Sequential Organ Failure Assessment (SOFA) score was 5 (range, 4–6), and six patients (54.5%) suffered shock during their initial intensive care unit admission. At AE-ILD diagnosis, the median initial P/F ratio was 86 (range, 64–107), representing severe hypoxemia, and 90% of patients received steroid pulse therapy, while 36.4% were concomitantly treated with immunosuppressants. During treatment with PMX-DHP, six patients (seven episodes) required mechanical ventilator support, and four patients were supported by high-flow nasal cannula (HFNC). After treatment with PMX-DHP, four patients (36.4%) were treated with venovenous extracorporeal membrane oxygenation (ECMO) therapy, one of whom survived (Table 1).
Details and Effects of PMX-DHP
The median time from AE-IPF onset to PMX-DHP application was 2 days (range, 0–3 days). The median number of PMX-DHP cycles was 2 (range, 2–3), and the median cycle duration was 6 hours (range, 6–8 hours). The median total duration of PMX-DHP treatment was 12.0 hours (range, 12–24 hours). NM was used as an anticoagulant in all patients, and there were no complications associated with hemoperfusion. At 48 hours after treatment, IL-6 and CRP decreased (Table 2), change in WBC was not significant, and the P/F ratio was improved significantly (86 [range, 63–106] vs. 145 [range, 86–260], P=0.030) compared to pretreatment values (Figure 1). However, after adjusting for mechanical ventilation, use of steroid pulse therapy, steroid dose, and use of immunosuppressant, improvement of P/F ratio and decrease of CRP remained significant, but change in IL-6 did not.

Comparison of white blood cell (WBC), C-reactive protein (CRP), interleukin (IL)-6, and the ratio of partial pressure arterial oxygen to fraction of inspired oxygen (P/F ratio) at 48 hours after polymyxin B-immobilized fiber column (PMX-DHP) treatment. (A) WBC count, (B) CRP, (C) IL-6, (D) P/F ratio. Serum levels of CRP and IL-6 were decreased after 48 hours of PMX-DHP. The P/F ratio was improved after 48 hours of PMX-DHP. There was no change in WBC count at after 48 hours of PMX-DHP. The horizontal bars indicate median values.
Survival Analysis
During the follow-up period, the survival rate from the time of AE onset using Kaplan-Meier analysis was 72.7% at 30 days and 27.3% at 90 days (Figure 2). The median survival period was 48 days (range, 8–570 days). Among four patients who received ECMO, 1 (25%) was weaned from treatment and survived for 570 days. The most common cause of death was acute respiratory failure including pneumonia (57.1%), followed by multi-organ failure (28.6%) and cardiovascular events (14.3%).
DISCUSSION
In our study, the clinical effects of PMX-DHP in AE-ILD patients were similar to those reported previously. PMX-DHP improved the P/F ratio and removal of inflammatory markers compared with pre-PMX-DHP treatment levels. Despite clinical improvement including P/F ratio and removal of inflammatory markers, mortality was high and clinical improvement did not result in survival for most patients. There were no adverse events associated with PMX-DHP treatment.
Several previous studies reported that PMX-DHP treatment reduces many kinds of inflammatory markers [6,20,21]. Oishi et al. [22] reported that, in nine AE-IPF patients who were treated with PMX-DHP, serum CRP, IL-9 (median, 35.6 pg/ml vs. 28.2 pg/ml; P = 0.011), and IL-12 (median, 38.8 pg/ml vs. 12.6 pg/ml; P = 0.004) significantly decreased after treatment. In a study including 14 patients (20 episodes) treated with PMX-DHP, Enomoto et al. [23] reported a significant change in neutrophil count (1,095 ± 1,649 vs. 6,166 ± 1,406, P = 0.031) and angiopoietin-2 before and after PMX-DHP. Our study also confirmed the effectiveness of PMX-DHP as an inflammatory-marker reducer, where we found significant differences in IL-6 and CRP but not in WBC. Among many inflammatory markers or cytokines, we examined CRP and serum IL-6 because IL-6 was previously established as a useful biomarker for AE-ILD given its roles as a soluble mediator with pleiotropic effects on inflammation, immune response, and fibrosis [24]. Papiris et al. [25] compared cytokine levels between 23 stable IPF and 19 AE-IPF patients and reported that high IL-6 level is characteristic of AE-IPF, and that increased IL-6 is associated with worse outcomes.
Previous studies found that PMX-DHP treatment improved the P/F ratio in AE-ILD patients. Our study also showed that PMX-DHP treatment had a tendency to improve the P/F ratio. A previous study of five AE-IPF patients who received PMX-DHP corroborates our findings of improved P/F ratio (170.0 ± 119.4 vs. 217.1 ± 129.6, P = 0.02) [26]. The actual mechanism of oxygenation improvement by PMX-DHP is not understood fully, but Abe et al. [27] suggested that active and inflammatory neutrophils that are absorbed by the PMX column play a role.
Our 30-day mortality rate was 27.3% and the 90-day mortality rate was 72.7%, which is higher than in other reports [28,29]. In our study, the pre-PMX-DHP treatment P/F ratio was very low (median, 86; range, 63–106), suggesting that our patient population was critically ill despite rapid application of PMX-DHP (median time from AE-IPF onset to PMX-DHP: 2 days). In addition, all patients were treated for severe respiratory failure with mechanical ventilation and HFNC in the intensive care unit. We assume that the disease severity of our enrolled AE-ILD patients might be the reason for our high mortality rate. In addition, clinical improvement did not result in longterm survival in most patients. This could be because short-term clinical improvement was not enough to avoid continuous mechanical ventilation or ECMO care, which is critical to survival. However, the effect of clinical improvement from PMX-DHP on survival benefit was uncertain in this study. As a pilot study, this will be helpful for additional large-scale and well-designed studies in the near future in Korea.
This study has some limitations. First, this was a small retrospective study at a single center. However, patients were treated with almost the same protocol, including PMX-DHP and conventional AE-ILD treatment by a single pulmonologist. All patients were treated with PMX-DHP within 4 days of AE-ILD onset. This study was composed of data gathered within a twoyear period, reducing bias from varied medical conditions over time. Second, there was no control group (without PMX-DHP treatment), so it was difficult to accurately assess the effect of PMX-DHP treatment. Third, we could not examine all inflammatory markers and cytokines that could be important. It is possible that other inflammatory markers and cytokines are relevant to AE-ILD pathogenesis.
In conclusion, PMX-DHP treatment for AE-ILD patients showed improvement in the P/F ratio and a decrease in inflammatory markers. For some patients, these improvements in clinical aspects might lead to better clinical outcomes. To confirm the clinical effects and survival benefits of PMX-DHP in AE-ILD patients, more studies are needed, including a double-blind, randomized, controlled study.
KEY MESSAGES
▪ Polymyxin B-immobilized fiber column (PMX-DHP) treatment for acute exacerbation of interstitial lung disease (AE-ILD) patients improved the ratio of partial pressure arterial oxygen to fraction of inspired oxygen and decreased inflammatory markers, which might improve clinical outcomes.
▪ Further studies including a double-blind randomized controlled study are needed to confirm the clinical effects and survival benefit of PMX-DHP in AE-ILD patients.
Notes
CONFLICT OF INTEREST
No potential conflict of interest relevant to this article was reported.
AUTHOR CONTRIBUTIONS
Conceptualization: JHL, HJJ. Data curation: JHL, JHP, JHJ, SHK, WH. Formal analysis & Methodology: JHL, JHP, HJK, HKK, YKK, IHK. Visualization: JHL, JHP, BSP, SHP. Writing–original draft: JHL, JHP. Writing–review & editing: all authors.