Predicting factors associated with prolonged intensive care unit stay of patients with COVID-19
Article information
Abstract
Background
Predicting the length of stay (LOS) for coronavirus disease 2019 (COVID-19) patients in the intensive care unit (ICU) is essential for efficient use of ICU resources. We analyzed the clinical characteristics of patients with severe COVID-19 based on their clinical care and determined the predictive factors associated with prolonged LOS.
Methods
We included 96 COVID-19 patients who received oxygen therapy at a high-flow nasal cannula level or above after ICU admission during March 2021 to February 2022. The demographic characteristics at the time of ICU admission and results of severity analysis (Sequential Organ Failure Assessment [SOFA], Acute Physiology and Chronic Health Evaluation [APACHE] II), blood tests, and ICU treatments were analyzed using a logistic regression model. Additionally, blood tests (C-reactive protein, D-dimer, and the PaO2 to FiO2 ratio [P/F ratio]) were performed on days 3 and 5 of ICU admission to identify factors associated with prolonged LOS.
Results
Univariable analyses showed statistically significant results for SOFA score at the time of ICU admission, C-reactive protein level, high-dose steroids, mechanical ventilation (MV) care, continuous renal replacement therapy, extracorporeal membrane oxygenation, and prone position. Multivariable analysis showed that MV care and P/F ratio on hospital day 5 were independent factors for prolonged ICU LOS. For D-dimer, no significant variation was observed at admission; however, after days 3 and 5 days of admission, significant between-group variation was detected.
Conclusions
MV care and P/F ratio on hospital day 5 are independent factors that can predict prolonged LOS for COVID-19 patients.
INTRODUCTION
In 2020, the World Health Organization declared a pandemic state for the coronavirus disease 2019 (COVID-19) after a steep increase in the number of infected patients worldwide, including South Korea [1]. Most patients recover after experiencing mild symptoms accompanied by upper respiratory tract infection; however, approximately 10%–15% of patients require oxygen supply, and approximately 5% are classified as severe patients that require high-flow nasal cannula (HFNC) oxygen delivery, mechanical ventilation (MV), and extracorporeal membrane oxygenation (ECMO) in the intensive care unit (ICU) [2,3].
Medical resources, including ICUs, were prepared around the world in the event of pandemic spread and severe disease. Moreover, studies have reported that during the 2009 H1N1 pandemic, patients’ underlying disease and disease progression, economic status of the pandemic region, and regional characteristics affected mortality [4]. Likewise, during the COVID-19 pandemic, after 2020, mortality varied according to ICU resources in each country [5,6]. However, even in countries or regions that are relatively well prepared for medical emergencies, the steep increase in ICU patients during the pandemic and consequent saturation of resources and overloading of medical staff was associated with a rise in mortality from 28.1% to 65.7% [7,8].
For instance, in South Korea, the number of COVID-19 patients rapidly increased after June 2021 with the emergence of the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) Delta variant. Yet, the number of severe patients was below 200 until June 2021, and then increased to approximately 1,000 at the peak of the outbreak in December 2021. Although mortality remained below 0.2% before the outbreak based on disinfection policies, vaccination, and sufficient medical resources, it increased to 1% on average by December 2021, suggesting that efficient use of ICU resources, including medical instruments and staff, significantly impacted mortality during pandemic conditions [9,10].
To ensure that ICU resources are efficiently distributed, it is necessary to predict the length of stay (LOS) of COVID-19 ICU patients by analyzing disease progression and assessing severity [11,12]. This relationship remains controversial and needs to be clarified in future research, and several studies have shown that the clinical features of COVID-19 differ from acute respiratory distress syndrome (ARDS) [13,14]. Therefore, this study analyzed the clinical characteristics of patients with severe COVID-19 based on their clinical care and identified the predictive factors associated with prolonged ICU LOS of 2 weeks or longer.
MATERIALS AND METHODS
This study was approved by the Institutional Review Board of the National Cancer Center (No. NCC 2022–0215). Informed consent was waived in accordance with the Institutional Review Board due to the retrospective nature of this study.
Study Design and Patients
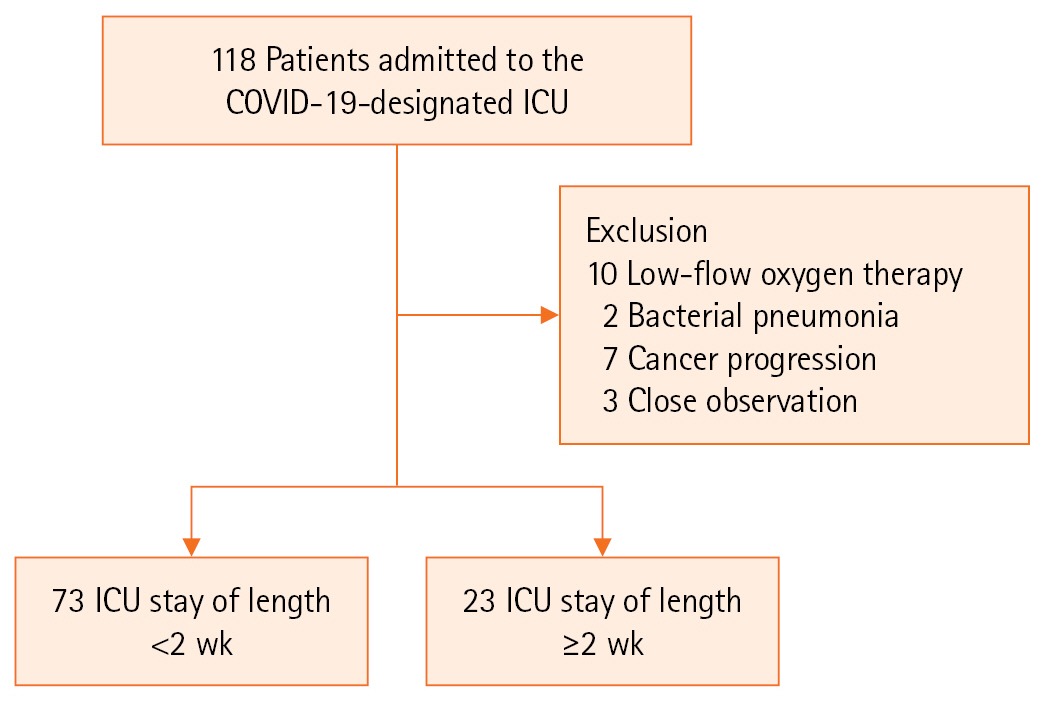
This retrospective study was conducted at a single center. The participants were patients admitted to the ICU due to COVID-19 infection between March 2021 and February 2022. The center had eight beds in the ICU for patients with severe COVID-19 since March 2021. Among the severe COVID-19 patients in the ICU, those who showed a low probability of pneumonia related to COVID-19 infection (bacterial pneumonia, malignancy) among those who received oxygen therapy at the HFNC level or above were excluded through medical review (Figure 1).
ICU admission and discharge criteria for all patients included in this study were defined in accordance with government quarantine policies. All patients admitted to the ICU were administered oxygen therapy at the HFNC level or above at the time of admission; MV or ECMO was applied depending on the stage of pneumonia and ARDS. Additionally, all patients were treated according to the clinical practice guidelines for ARDS and the Sepsis-Surviving Campaign guidelines [15,16]. Treatment included administering remdesivir and steroids, as well as antibiotics and anticoagulants, to prevent secondary bacterial pneumonia. For steroid therapy, 6 mg dexamethasone was administered once a day for 10 days. For ARDS aggravation, despite steroid therapy, dosage was increased to 20 mg once a day for 5 days, with tapering depending on patient status [17]. Tocilizumab 8mg/kg was administered when the oxygen demand increased within 24–48 hours and C-reactive protein (CRP) level was ≥5.0 mg/dL even after starting steroid administration [18]. When the patient showed a decrease in oxygen requirement following ICU care, treatment was switched to a 4 L/min nasal prong. Patients were transferred to the general ward when they were deemed stable based on clinical symptoms, blood test results, and X-ray results. However, considering the possibility of further deterioration, some cases were transferred after 1–2 days of follow-up observation at the discretion of the attending physician.
Data Collection
To identify the factors associated with prolonged ICU LOS ≥2 weeks, patient age, sex, body weight, underlying disease, and smoking history were investigated. Sequential Organ Failure Assessment (SOFA) and Acute Physiology and Chronic Health Evaluation (APACHE) II scores were determined immediately after hospitalization to assess severity. Following ICU admission, blood test results (neutrophil-lymphocyte ratio (NLR), CRP level, D-dimer level, and PaO2 to FiO2 ratio (P/F ratio) on days 3 and 5 of admission were compared to analyze the association between prolonged ICU LOS and changes in blood parameters.
Statistical Analyses
Patient characteristics and laboratory results were summarized as frequencies with percentages for categorical variables. Normality was assessed using the Shapiro-Wilk test. If the assumption of normality was satisfied, continuous variables were presented as mean±standard deviation, and a t-test was used. If the normality assumption was not satisfied, continuous variables were presented as median (range) and the Wilcoxon rank-sum test was used. A logistic regression model was used to identify the prognostic factors associated with a prolonged ICU stay. In the multivariable logistic model, we included all risk factors for prolonged ICU stay with a P-value <0.05, and a risk factor for prolonged ICU stay was selected using backward elimination in the final model. All analyses were performed using SAS software ver. 9.4 (SAS institute).
RESULTS
Patient Characteristics
A total of 118 patients were admitted to the COVID-19-designated ICU, and 96 patients were included in the study after excluding those who did not meet the study criteria (Figure 1). After HFNC treatment, 65 patients showed improvement, while 31 received MV care. Most MV care (28/31, 90%) and continuous renal replacement therapy (CRRT) were performed within 3 days of initial admission. ECMO was performed in five cases on admission to the ICU, and for the remaining three cases, ECMO treatment was applied due to ARDS exacerbation even after 5–10 days of MV care. A neuromuscular (NM) blocking agent was injected in 61.2% (19/31) of patients who received MV care, while 32.3% (10/31) were placed in a prone position. Eight patients (8.3%) showed ARDS aggravation despite MV care, and ECMO was applied. Twenty-three patients (24%) received treatment in the ICU for 2 weeks or more. Of the 96 patients, four died in the ICU due to sepsis from pneumonia aggravation (n=3) and pulmonary hemorrhage during ECMO for ARDS (n=1). All four deceased patients died after being in the ICU for more than 2 weeks. The median ICU LOS was 14 days as a cut off for prolonged ICU LOS (Table 1).
Tracheostomy was performed on nine patients. Of these, seven underwent tracheostomy after undergoing ECMO treatment and two patients due to prolonged intubation (more than 2 weeks). Tracheostomy was performed by an otolaryngologist in the ICU by an open method.
Predicting Factors for Prolonged ICU LOS
Univariable analyses showed that SOFA score at the time of ICU admission, CRP level, high-dose steroids, MV care, CRRT, ECMO, and prone position was significantly correlated (Table 2) with ≥2 weeks of ICU care (prolonged ICU stay). Multivariable analysis showed that MV care and P/F ratio on hospital day 5 were independent factors for prolonged ICU LOS (Table 3).
Laboratory Tests Associated with Prolonged ICU LOS
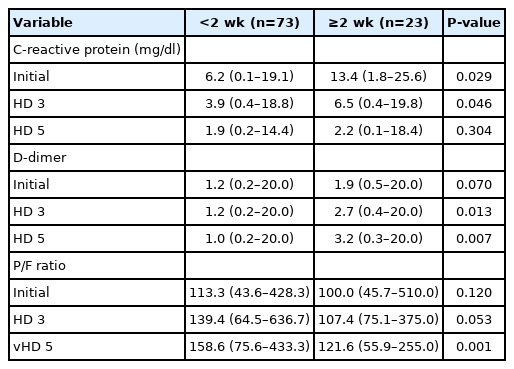
CRP level showed a decrease between days 3 and 5 in both groups, but D-dimer showed a decrease only in the non-ICU LOS group with LOS <2 weeks. The P/F ratio showed an increase between days 3 and 5 in both groups. CRP level was significantly higher in the prolonged ICU LOS patients at 6.2 mg/dl (range, 0.1–19.1) vs. 13.4 mg/dL (range, 1.8–25.6) (P=0.029) at the time of ICU admission; however, the level was reduced on hospital days 3 and 5. For the D-dimer level, no significant variation was observed at ICU admission; however, after days 3 and 5, significant between-group variation was detected (1.0 [range, 0.2–20.0] vs. 3.2 [range, 0.3–20.0], P=0.007). Finally, for the P/F ratio, an increasing trend was observed in the non–ICU LOS group with LOS <2 weeks on days 3, but no significant difference was observed, and after 5 days a significant difference between the two groups was noted (158.6 [range, 75.6–433.3] vs. 121.6 [range, 55.9–255.0], P=0.001) (Table 4).
Medications for Mechanical Ventilator Care Patients
For MV care, dexmedetomidine was preferentially administered as a sedative, and midazolam or propofol was added when additional deep sedation was required during the first few days of MV care according to the attending physician’s discretion. Administration of midazolam (38.5% vs 94.4%, P=0.001) and NM blocking agent (38.5% vs 77.8%, P=0.027) was significantly associated with prolonged ICU LOS. Duration of sedative use (3 [2–5] vs. 7 [3–12], P=0.030) and NM blocking agents (2 [1–3] vs. 6 [4–9], P=0.011) were also statistically significant with prolonged ICU LOS (Table 5).
DISCUSSION
To ensure the appropriate distribution of medical resources and efficient treatments, it is important to determine the current status of medical resource use. During a pandemic, a high burden of patients requiring intensive care is associated with ICU mortality, which places a special emphasis on ICU management [19]. Most previous studies have reported on COVID-19 patient mortality and the risk factors for ICU admission; however, only a few studies have investigated the factors that influence ICU LOS. In this study, the predictive factors associated with prolonged ICU LOS (≥2 weeks) were examined, and MV care alone was observed to be an independent factor, while an increase in the D-dimer level after days 3 and 5 of ICU admission was shown to be significantly correlated with ICU admission.
Age, underlying disease, smoking history, body mass index, APACHE II, and SOFA scores are well-known clinical factors associated with COVID-19 prognosis [20-22]. However, in this study, MV care was the only factor that significantly impacted ICU LOS. MV care is one of the most important treatments for ARDS due to COVID-19, which has been reported to necessitate a greater level of deep sedation than ARDS from other causes [23,24]. Moreover, deep sedation can delay MV weaning and induce muscle weakness and exercise intolerance. Similarly, in this study, midazolam and a NM blocking agent were used to deeply sedate numerous patients, which is presumably why MV care had a greater impact on ICU LOS than any other factor.
Previous studies have reported that CRP and D-dimer levels and NLR and P/F ratios predict the severity and prognosis of COVID-19 patients [25-27]. In this study, the blood parameters measured at the time of ICU admission did not show significant between-group variations while D-dimer levels showed an increasing trend in the prolonged ICU LOS group. Progression of COVID-19 infection is correlated with coagulopathy. Because the D-dimer test is sensitive to measuring coagulopathy, analyzing its dynamic changes may serve as a predictor of prolonged ICU LOS [28,29].
A limitation of this study is that it was conducted as a small-scale, single-center study. Additionally, as a retrospective study, some variables that affect ICU LOS could not be analyzed at the same time points. Also, factors such as nutritional status and sarcopenia that were reported in other studies to influence ICU LOS could not be included. Future studies should investigate ways to reduce MV care duration for ICU rehabilitation, early MV weaning, and nutritional support to reduce ICU LOS and mortality.
MV care and P/F ratio on hospital day 5 are independent factors that predict prolonged ICU LOS in COVID-19 patients. Further studies should investigate ways to reduce MV care duration to reduce ICU LOS.
KEY MESSAGES
▪ Predicting the length of stay of intensive care unit (ICU) patients with coronavirus disease 2019 (COVID-19) is essential for efficient use of ICU resources.
▪ Mechanical ventilator care and the ratio of arterial oxygen partial pressure to fractional inspired oxygen on hospital day 5 are independent factors that can predict prolonged length of stay for COVID-19 patients.
Notes
CONFLICT OF INTEREST
No potential conflicts of interest relevant to this article are reported.
FUNDING
None.
ACKNOWLEDGMENTS
None.
AUTHOR CONTRIBUTIONS
None.
AUTHOR CONTRIBUTIONS
Conceptualization: JHK. Data curation: WHH, JHL, JYC, YJC, YK. Formal analysis: MH. Methodology: JHL. Project administration: JHK. Visualization: WHH. Writing–original draft: WHH. Writing–review & editing: JYC, YJC, YK. All authors read and approved the final manuscript.