Assessment and Treatment of Pain in Adult Intensive Care Unit Patients
Article information
Abstract
In most cases, patients admitted to an intensive care unit (ICU) have suffered from severe trauma, undergone major surgery or been treated for a serious medical illness. Although they often experience more intense pain than general ward patients, they are frequently unable to communicate their experiences to health care providers, thus preventing accurate assessment and treatment of their pain. If appropriate measures are not taken to treat pain in critically ill patients, stress response or sympathetic overstimulation can lead to complications. The short-term consequences of untreated pain include higher energy expenditure and immunomodulation. Longer-term, untreated pain increases the risk of post-traumatic stress disorder. Because pain is quite subjective, the accurate assessment of pain is very difficult in the patients with impaired communication ability. The current most valid and reliable behavioral pain scales used to assess pain in adult ICU patients are the Behavioral Pain Scale and the Critical-Care Pain Observation Tool. Once pain has been accurately assessed using these methods, various pharmacologic and non-pharmacologic therapies should be performed by the multidisciplinary care team. Accurate assessment and proper treatment of pain in adult ICU patients will improve patients outcome, which reduces the stress response and decreases the risk of post-traumatic stress disorder.
Introduction
The pain management is a key aspect of medical professionalism. Since the American Pain Society declared “pain: the fifth vital sign” in 1995, medical professionals became more aware of the importance of management of a wide variety of pain symptoms in their patients, and their level of interest in research on pain management methods have increased.[1] Patients in intensive care unit (ICU) are vulnerable to severe pain as a result of major surgery, severe trauma and serious medical illness requiring invasive monitoring, mechanical ventilation and prolonged hospitalization.[2–4] In addition, even routine nursing care, such as dressing change and airway suctioning can be the source of substantial pain for ICU patients.[2–4] ICU patients are in greater need of adequate pain management, compared with general ward patients. However, ICU patients may not receive adequate care and treatment for the pain they go through because of several reasons. In fact, medical professionals may have difficulties identifying and evaluating pain in adult ICU patients, and their pain is often undetected or underestimated in many cases.[5–7] Any difficulty in identifying and evaluating pain in adult ICU patients appears to be associated with adult ICU patient’s limited ability to communicate.[8] Patient’s communication abilities are further limited when they are intubated. Payen et al.[2] estimated accurate assessment of pain was made in less than 40% of total mechanically ventilated patients. ICU patient’s alertness is also affected by the administration of sedatives and/or analgesics and disease itself progression. Thus most ICU patients may have difficulty communicating the experience of pain and describing the intensity of pain, making it difficult or impossible for medical professionals to evaluate their pain accurately.[9]
The present article aimed to describe adverse effects that may occur in adult ICU patients when their pain is not properly controlled. The article also addressed pain assessment tools, which have shown to be valid in evaluating pain in adult ICU patients with communication difficulties, and the effectiveness of both pharmacologic and non-pharmacologic management techniques available for them.
The Importance of Adequate Pain Management in ICU
Stein-Parbury and McKinley[10] reported in their prospective study that most ICU patients suffer pain. To manage the pain properly that ICU patients feel, accurate assessment and appropriate treatment of the pain is the most crucial factor. If pain in ICU patients is not properly managed, short-term side effects such as higher energy expenditure and immunomodulation can occur.[11,12] In addition, the long-term side effects of uncontrolled persistent pain can include even post-traumatic stress disorder.[13]
International Association for the Study of Pain (IASP) defines pain as an unpleasant sensory and emotional experience associated with actual or potential tissue damage, or described in terms of such damage.[14] As shown in this definition, pain is quite subjective, meaning that pain can be described very differently among patients suffering the same medical condition. Nonetheless, certain physiologic signs are commonly anticipated in patients when their pain is not controlled properly. And these responses may result in further adverse effects. Uncontrolled pain can cause deleterious systemic effects on the body by activating neuroendocrine stress, sympathetic overstimulation, excessive catecholamine release and inducing general stress response.[15,16] Then physiological reactions to uncontrolled pain include anxiety, tachycardia, diaphoresis and catabolic metabolism. As indicated in Table 1, those physiological reactions can lead to increased cardiovascular oxygen consumption, decreased gastrointestinal motility, tachypnea, altered pulmonary mechanics, water and sodium retention as a result of activation of the renin-angiotensin-aldosterone axis and cytokine production. Persistent chronic pain, when it remains unrelieved, will cause manifold effects, including immune system dysfunction, hypercoagulable states and an increase in thromboembolic disease, altered glucose control, myocardial ischemia, ventilator dyssynchrony, acute restrictive respiratory physiology, and disrupted sleep quality.[17,18] In addition, ICU patients can experience functional impairment as a result of pain-induced respiratory dysfunction and failure of early ambulation, resulting in poor patient outcome and increased mortality risk.[19]
Most adult ICU patients had major surgery, severe trauma or serious medical illness. Their life-threatening critical condition acts as a potent stimulus to sympathetic nervous system (SNS).[20] In response to any stimulus, the SNS triggers the fight-or-flight or stress response, which is the most efficient physiological mechanism contributing to our survival.[21] The stimulation of SNS is also crucial to critically ill patients because it boosts oxygen supply into tissues and allows the vital organs remain intact. However, overactive SNS or persistent SNS stimulation is commonly present in adult ICU patients with critical illness. Chronic overstimulation of the SNS activates the stress response beyond tolerable time period and adaptive scope that exert beneficial effects, causing damaging effects.[20] In particular, the stress response system easily gets out of control when the SNS remains overstimulated during critical illness and causes adverse side effects.[20] Although the ICU is designed to provide intensive care for critically ill patients, the ICU environment itself can be the primary factor inducing stress responses in adult ICU patients. In the critical care setting, noise, sleep deprivation, disturbance of circadian rhythms, a lack of natural light and loss of entitlement increase stress in patients.[22]
Recently, the relationship between the SNS and post-traumatic stress disorder (PTSD) has drawn attention. A study claimed 20% to 25% of ICU patients developed PTSD.[23] Also, PTSD patients were characterized by the SNS chronically activated over the normal baseline levels.[24,25] PTSD patients also show elevated urinary norepinephrine[26] and significantly higher level of norepinephrine concentrations in cerebrospinal fluid, compared with the control group.[27] Norepinephrine concentration levels were correlated with the severity of PTSD symptoms, according to the latter study. The findings of increased noradrenergic activity among PTSD patients suggest a decrease in central nervous system noradrenergic activity would effectively treat arousal symptoms such as nightmares and startle reactions.[28]
Pain Assessment for Adult ICU Patients
As mentioned above, pain is defined as a subjective experience by IASP. That is, patients should be able to describe the pain they are experiencing to have their pain assessed accurately and treated properly. At the same time, the intensity of pain felt can vary among patients even when the source of their pain is identical. While patient’s subjective descriptions of pain characteristics are required for accurate assessment of their pain, many patients in the ICU have impaired communication ability, meaning that they may be unable to express their pain experience although their critical physical and mental conditions amplify their pain. Thus accurate pain assessment can be difficult when patients are impaired in their ability to communicate.[9] In fact, it is known that adult ICU patients experience pain on a daily basis even during routine care. Procedural pain is also common in the ICU.[29] Therefore, accurate understanding of their chronic and severe pain should be the first step in managing pain effectively. In addition to the essential understanding of the presence of pain, a routine and repetitive systemic assessment approach is necessary to evaluate the intensity and characteristics of the perceived pain.[29–31] As the IASP described, “the inability to communicate verbally does not negate the possibility that an individual is experiencing pain and is in need of appropriate pain-relieving treatment,” it can be wrong if medical professionals do not provide adequate pain management in adult ICU patients who are not able to describe the pain in the mistaken assumption that they have no pain.[14] Routine pain assessment leads to improved clinical outcomes, and the newly released clinical practice guidelines of the Society of Critical Care Medicine (2012 SCCM guideline) strongly recommends the implementation of routine pain assessment.[29] In particular, a standardized pain management protocol can significantly reduce sedative dose, the length of ICU stay and the length of intubation period.[2,32]
Ideally, pain assessing practices in the ICU should identify the characteristics of pain, including location, duration, type, intensity and exacerbating and relieving factors, if possible, not to mention the presence or absence of pain.[33] There are also subtypes of pain, which are categorized as somatic, visceral, neuropathic and mixed. Because these subtypes also affect pain characteristics and management, they should be identified.[30] Their pain is mostly somatic pain, which is typically dull and aching pain and often well-localized while responds well to opioids and non-steroidal anti-inflammatory drugs (NSAIDs). Visceral pain occurs when bowels are not properly treated and when there is underlying gastrointestinal pathology. This abdominal pain is cramping and colicky but responds promptly to anticholinergics. Although it is not well documented in ICU patients, neuropathic pain also exists in symptoms of burning and shooting pain along the distribution of the affected nerve root. Antidepressants and anticonvulsants are commonly used for the management of neuropathic pain.[30,33]
It is however difficult for ICU physicians to predict the onset of pain and clarify the correlation between the new-onset pain and physiological changes such as hypertension, tachycardia and tachypnea.[29] Therefore, patient’s self-reporting is regarded as the gold standard of pain assessment.[9,34] Pain scales such as visual analog pain scale (VAS) and numerical rating pain scale (NRS) are used to measure intensity of pain in patients with self-report abilities.[35] These tools cannot be applied to many adult ICU patients because they are unable to self-report with limited verbal capabilities.
American Society for Pain Management Nursing recommended proper treatment after the cause of pain is identified through the meticulous evaluation of pain-related behaviors of patients being intubated or having cognitive deficits.[36] Since these patients tend to have communication difficulties, their vital signs are sometimes used as alternative pain indicators. However, the 2012 SCCM guidelines recommend not using vital signs alone for pain assessment in critically ill adult patients.[29] Nonetherless, it does not mean that changes in vital signs are useless for pain assessment. It is a well-known fact that changes in vital signs can provide important clues to enable closer examination or diagnosis.[36,37] Medical professionals therefore rely on behavioral measures when evaluating pains in adult ICU patients with inability to communicate verbally.[5,9,37,38] Behavioral measures in cognitively impaired adults examine common pain-related responses or actions, including muscle tension, differences in body postures, startling facial expression, psychomotor agitation and patient-ventilator dyssynchrony (bucking the ventilator).[39–41]
When ICU patients’ motor function remains intact and their pain behavior is observable except brain injury patients, even though they cannot provide a self-report, the 2012 SCCM guidelines recommends Behavioral Pain Scale (BPS) and Critical-Care Pain Observation Tool (CPOT) as valid tools for accurately assessing pain.[29] These nonverbal scales proved successful assessing pain among adult ICU patients given the quality of psychometric evidences although the scales still have some limitations.[29] BPS has been most commonly used to assess pain through evaluate facial expression, upper limb movements, ventilation compliance. The BPS score (total score: 3–12) is the sum of three behavioral domains that had a range of score of 1–4.[42] Although BPS is known to be relatively accurate in evaluating pain in mechanically ventilated patients or those receiving sedation, the scale cannot be applied to non-intubated patients. Behavioral Pain scale-Non-Intubated (BPS-NI) is a modified BPS to evaluate pain in spontaneously breathing patients who cannot self-report. In BPS-NI, compliance with ventilation of BPS is replaced with vocalization.[43] CPOT is useful in assessing pain of spontaneously breathing patients.[44] The CPOT score (total score: 0–8) indicates the sum score of four behavioral domains: facial expression, body movements, muscle tension and compliance with the ventilator or vocalization. Each item is scored from 0 to 2. The item of ‘compliance with the ventilator or vocalization’ makes this tool suitable for assessing pain in spontaneously breathing patients. A CPOT sore of 2 or higher show a sensitivity of 86% and a specificity of 78% when predicting significant pain in postoperative adult ICU patients who underwent a nociceptive procedure.[40,45] These sensitivity and specificity levels of the CPOT are considered similar to a BPS score of greater than 5, making it possible to predict that nonverbal adult ICU patients have significant pain during nociceptive procedure, compared with patients at rest.[32]
While studies conducting pain assessment based on pain-related behaviors are still not enough, either BPS or CPOT has become the mainstay of pain assessment in ventilated patients. CPOT is widely used for pain assessment in spontaneous breathing patients who cannot self-report. Separately, BPS-NI is regarded as an effective tool for assessing pain in non-intubated patients unable to self-report due to delirium and subsequent cognitive impairment.[5]
Pain Treatment for Adult ICU Patients
Pain management methods for adult ICU patients are largely classified into pharmacologic and non-pharmacologic methods.[29] However, the most important requisite for pain management is to check whether there are any other possible causes of pain before undertaking any of these therapies.[3,4] Identification of potential causes of pain should be prioritized. Once other causes of pain are identified, medical professionals should implement corrective actions to remove them. Their actions may include patient repositioning, stabilization of injury and/or fractures, removal of noxious or irritating stimuli (e.g., proper positioning of ventilator tubing to avoid traction on the endotracheal tube), and application of heat or cold.[3,4] If pain remains persistent even after doing corrective actions properly, physicians can undertake either pharmacologic or non-pharmacologic therapy.
1) Pharmacologic therapies
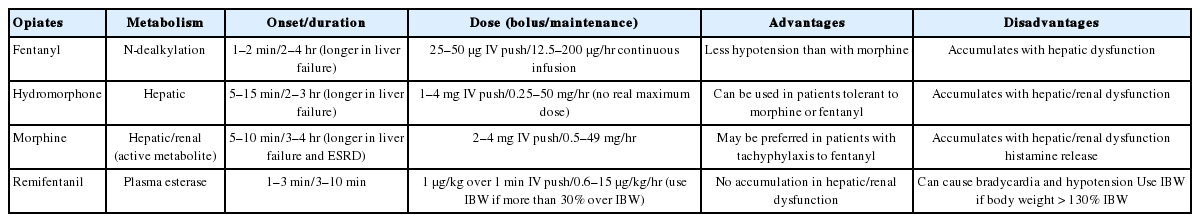
Medical professionals may be surprised to learn that one of the most painful and distressing procedures for adult ICU patients is turning.[46] Nonetheless, adult ICU patients are rarely premedicated to undergo pain provoking procedures such as turning. Patient’s communication difficulties may be one of the reasons for rare premedication. Given limited communication ability among adult ICU patients, pharmacologic therapy through intravenous (IV) route is the most commonly used method for the management of pain in adult ICU patients because it is easy to administer drugs without changing the posture of the patients.[3,47,48] A pharmacologic therapy for pain management should be chosen on the basis of pharmacokinetic and pharmacodynamic properties as described in Table 2. Potential drug-related side effects should be also noted.[31]
The 2012 SCCM guidelines recommends IV opioids such as fentanyl, hydromorphone, morphine, methadone and remifentanil as the first-line treatment for non-neuropathic pain in adult ICU patients.[29] IV opioids are widely used because they are adequately potent without ceiling effects and have mild sedative and anxiolytic effects.[30] These drugs are therapeutically equivalent in terms of analgesic effects. Opioids can cause side effects, and therapeutic doses should be reduced or discontinued in the presence of side effects, which include respiratory depression, altered mental status, decreased gastrointestinal motility, hypotension, nausea and/or vomiting, pruritus, flushing, urinary retention, and delirium.[31] However, these side effects can be very serious, depending on patient condition. As described in Table 2, properties and side effects can vary with the class of opioids, in addition to properties and side effects common to all opioids. Consideration should be therefore given to the expected effectiveness and side effects of opioids before administration.[29] Differences in lipid solubility, volume of distribution, half-life, clearance, metabolism, and active metabolites, caused by different opioids, produce different effects on onset, peak, and offset of action, potency, and the influence of renal or hepatic dysfunction.[31] The use of morphine sulfate requires caution because morphine-6-glucuronide, which is active as much as morphine sulfate as one of major active metabolites of morphine, can be accumulated in the presence of renal failure. Accumulation of morphine-6-glucuronide can cause unnecessarily excessive prolongation of analgesic effect, somnolence and respiratory depression.[49] Fentanyl is a synthetic opioid that achieves rapid equilibration from plasma to lipid-rich brain tissue for its rapid onset of action, which leads to high lipophilicity-induced large volume of distribution and delayed offset of action after prolonged infusion.[50] Remifentanil, one of fentanyl derivates, has small volume of distribution and is rapidly metabolized by esterases, showing short half-life and rapid offset of action regardless renal and hepatic function.[51,52] Hydromorphone has pharmacokinetic and pharmacodynamic profiles similar to those of morphine. As a more potent congener of morphine, hydromorphone produces little histamine release without much adverse effects on the central nervous system, making it an alternative to morphine.[53] Methadone delays development of tolerance when used with other opioids, but its continuous infusion is not possible because of the difficulty in predicting pharmacokinetics.[38] Methadone is a strong medicine that prolongs the rate-corrected QT interval.[54,55] Meperidine has the potential to cause neuroexcitatory adverse effects such as seizure.[49] It is not therefore recommended for ICU patients.[29]
Pain management for adult ICU patients requires accurate knowledge about possible side effects associated with these drugs. At the same time, it is also crucial to avoid unnecessary side effects and treat unavoidable side effects with accurate information. If side effects are intolerable, the dose should be reduced to the lowest effective level.[33] If pain is not relieved by opioids alone or side effects by opioids become intolerable, the use of non-opioids or atypical analgesics is recommended or IV opioids can be replaced by epidural analgesia, other peripheral nerve block techniques, or complementary non-pharmacologic interventions.[31]
Although pain management for adult ICU patients by nonopioid analgesics is supported by limited evidence until now, the 2012 SCCM guidelines suggested the use of non-opioid analgesics for non-neuropathic pain to decrease total quantity of opioids, eliminate the need for IV opioids altogether and decrease opioid-related side effects.[29] Non-opioid analgesics include corticosteroids, local anesthetics (lidocaine, bupivacaine), NSAIDs (ketorolac, ibuprofen), IV acetaminophen, ketamine, adenosine and anticonvulsants. The efficacy of non-opioid analgesics has been proven in postoperative patients and various categories of non-critically ill populations.[29,56–61] However, the efficacy and safety of non-opioid analgesics alone in ICU patients have been less investigated while the previous studies were limited to postoperative settings. Therefore, results cannot be generalized to all ICU patients.[29] The previous studies claimed IV acetaminophen and ketamine were relatively effective to manage pain in ICU patients.[62–64] It is confirmed that IV acetaminophen is effective and safe to control postoperative pain in combination with opioids after major or cardiac surgery. Recently the United States approved use. However, chronic use of IV acetaminophen can increase to the risk of developing hepatotoxicity, and long-term use should be avoided.[30,62,65,66] Corticosteroids, ketamine, and adenosine also have potential benefit as adjuvant due to opioids sparing effects. However, the efficacy of these medications on ICU patients is not supported by enough evidence.[60,61] Ketorolac, one of IV NSAIDs, has anti-inflammatory effects but increases the risk of gastrointestinal toxicity and bleeding, making its use restricted in the ICU setting if patients have the risk of bleeding. In a latter case, Ketorolac should be administered for less than 5 days.[30] Despite its possible role as an adjuvant treatment for complex or neuropathic syndromes, lidocaine is also restricted to use for ICU patients, because of the potential to cause arrhythmia.[67] Clonidine and its isomer dexmedetomidine are alpha agonists having mild sedative and analgesic effects. Dexmedetomidine is increasingly used as a sedative for adult ICU patients breathing spontaneously or weaning from ventilation because it causes less respiratory depression.[68]
To control neuropathic pain in adult ICU patients, enterally administered gabapentin or carbamazepine, in addition to IV opioids, are shown to be more effective than opioids alone, as strongly recommended by the 2012 SCCM guidelines.[29] Drug-based neuropathic pain treatment has been widely explored in non-ICU patients through randomized controlled trials despite some limitations of trials, including small sample sizes and the presence of side effects. Drugs used for neuropathic pain treatment include tricyclic antidepressants, selective serotonin receptor inhibitors, serotonin and norepinephrine reuptake inhibitors, anticonvulsants, tramadol, dextromethorphan, ketamine, topical capsaicin, lidocaine patch or gel and cannabinoids.[69]
When choosing drugs for pain management, dosing method along with types of drug are important.[29] Once the type of drugs is decided, whether the drug will be administered intermittently or continuous IV should be chosen based on comprehensive factors, including pharmacokinetics, pain frequency and severity, and/or patient consciousness.[70] For enteral administration of opioids and other drugs, patient’s gastrointestinal absorptive capacity and motility should be properly maintained.
2) Non-pharmacologic therapies
The use of systemic opioids alone is not often enough to manage pain in adult ICU patients, and sometimes other target-oriented techniques are performed to boost pain relief.[18] In case of an aged patient having difficulty breathing due to pain associated with multiple rib fractures, the use of opioids may increase the risk of too deep sedation and obtundity. But epidural or paravertebral catheterization can be a better choice in that case because it makes respiratory and physical therapy possible at the same time while the patient breathe on his or her own. Regional analgesic techniques are employed when pain is not relieved by opioids alone, pain is well localized or risky side effects are expected as a result of opioids. These techniques are also used when they are considered as effective as IV opioids but their systemic side effects will be far less than opioid-induced side effects.[18,30] In addition to the most commonly used epidural analgesia technique, various peripheral nerve block techniques are available for pain relief in adult ICU patients.[71]
Epidural analgesia (EA) is the best known regional analgesic technique for pain management in adult ICU patients.[71] The 2012 SCCM guidelines recommends thoracic epidural anesthetic/analgesic technique for postoperative pain management in patients exposed to abdominal aortic surgery (AAA) or traumatic rib fractures.[29] However, lumbar EA is not recommended over parenteral opioids in patients exposed to AAA surgery. Also, thoracic EA is not recommended in patients exposed to thoracic or non-vascular abdominal surgical procedures.[29] Neuraxial/regional analgesia is not recommended over systemic analgesia in medical ICU patients.[29] EA in pain management has provided more pain relief and less opioid side effects than IV opioids alone when thoracic EA was combined with bupivacaine and morphine.[72,73] Also, EA led to reduced incidence of postoperative thromboembolism or myocardial infarction and improved bowel and pulmonary function in high risk group of patients.[74] In Cochrane reviews comparing the effectiveness of EA and systemic opioids in patients exposed to elective abdominal surgery, EA not only outpaced opioids in pain relief but reduced the length of intubation period and the incidence of cardiovascular and gastrointestinal complications and acute renal failure.[75] A study that used National Trauma Data Bank revealed a positive correlation between the number of rib fractures and extrapulmonary complications and that mortality risk significantly increased in patients with more than 6 rib fractures, compared with those with less than 6 rib fractures.[18] EA resulted in reductions in morbidity and mortality, particularly in patients with more than 4 rib fractures. Despite these positive benefits, most studies failed to verify the effects of EA on other factors such as ICU stay, hospital stay and mortality. Aside from the above mentioned benefits, EA has some limitations, which make it difficult for medical professionals to utilize for adult ICU patients. Low[76] reported that 89% of 159 units performed epidural analgesia in a postal questionnaire survey conducted with 216 general ICU in England in 2002. However, just only 51 units (32%) used guidelines for EA administration in ICU patients. In fact, there were no consensus guidelines for the management of EA in many cases of bacteremia, sepsis, systemic inflammatory response syndrome or coagulopathy. Because of immunomodulating properties of opioid, EA also increase the risk of infection in the epidural space when performed for ICU patients receiving IV opioids.[77,78] When an epidural catheter is required for prolonged analgesia, EA becomes more difficult to perform, requiring a longer procedure time and increasing risk of infection, depending patient’s condition.
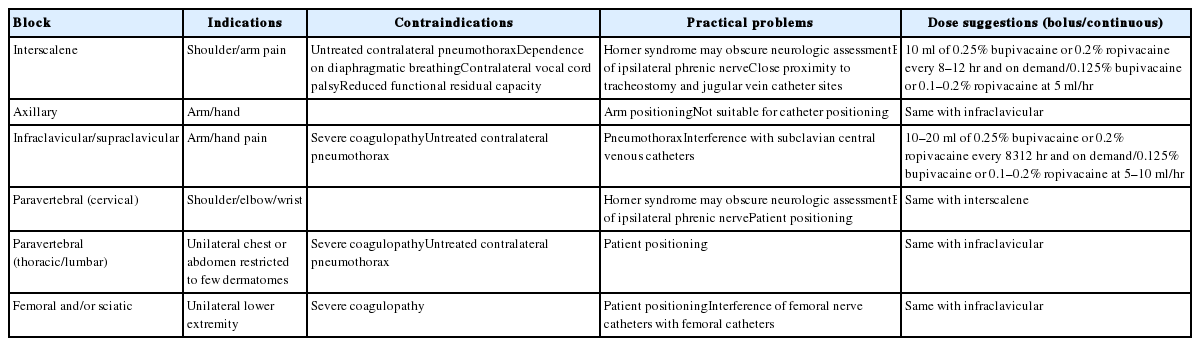
As described in Table 3, there are also peripheral nerve block techniques useful for adult ICU patients. These regional analgesic techniques are particularly effective as a pain management tool for patients who have surgery that required a short operation time.[18] The techniques have also proven effective in morbidly obese patients with obstructive sleep apnea and those who develop hypersensitivity to the respiratory depression with opioids. Nowadays nerve block techniques have become safer and simpler by the use of ultrasound as an aid. A limited number of peripheral nerve block techniques are available for adult ICU patients, because they are able to maintain only supine or lateral decubitus position in many cases.[79] The peripheral nerve blocks that can be performed in the upper part of the body include interscalenene, paravertebral (cervical and thoracic), infraclavicular and axillary blocks. The procedures can be performed in the lower part of the body include paravertebral (lumbar), femoral and sciatic nerve blocks. Indications and contraindications for these procedures are shown in Table 3.
Paravertebral block can be performed on either side of the patient and limit diffusion of anesthetic up to 1 or 2 dermatomes from the needle insertion site, meaning that side effects that may occur with a wide block can be therefore prevented.[18] If a wider diffusion or continuous infusion of anesthetic is necessary, catheterization can be performed. Multiple catheterizations also enable a wider diffusion of anesthetic on one side of the chest. However, there is always a possibility of pneumothorax and hemothorax. Multiple catheterizations are not recommended over EA in patients with localized chest trauma or chest tube induced pain.[18] For patients with rib fractures, intercostal nerve block is effective for pain management when they need large amounts of opioids to relieve severe pain caused by movements because even a single fracture can affect the ability to breath and induces pulmonary complication. When EA is contraindicated, intercostal nerve block can be a good alternative. However, like other regional analgesic techniques, the effectiveness of intercostal nerve block in adult ICU patients is limitedly known. Besides, multiple injections above and below the injured rib are required for proper pain management and duration of analgesic effect is short up to 4 to 8 hours. Intercostal nerve block cannot be continuously infused because of a high risk of complications. In addition, intercostal nerve block can increase the risk of systemic local anesthetic toxicity as a result of higher local anesthetic serum concentrations in patients with more than 1 or 2 rib fractures, compared with those of peripheral or neuraxial nerve blocks.[18] Femoral nerve block is very useful approach for acute preoperative pain after femoral neck fracture occurs.[80,81] Fascia iliaca compartment block can be also employed in that case.[82,83] A combination of femoral nerve block and sciatic nerve block is also used as a anesthetic in surgical treatment like external fixation apart from its role in pain control.[84]
Although clinical outcomes of regional analgesia were not always beneficial, some studies showed good results.[72,75,85,86] A study reported that neuraxial analgesic techniques reduced the length of intubation period and incidence of pneumonia.[87] Although further evidence is required, increasing studies stress a correlation between the use of narcotics and sedatives and occurrence of delirium and cognitive dysfunction.[68,88,89] Regional analgesia can reduce these adverse effects when properly used.[18] To choose an adequate procedure for patients, physicians should make sure patient’s systemic and regional state and have accurate anatomical knowledge on procedure site. Physicians appear to be capable of devising a new technique when they have established enough experiences in nerve block procedures with deep broad knowledge in anatomy and physiology.
Adult ICU patients do not have conditions ideal for regional analgesia, excluding regional anesthesia for postoperative analgesia.[18,30] Most of adult ICU patients cannot maintain body posture required by different anesthetic techniques, and their tissue edema and anatomic deformity may make a needle or catheter placement difficult. Recently advanced ultrasound scanners are lightweight and miniaturized, making them easy to use for patients with limited capability to make movements or postural adjustments. Physicians can carry this portable device to them to perform regional analgesia and check patient’s anatomy via ultrasound on a real time bases.[90,91] Ultrasoundguided peripheral nerve blocks demonstrated high success and low complication rates. When patients undergo opioid-based pain treatment, opioid dose is a major factor to decide, and dose is frequently adjusted or discontinued even in patients, who need substantial amounts of opioids, to prevent side effects. As part of the efforts to prevent side effects of opioids, peripheral nerve blocks were employed, and these procedures successfully controlled pain using small amounts of opioid or no opioid. [92,93] In adult ICU patients, keeping a needle or catheter in place is hard as much as placing them in the right place.[30] A catheter lodged in the right place can be easily dislodged by the patient’s agitation, routine nursing care, radiological test and routine daily activities in the ICU. Nonetheless, catheter malposition cannot be checked in many cases as a consequence of altered states of consciousness, coagulation status and risk of transport. In the perioperative setting, a needle or catheter placement increases the risk of complications in adult ICU patients with coagulopathy or thrombocytopenia and taking drugs that affect hemostasis. This kind of risk can be reduced by consistent check about clotting function. However, catheter-associated complication can happen in the course of treatment of critical illness and even after the placement of potentially risky catheter. With regard to patients receiving anticoagulation therapy or otherwise at increased risk of bleeding, there are regularly updated guidelines available from the American Society of Regional Anesthesia that should be followed for such an regional analgesia.[94]
For long-term pain management, local anesthetic should be continuously infused through a catheter, and infection risk should be clearly considered.[18] As mentioned above, opioids have immunomodulating properties, and a regional analgesia through a catheter requires extra caution in patients taking opioids frequently.[77,78] Regional analgesia should be performed after considering possible infections associated with catheterization or patient’s underlying illness such as sepsis.[18] When Regional analgesia is performed with a continuous catheter, the catheter should be not removed unless there are any signs of an infection.[79]
Table 4 summarizes information that should be noted to perform a regional analgesic technique in adult ICU patient. More importantly, regional anesthetic techniques can be used appropriately for pain management in adult ICU patients provided the following four conditions are met.[18,30] First, the extent to which regional anesthesia influences patient conditions should be accurately considered. Secondly, fully experienced anesthesiologist, pain physician or intensivist trained in anesthesiology and pain medicine is available to perform a variety of anesthetic techniques. Thirdly, well-experienced nurses who are able to identify complications related to surgical procedures or early warning signs of complications are available. Lastly, the ICU should be operated in a systematic way to provide an aseptic condition for catheter placement, regular inspection of devices and early detection of infection. However, these conditions are not met in many ICU settings, and the methods described in Table 3 is not widely used. Above all, there is not enough evidence that support the use of regional analgesia for pain management in the ICU. Further investigations are needed to improve the current pain control practices in the critical care setting, which demands more evidence to accept benefits of regional analgesia and further information to count adverse effects associated with regional analgesic techniques.[30]
In addition to regional analgesic techniques, non-pharmacologic pain treatments also include music therapy, relaxation techniques, and non-pharmacologic complementary interventions like provision of information.[30,31,95–97] These techniques are easy, safe and economic to perform. Regional analgesia is not widely used for ICU patients amid insufficient investigations and lack of evidence to support benefits. However, regional analgesia will provide a new direction toward pain management in adult ICU patients by reducing the use of opioids and improving pain relief.[29,30,33]
Conclusion
The assessment and treatment of pain is not done properly for adult ICU patients, even though they may suffer from quite severe pain, largely because they cannot verbally express their pain. If pain in adult ICU patients can be assessed more accurately based on patient characteristics and treated by multidisciplinary cares, including pharmacologic and non-pharmacologic methods, their pain and stress response can be controlled successfully, leading to a decrease in PTSD.
Notes
No potential conflict of interest relevant to this article was reported.