The correlation between carotid artery Doppler and stroke volume during central blood volume loss and resuscitation
Article information
Abstract
Background
Using peripheral arteries to infer central hemodynamics is common among hemodynamic monitors. Doppler ultrasound of the common carotid artery has been used in this manner with conflicting results. We investigated the relationship between changing common carotid artery Doppler measures and stroke volume (SV), hypothesizing that more consecutively-averaged cardiac cycles would improve SV-carotid Doppler correlation.
Methods
Twenty-seven healthy volunteers were recruited and studied in a physiology laboratory. Carotid artery Doppler pulse was measured with a wearable, wireless ultrasound during central hypovolemia and resuscitation induced by a stepped lower body negative pressure protocol. The change in maximum velocity time integral (VTI) and corrected flow time of the carotid artery (ccFT) were compared with changing SV using repeated measures correlation.
Results
In total, 73,431 cardiac cycles were compared across 27 subjects. There was a strong linear correlation between changing SV and carotid Doppler measures during simulated hemorrhage (repeated-measures linear correlation [Rrm]=0.91 for VTI; 0.88 for ccFT). This relationship improved with larger numbers of consecutively-averaged cardiac cycles. For ccFT, beyond four consecutively-averaged cardiac cycles the correlation coefficient remained strong (i.e., Rrm of at least 0.80). For VTI, the correlation coefficient with SV was strong for any number of averaged cardiac cycles. For both ccFT and VTI, Rrm remained stable around 25 consecutively-averaged cardiac cycles.
Conclusions
There was a strong linear correlation between changing SV and carotid Doppler measures during central blood volume loss. The strength of this relationship was dependent upon the number of consecutively-averaged cardiac cycles.
INTRODUCTION
Using a peripheral artery to transduce central hemodynamics is common among hemodynamic monitors. For instance, both calibrated and uncalibrated pulse contour analyses use the radial artery pressure waveform to infer volumetric flow from the left ventricle [1]. With the rise of point-of-care ultrasound use in acute care, a similar approach has been adopted with ultrasonographic measures. As an example, the common carotid artery Doppler pulse is used to assess both absolute [2,3] and changing [4-11] stroke volume (SV). With respect to the critically ill population, this approach is important during resuscitation as flow-guided preload administration reduces morbidity and mortality [12,13]; furthermore, detecting diminished SV could help diagnose early hemorrhage [14]. Nevertheless, there are conflicting results regarding carotid Doppler as an estimate of cardiac output and its change [15-17]. Various groups [18,19] have noted the importance of human measurement variability and cardiac cycle sample size as key mediators relating left ventricular output to the common carotid artery. Given this, Kenny et al. [4,18,20] previously reported on a wearable, wireless, adhesive Doppler ultrasound during central blood volume loss and resuscitation in healthy volunteers undergoing lower body negative pressure (LBNP) and release. Within this paradigm, this group showed that SV change across many cardiac cycles can be transduced by the carotid artery Doppler spectrogram. Therefore, we hypothesized that the correlation coefficient between changing SV and carotid Doppler measures such as the velocity time integral (VTI) and corrected flow time (ccFT) would depend on the number of consecutively-averaged cardiac cycles used to calculate change. Furthermore, we hypothesized that the relationship between the number of consecutively-averaged cardiac cycles and the correlation coefficient relating SV to carotid Doppler would be different between periods of falling SV (i.e., LBNP) and rising SV (i.e., LBNP release).
MATERIALS AND METHODS
Ethics Approval and Consent to Participate
All data collection was initially performed after approval of the local Institutional Ethics Committee (No. 19-010136). Written informed consent was obtained from all participants included in the analysis at the time of enrollment. Adults with no prior medical history and taking no cardiovascular medications were recruited.
SV Monitoring
A non-invasive SV monitor, the Nexfin (Edwards Lifesciences), was used throughout the study and synchronized with the carotid Doppler monitor for each cardiac cycle. Alignment of the Doppler spectrograms to the non-invasive SV monitor was performed by cross-correlating heart rate (HR) to find the optimal lag in the Doppler signal. Systolic, diastolic and mean arterial pressure were also obtained from the Nexfin.
Doppler System
The U.S. Food and Drug Administration-cleared, wireless, wearable, four MHz Doppler ultrasound patch (Flosonics Medical) was placed and the ccFT and VTI captured, as described previously [4,18,20].
Lower Body Negative Pressure
Beginning with resting baseline, each stage of the LBNP protocol required five minutes. LBNP was reduced by 15 mm Hg per stage down to and including –60 mm Hg and then by 10 mm Hg down to and including –80 mm Hg if this was tolerated by the patient. The final stage was release of LBNP to atmospheric pressure. All subjects underwent this seven-stage protocol in duplicate.
Statistical Analysis
Cardiac cycles with artifact or during LBNP stage transition were excluded. Artifact was detected by HR discordance of < 0.8 or > 1.3-fold change between devices and visual inspection of the Doppler spectrograms for phonation or deglutition signatures. Both ccFT and VTI were referenced to resting baseline to model increasingly severe hypovolemia. Change from the lowest-achieved LBNP stage back to atmosphere modeled rapid blood transfusion.
To account for multiple measurements per person, repeated-measures linear correlation (Rrm) was performed across negative pressure stages. Spearman correlation was used to model lowest pressure stage back to release when using whole stage averages. To detect a Rrm of 0.8, with a power of 0.8 and significance level of 0.05, we calculated the minimum sample size for a multilevel model with seven stages. The minimum required sample size for each stage was approximately 11.
RESULTS
The baseline characteristics and vital signs of all included subjects are listed in Table 1. Healthy volunteers were recruited between July 2020 and December 2021 for this physiology laboratory study. In total, data from 27 subjects were included in this analysis. The wearable ultrasound patch, its graphical user interface and representative results from a single session are shown in Figure 1. Across all sessions and subjects, a total of 91,975 cardiac cycles were captured; but 20.16% of the beats were excluded for these reasons: 8.34% stage transition, 1.86% misaligned HR between the Doppler and traditional HR vital sign monitor (i.e., < 0.8 or > 1.3-fold change as compared to vital sign monitor), 6.86% Doppler feature detection (e.g., dicrotic notch) failure as determined by either ccFT or VTI value more than two standard deviations as compared to the respective mean values for any given LBNP stage, and 3.10% Nexfin feature detection failure as determined by SV value more than two standard deviations as compared to the mean value of any given LBNP stage. Stage transition was excluded to ensure hemodynamic stability during comparison between SV and carotid Doppler measures. A total of 73,431 cardiac cycles comprised the analysis and included 8,655 release-stage beats.
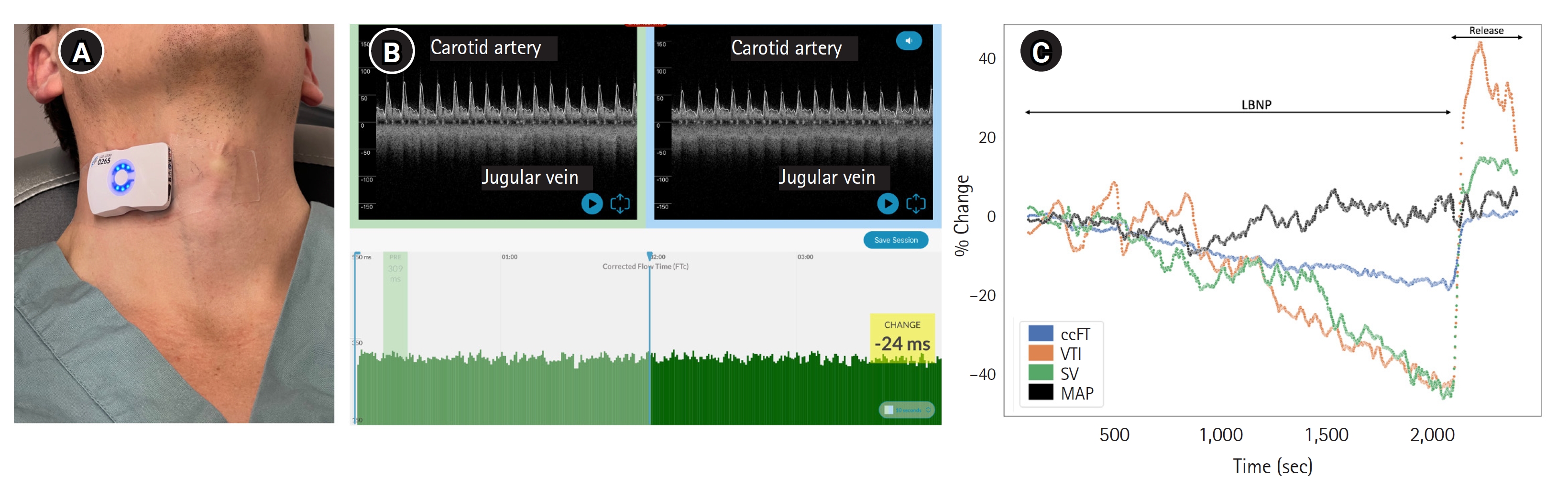
Wearable Doppler ultrasound and representative results. (A) The wearable Doppler ultrasound worn over the right common carotid artery of a healthy subject. (B) Representative Doppler spectrograms from left to right showing increasingly severe lower body negative pressure (LBNP). The green bars at the base are the common carotid corrected flow time (ccFT) per cardiac cycle and time on the X-axis. (C) Representative results from a single LBNP protocol showing two carotid ultrasound measures: ccFT and velocity time integral (VTI) compared to stroke volume (SV) and mean arterial pressure (MAP). Release was when the LBNP chamber returned to atmospheric pressure.
The correlations between changing ccFT and VTI against SV are shown in Figure 2. In Figure 2, the first row (Figure 2A-C) is % change ccFT, the second row (Figure 2D-F) is % change in carotid VTI. The first two panels for each row illustrate the relationship (i.e., repeated measures correlation) between % change SV on the X-axis and carotid Doppler change on the Y-axis during LBNP. The final panel in each row demonstrates the relationship between the number of consecutively-averaged cardiac cycles on the X-axis and strength of the repeated measures correlation coefficient on the Y-axis.
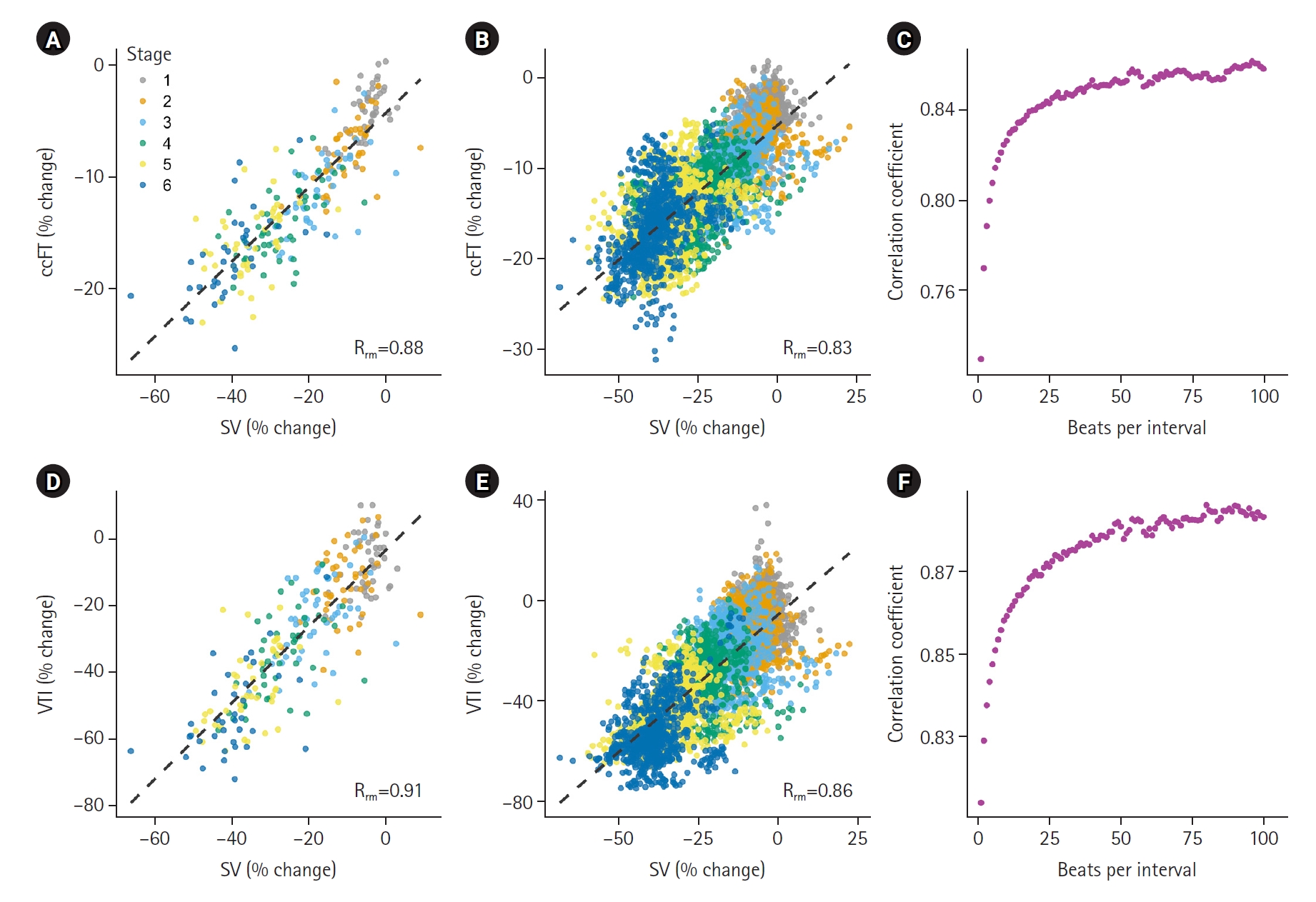
The relationship between change in common carotid artery corrected flow time (ccFT) and velocity time integral (VTI) with stroke volume (SV) in a model of central blood volume loss. (A) The relationship between SV % change from baseline and ccFT % change from baseline using whole stage averages. Across decreasing pressure stages, the repeated-measures linear correlation (Rrm) is 0.88 and the slope is 0.33. (B) The relationship between SV % change from baseline and ccFT % change from baseline using 10-beat averages. Across decreasing pressure stages, the Rrm is 0.83 and the slope is 0.30. (C) The effect of the number of consecutively-averaged cardiac cycles on repeated-measures correlation coefficient between % change SV and % change ccFT. (D) The relationship between SV % change from baseline and VTI % change from baseline using whole stage averages. Across decreasing pressure stages, the Rrm is 0.91 and the slope is 1.18. (E) The relationship between SV % change from baseline and VTI % change from baseline using 10-beat averages. Across decreasing pressure stages, the Rrm is 0.86 and the slope is 1.16. (F) The effect of the number of consecutively-averaged cardiac cycles on repeated-measures correlation coefficient between % change SV and % change VTI.
DISCUSSION
Our findings are important for the acute and critically ill populations. During progressive central blood volume loss, there was a strong, linear correlation between falling SV, ccFT and carotid VTI. Moreover, the correlation between changing ccFT and VTI with SV improved with increasing number of consecutively-averaged cardiac cycles in both the models of volume loss and resuscitation. Both ccFT and VTI also increased during central blood volume resuscitation, however the strength of their direct correlations with SV was less strong.
The finding that both VTI and ccFT correlated with falling SV during LBNP is consistent with previous work [4] and adds credence to measures such as the Doppler shock index, i.e., HR divided by ccFT or carotid artery VTI [14]. The slopes of the relationships found in this investigation imply that a 10% fall in SV correlates with a 11.8% fall in VTI and a 3.3% or 10.5 millisecond (ms) fall in ccFT. These values are consistent with previously-reported regression slopes between changing SV and carotid Doppler measures [4, 21]. Given that during exsanguination, the fraction of blood volume loss can be estimated as one-half of the % fall in SV, our measured VTI and ccFT values could be used as crude estimates of total blood volume loss. For example, 15% total blood volume loss is expected to decrease VTI by 35.4% and ccFT by 9.9% or 31.5 ms when hemorrhage is the sole etiology of reduced SV. Therefore, trending carotid artery Doppler measures in the acute trauma population could predict impending hemodynamic collapse due to blood loss prior to more traditional vital signs such as mean arterial pressure and HR [4,14,22].
Second, we observed stronger correlation between changing SV and carotid Doppler measures with greater numbers of consecutively-averaged cardiac cycles. While this is unsurprising, we note that for ccFT the relationship did not improve (i.e., above 0.8) until at least five cardiac cycles were averaged. In contrast, the relationship with VTI was strong with fewer cardiac cycles. We suspect that this is because of the precision required to infer SV change from the ccFT. For example, Barjaktarevic et al. [6] found that a +7 ms ccFT augmentation most accurately detected a +10% change in SV. In their cohort, the baseline ccFT was 301 ms; therefore, +7 ms approximates a +2% change in ccFT. These values are consistent with our reported findings; a 10% change in SV correlated with a 3% or 10.5 ms change in ccFT. Our observations have important implications for investigations relating cardiac output to the ccFT; averaging too few cardiac cycles will degrade the relationship [15]. The theoretical benefit of ccFT over carotid VTI is that ccFT is, ostensibly, more resistant to changes in carotid artery diameter, fraction of flow directed towards the head and error introduced by insonation angle selection [3,23,24].
Last, during the resuscitation phase of the LBNP model, the relationship between changing SV and carotid Doppler measures was less strong than during blood volume loss. We suspect that this is due to the relatively large change in SV between the final LBNP stage and recovery. Therefore, a more gradual rise in SV may have shown a stronger correlation. Additionally, the slope of the changing VTI to SV relationship was steeper during the release phase for VTI. The reason for this is not clear; however, increased carotid artery flow fraction at LBNP release could be the reason for this [3]. More specifically, a combination of increased peripheral resistance and downstream cerebrovascular dilation would preferentially direct flow to the carotid artery at the moment of LBNP release [3,20]; therefore, the rise in carotid VTI per unit SV augmentation would be greater.
There are limitations to our analysis. First, given that this was a convenience sample of healthy volunteers, these data may be difficult to generalize to patients in the hospital. Nevertheless, our observations are similar to a previous LBNP investigation [4], and carotid Doppler has been used to infer changes in SV in the critically-ill [6,25]. Second, the volunteers were cooperative and able to remain still for the entirety of the protocol; movement and vocalization by patients may make implementing this paradigm in the clinical sphere more challenging. Third, the release phase of our model was a relatively large preload bolus (i.e., immediately replenishing an approximate 25% central blood volume deficit [14]. A gradual increase in preload and, therefore, SV would clarify the relationship between carotid Doppler and central hemodynamics during the release phase.
In conclusion, this study comprised the largest known dataset comparing changing SV with the common carotid Doppler pulse. This investigation included 27 healthy subjects and 73,431 cardiac cycles, while most clinical studies sample fewer than three cardiac cycles per patient. There was a strong linear correlation between changing SV and ccFT as well as changing SV and VTI during central blood volume loss. Furthermore, the strength of this relationship was dependent upon the number of consecutively-averaged cardiac cycles. These results partly explain discordant outcomes when relating the carotid artery Doppler pulse to central hemodynamics.
HIGHLIGHTS
▪ Inferring central hemodynamics from a peripheral artery is a common approach amongst hemodynamic monitors; however, many cardiac cycles are typically averaged with commercially-available devices.
▪ When changing carotid Doppler is compared to changing stroke volume measured by non-invasive pulse contour analysis, the correlation is strengthened when more cardiac cycles are averaged consecutively.
▪ There was a strong linear correlation between falling stroke volume and carotid artery Doppler pulse with progressively-severe central hypovolemia.
▪ This suggests monitoring carotid Doppler could help detect cryptic blood loss.
Notes
CONFLICT OF INTEREST
IK, SA, ME, AME, JKE, JESK are employees of Flosonics Medical. No other potential conflicts of interest relevant to this article were reported.
FUNDING
None.
AUTHOR CONTRIBUTIONS
Conceptualization: ME, AE, JKE, JLT, CHK, BDJ, JESK. Methodology: ME, AE, JKE, JLT, CHK, BDJ, JESK. Formal analysis: IK, SA. Data curation: IK, JLT. Visualization: IK, SA. Project administration: ME, AE, JKE, JESK. Funding acquisition: AE, JKE, JESK. Writing–original draft: IK, JESK. Writing–review & editing: all authors. All authors read and agreed to the published version of the manuscript.
ACKNOWLEDGMENTS
None.

