Outcomes of patients with COVID-19 requiring extracorporeal membrane oxygenation and continuous renal replacement therapy in the United States
Article information
Abstract
Background
Coronavirus disease 2019 (COVID-19) infection is associated with significant morbidity and mortality. Some patients develop severe acute respiratory distress syndrome and kidney failure requiring the combination of extracorporeal membrane oxygenation (ECMO) and continuous renal replacement therapy (CRRT).
Methods
Retrospective cohort study of 127 consecutive patients requiring combined ECMO and CRRT support in intensive care units at an ECMO center in Marietta, GA, United States.
Results
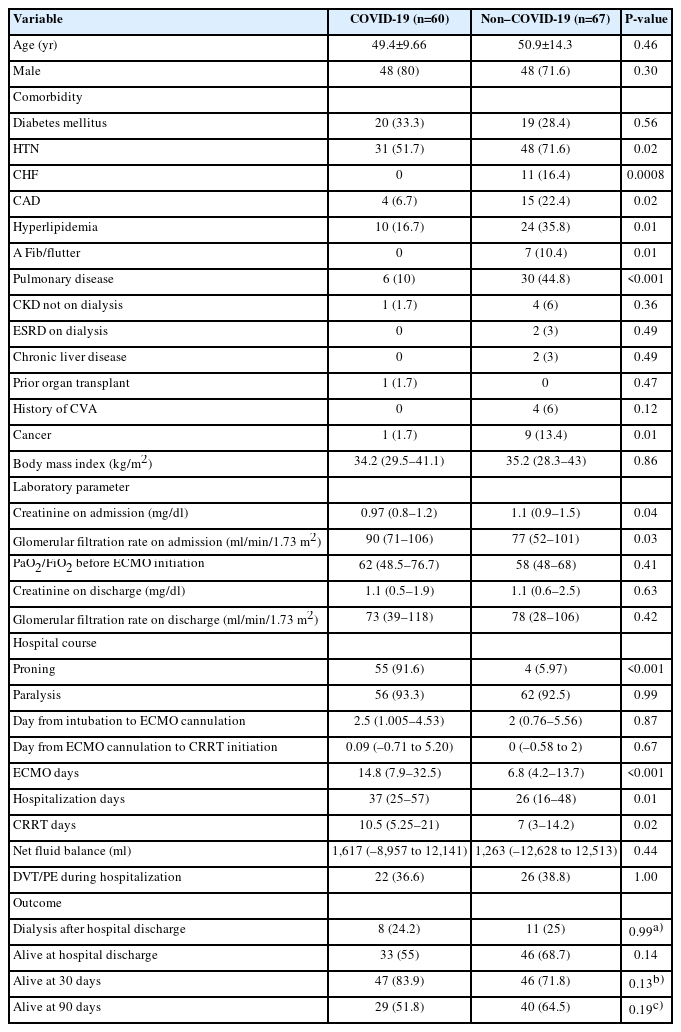
Sixty and 67 patients with and without COVID-19, respectively, required ECMO-CRRT support. After adjusting for confounding variables, patients with COVID-19 had increased mortality at 30 days (hazard ratio [HR], 5.19; 95% confidence interval [CI], 2.51–10.7; P<0.001) and 90 days (HR, 6.23; 95% CI, 2.60–14.9; P<0.001).
Conclusions
In this retrospective study, patients with COVID-19 who required ECMO-CRRT had increased mortality when compared to patients without COVID-19.
INTRODUCTION
Severe acute respiratory syndrome secondary to coronavirus disease 2019 (COVID-19) is associated with significant morbidity and mortality. Unfortunately, some patients develop acute respiratory distress syndrome (ARDS) requiring mechanical ventilation. Despite the best standard of care and lung protective ventilation, a subset suffers from worsening disease and requires extracorporeal membrane oxygenation (ECMO). ECMO is a rescue therapy for patients with severe respiratory failure, refractory cardiogenic shock, and combined cardiorespiratory failure. The use of ECMO has increased significantly over the last couple of years, partly due to the high burden on patients with severe ARDS during the COVID-19 pandemic.
Recent studies have shown that patients with COVID-19 requiring ECMO in the setting of severe ARDS have a longer duration of ECMO support and hospitalization than patients with ARDS due to influenza [1]. A distinct feature of COVID-19 is the propensity to develop multi-organ dysfunction, with acute kidney injury (AKI) occurring in over 50% of patients [2]. Furthermore, critically ill patients with kidney failure requiring renal replacement therapy have significantly higher hospital mortality rates than that of matched controls [3].
The significant morbidity and mortality associated with respiratory and kidney failure in the setting of COVID-19 has created complex medical decisions regarding the use of artificial life support, particularly ECMO and continuous renal replacement therapy (CRRT) or their combination. Therefore, we performed a retrospective analysis of patients with COVID-19 who required combined ECMO-CRRT and compared their outcomes to those of a wide cohort of non–COVID-19 patients requiring the same level of support. We hypothesized that patients with COVID-19 requiring combined ECMO-CRRT would have higher mortality than those without COVID-19.
MATERIALS AND METHODS
The retrospective study (protocol no. 1860629-1, ECMO and CRRT in patients with COVID-19, February 3, 2022) was approved by the Institutional Board Review at WellStar Kennestone Hospital, Marietta, GA, USA. The study was performed in accordance with the ethical standards of the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards. Informed consent was waived as it was determined that the proposed activity was exempt from further review by the Human Protections Administrator or the Institutional Review Board, based on 45 CFR 46.101(b) of the Code of Federal Regulations, Category 4: Research involving collection or study of existing identifiable private information or identifiable biospecimens, documents, records, or specimens if the sources are publicly available or if data are recorded in a way that subjects cannot be identified. Consecutive patients requiring combined ECMO-CRRT due to respiratory and kidney failure at WellStar Kennestone Hospital (Marietta, GA, USA) from June 2016 to February 2022 were studied. These patients were divided into those with severe respiratory failure due to COVID-19 and non–COVID-19 causes.
Indications for ECMO included but were not limited to severe ARDS and refractory hypoxemia (PaO2/FiO2 <80 mm of mercury [mm Hg]), or severe hypercapnic respiratory failure (pH <7.25 with a PaCO2 ≥60 mm Hg), after optimal conventional management (including, in the absence of contraindications, a trial of prone positioning).
ARDS, defined as an acute disorder that starts within seven days of the inciting event and characterized by bilateral lung infiltrates and severe progressive hypoxemia in the absence of any evidence of cardiogenic pulmonary edema, was classified as mild, moderate, or severe according to the following: mild: an initial PaO2/FiO2 of 201–300 mm Hg with invasive or non-invasive ventilation with a tight-fitting mask and positive end expiratory pressure or continuous positive airway pressure of at least 5 cm H2O; moderate: PaO2/FiO2 of 101–200 mm Hg; severe: PaO2/FiO2 of ≤100 mm Hg [4].
The diagnosis of COVID-19 was confirmed by a positive real-time fluorescent reverse transcription-polymerase chain reaction test for 2019-nCoV nucleic acid at the time of hospital admission. AKI was defined as an: (1) increase in serum creatinine by ≥0.3 mg/dl within 48 hours; or (2) increase in serum creatinine to ≥1.5 times baseline, which is known or presumed to have occurred within the prior 7 days; or (3) urine volume <0.5 ml/kg/hr over a 6-hour period. Chronic dialysis patients requiring ECMO-CRRT were also included in the study.
The indications for initiating CRRT included metabolic acidosis, hyperkalemia, uremia, and fluid overload. CRRT was administered during ECMO via a dialysis catheter, placement of a circuit in-line hemofilter, or a direct connection of CRRT in line with the ECMO circuit. Patients who required ECMO due to primary cardiac indications, those younger than 18 years, patients not requiring CRRT and those not requiring ECMO were excluded from this study.
Statistical Analyses
Patients’ demographic, clinical, radiological, and laboratory data were collected for analysis. Data are presented as mean and standard deviation if normally distributed and median (interquartile range) or range if not. For parametric data, differences in the mean were compared using the Student t-test or analysis of variance. The Wilcoxon-Mann-Whitney/Kruskal-Wallis test was used for highly skewed data. Normality of the data was assessed using a frequency distribution histogram. Differences in proportions were assessed by the chi-square or Fisher’s exact test. Cox proportional hazards models were used to study the associations and adjust for confounding factors. Variables with P≤0.05 in univariate analyses were included. Statistical significance was set at P<0.05. All analyses were performed using the JMP statistical software version 16.
RESULTS
All the patients with COVID-19 were seen between 2020 and 2022, on the other hand, non–COVID-19 patients were seen between the years 2016 and 2022. Most of the patients required venovenous (VV) ECMO support. Hybrid ECMO support was defined as venovenous-venous ECMO, venovenous-venous ECMO, venovenous-arterial ECMO, extracorporeal CO2 removal, VV/right ventricular assist device. Two hundred and fifty patients required ECMO during the study period. Of these, 127 also required CRRT. Patients with COVID-19 on ECMO had higher need for CRRT compared to those without COVID (58.8% [60/102] vs. 45.2% [67/148], P=0.002) (Figure 1).
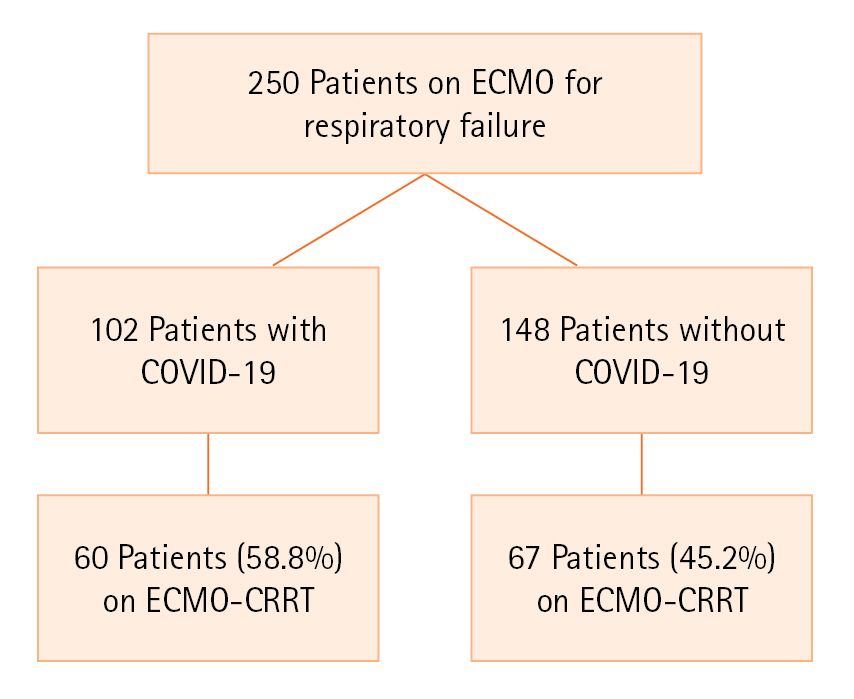
Flowchart of the study. ECMO: extracorporeal membrane oxygenation; COVID-19: coronavirus disease 2019; CRRT: continuous renal replacement therapy.
In the non–COVID-19 group, 54 patients (80.6%) were on VV ECMO, and 13 patients (19.4%) were on hybrid ECMO. In the COVID-19 group, 52 patients (86.7%) were on VV ECMO, and 8 patients (13.3%) were on hybrid ECMO (P=0.47). In the non–COVID-19 group requiring ECMO-CRRT, there were 10 cases of viral pneumonia, including influenza, parainfluenza, and human metapneumovirus (14.9%); 42 cases of bacterial pneumonia (62.8%); 8 cases of post-surgical ARDS (11.9%); 3 cases of aspiration pneumonia (4.47%); 3 cases of ARDS due to burns (4.47%); and 1 case of ARDS due to pancreatitis (1.49%). Patients in this group had more comorbidities and lower renal function at baseline.
Patients with COVID-19 on ECMO-CRRT had longer hospitalizations, longer duration of ECMO-CRRT support and increased hospital mortality. We observed no differences in the time of delivery of ECMO, CRRT or in the fluid balance. CRRT was started simultaneously with ECMO in 28 (46.7%) patients with COVID-19 and 30 non-COVID patients (44.7%); before ECMO in 8 patients with COVID-19 (13.3%) and 11 patients without COVID (16.4%); and after ECMO in 24 patients with COVID-19 (40%) vs 26 patients without COVID (38.8%) (P=0.94). There was a similar need for dialysis after hospital discharge (Table 1).
After adjusting for confounding variables, patients with COVID-19 on ECMO-CRRT showed significantly increased 30 day-mortality (Table 2). Models using 90-day mortality yielded a similar result (HR, 6.23; 95% confidence interval [CI], 2.60–14.9; P<0.001). Patients with COVID-19 who survived spent fewer days on ECMO and CRRT compared to non-survivors (ECMO: median, 8.96 days; interquartile range [IQR], 6–25.1 vs. median, 21.3 days; IQR, 10.9–38.3; P=0.008 and CRRT: median, 8 days; IQR, 5–15 vs. median, 17 days; IQR, 6–34, P=0.10). Non–COVID-19 patients who survived had a median of 7.71 days on ECMO (IQR, 5–13.7) versus 5.86 days in those who died (IQR, 2.25–13.9) (P=0.14). Median CRRT days were 8 (IQR, 3–15) and 6 (IQR, 3–11) for survivors and deceased, respectively (P=0.53).
DISCUSSION
There is a paucity of studies on the outcomes of combined ECMO-CRRT in patients with COVID-19. Given this, and the high mortality rate associated with combined respiratory and kidney failure we sought to describe the outcomes of patients with COVID-19 requiring combined ECMO-CRRT, in comparison to patients without COVID-19 requiring a similar level of support. COVID-19 can lead to progressive ARDS that requires mechanical ventilation. Despite best practices, some patients develop severe respiratory failure requiring ECMO therapy. This population requires the highest level of hospital care and is at significant risk of multiorgan failure and mortality. Reportedly, 45%–80% of critically ill patients with COVID-19 develop AKI, and 20%–39% require renal replacement therapy during their hospitalization [5,6]. Furthermore, those intubated with kidney failure requiring CRRT have mortality rates of nearly 70% [7].
COVID-19 inflicts renal damage via several possible mechanisms, including direct cytotoxic effects on the renal epithelium via angiotensin converting enzyme II or by a cytokine storm, which causes shock and multiorgan dysfunction with subsequent pre-renal and acute tubular injury. Histopathological examination of biopsies from patients with AKI associated with COVID-19 has shown mostly focal segmental glomerulosclerosis and acute tubular necrosis [8-10].
Prior studies showed that ECMO patients had rates of AKI requiring renal replacement therapy of 59%, with 3.73 higher odds of hospital mortality and a 1-year mortality rate of 32%, along with a higher risk of post-discharge chronic kidney disease (CKD). Other studies have shown an even more dismal prognosis, with a 3-month survival of 17% [11-14]. Risk factors include multiorgan failure, a history of congestive heart failure, hypertension, diabetes, CKD, shock, diffuse inflammation, viral cytotoxicity, ischemic events, and nephrotoxins. ECMO-related factors contributing to kidney failure include hemodynamic changes during ECMO cannulation, hemolysis, hyperinflammatory state, microcirculatory dysfunction, and coagulopathy [15-17].
The mortality rate of patients with COVID-19 on ECMO has fluctuated depending on the treatment era. In a recent meta-analysis, the pooled mortality rate of these patients was 48.8%. Mortality was higher among studies that enrolled patients later as opposed to those enrolling patients earlier in the pandemic. Predictors of increased mortality included age, time of final patient enrolment, proportion of patients receiving corticosteroids, and ECMO duration [18].
We found that patients with COVID-19 required longer durations of ECMO and CRRT, had longer hospitalizations and lower survival rates at hospital discharge, 30 days, and 90 days when compared to non–COVID-19 patients. Furthermore, after adjusting for confounding variables, patients with COVID-19 on ECMO-CRRT had five to six times higher risk of death than those without COVID-19.
However, a previous study of a similar population reported a 90-day mortality rate of only 29% [19]. The cause of this survival discrepancy requires better understanding. For instance, a study comparing patients with COVID-19 on ECMO to a matched group with influenza found that those with COVID-19 had a 1.34 times higher risk of mortality. Another study suggested that the higher mortality seen in patients with COVID-19 on ECMO compared with H1N1 patients on ECMO was driven mostly by their older age and length of hospitalization [20,21].
In this study, ECMO and CRRT were initiated in close temporal proximity to each other. The pressor requirements and time to ECMO and CRRT initiation were similar between the two groups. We noticed no difference in thrombotic events. Nonetheless, patients with COVID-19 remained longer on ECMO and CRRT, indicating a more prolonged state of multiorgan failure. Furthermore, patients with COVID-19 who died required more days of ECMO and CRRT than survivors. This finding aligns with prior studies that reported that a longer need for ECMO support and prolonged renal failure are associated with increased mortality in hospitalized patients [22,23].
Kidney failure is associated with increased mortality due to multiple mechanisms, including underlying multiorgan failure, accumulation of uremic toxins, fluid overload, acidosis, electrolyte abnormalities, increased inflammation, impaired immunity, worsening sepsis, and cardiovascular complications [24-26]. We found no difference in fluid balance between the two groups. A prior study showed that patients who achieved a lower fluid balance had higher odds of survival to discharge, along with improved PaO2/FiO2 [27]. Unfortunately, we could not compare inflammatory markers between the two groups, as non–COVID-19 patients did not have consistent C-reactive protein, interleukin-6, D-dimer, or ferritin measurements.
Another consequence of kidney failure is the development of CKD. Patients with COVID-19 who develop AKI KDIGO stages 2 and 3 and patients with unresolved AKI at the time of discharge have demonstrated higher odds of CKD post-hospitalization [28]. In this study, 25% of the hospital survivors who required CRRT who were not previously on dialysis remained dialysis-dependent after hospital discharge. A prior study showed that among non-ECMO critically ill patients with COVID-19 who developed AKI, 44.4% remained dialysis-dependent at discharge, 41% at 3 months, and 33% at 6 months [5].
Main limitations of the study include its relatively small number of patients, single-center enrollment (although patients were referred from several hospitals for ECMO evaluation), and retrospective data collection. Given the fact many of the patients were included before COVID-19 vaccines were available we were unable to study vaccine status and association with clinical outcomes. In conclusion, patients with COVID-19 who required ECMO-CRRT had increased mortality when compared to patients without COVID-19.
KEY MESSAGES
▪ Coronavirus disease 2019 (COVID-19) patients requiring extracorporeal membrane oxygenation (ECMO)-continuous renal replacement therapy (CRRT) support had longer hospitalizations than non–COVID-19 patients.
▪ Patients with COVID-19 remained longer on ECMO and CRRT than patients without COVID-19.
▪ In multivariate models, patients with COVID-19 on ECMO-CRRT had an increased risk of 30- and 90-day mortality compared to non–COVID-19 patients.
Notes
CONFLICT OF INTEREST
No potential conflict of interest relevant to this article was reported.
FUNDING
None.
AUTHOR CONTRIBUTIONS
Conceptualization: CFP, RH, AB, AS. Methodology: CFP, CT, AS. Formal analysis: CFP. Data curation: CFP, RH, CT, JJ. Visualization: CFP, AS. Project administration: CFP, RH. Writing–original draft: CFP, RH, CT, JJ, AB, AS. Writing–review & editing: all authors.
Acknowledgements
we would like to thank the ICU staff (nurses, pharmacists, social workers, ECMO nurses, respiratory therapists, physical therapists, clerical staff) at the different WellStar Hospitals who worked tirelessly taking care of our patients.