Articles
- Page Path
- HOME > Acute Crit Care > Volume 33(3); 2018 > Article
- Original Article Feasibility of Immediate in-Intensive Care Unit Pulmonary Rehabilitation after Lung Transplantation: A Single Center Experience
-
Joo Han Song1, Ji-Eun Park1
 , Sang Chul Lee2
, Sang Chul Lee2 , Sarang Kim2, Dong Hyung Lee3, Eun Kyoung Kim4, Song Yee Kim1
, Sarang Kim2, Dong Hyung Lee3, Eun Kyoung Kim4, Song Yee Kim1 , Ji Cheol Shin2, Jin Gu Lee5, Hyo Chae Paik5, Moo Suk Park1
, Ji Cheol Shin2, Jin Gu Lee5, Hyo Chae Paik5, Moo Suk Park1
-
Acute and Critical Care 2018;33(3):146-153.
DOI: https://doi.org/10.4266/acc.2018.00129
Published online: August 31, 2018
1Division of Pulmonology, Department of Internal Medicine, Institute of Chest Diseases, Severance Hospital, Yonsei University College of Medicine, Seoul, Korea
2Department and Research Institute of Rehabilitation Medicine, Severance Rehabilitation Hospital, Yonsei University College of Medicine, Seoul, Korea
3Division of Nursing, Pediatric Intensive Care Unit, Severance Hospital, Seoul, Korea
4Division of Nursing, Neurointensive Care Unit, Severance Hospital, Seoul, Korea
5Department of Thoracic and Cardiovascular Surgery, Yonsei University College of Medicine, Seoul, Korea
- Corresponding author Moo Suk Park Division of Pulmonology, Department of Internal Medicine, Institute of Chest Diseases, Severance Hospital, Yonsei University College of Medicine, 50-1 Yonsei-ro, Seodaemun-gu, Seoul 03722, Korea Tel: +82-2-2228-1930 Fax: +82-2-393-6884 E-mail: pms70@yuhs.ac
Copyright © 2018 The Korean Society of Critical Care Medicine
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
-
Background
- Physical function may influence perioperative outcomes of lung transplantation. We investigated the feasibility of a pulmonary rehabilitation program initiated in the immediate postoperative period at an intensive care unit (ICU) for patients who underwent lung transplantation.
-
Methods
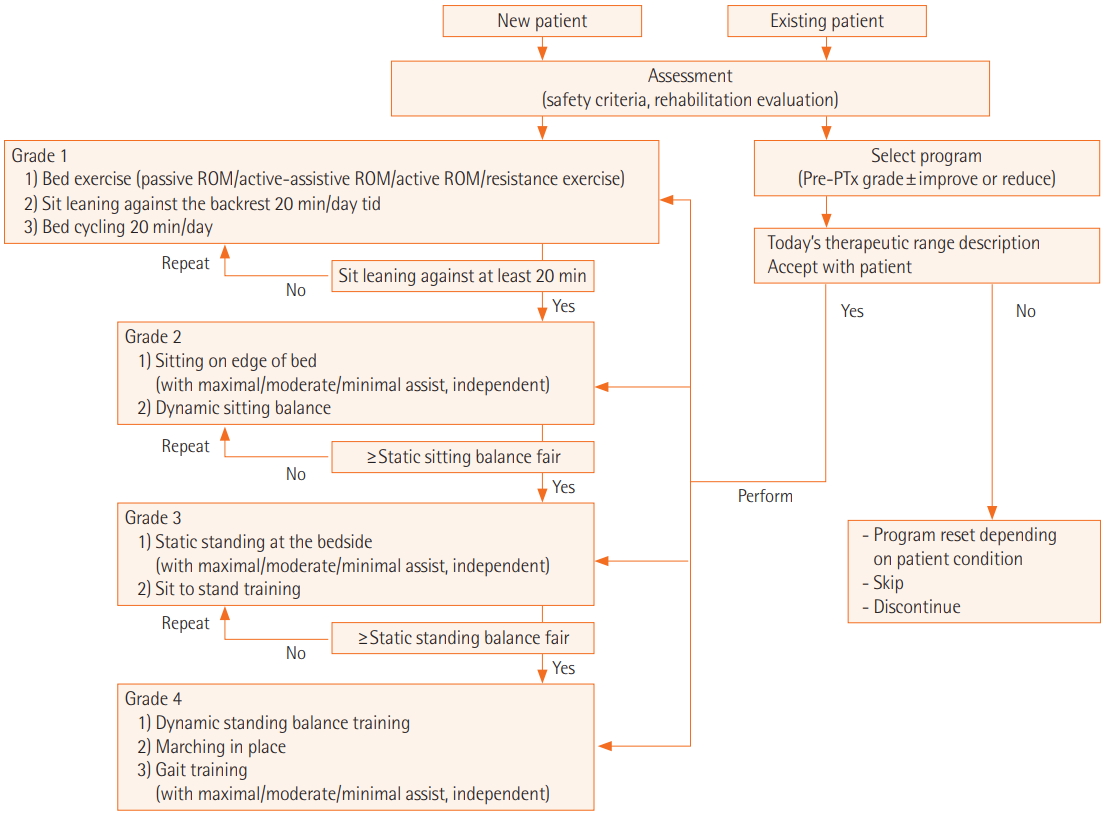
- We retrospectively evaluated 22 patients who received pulmonary rehabilitation initiated in the ICU within 2 weeks after lung transplantation at our institution from March 2015 to February 2016. Levels of physical function were graded at the start of pulmonary rehabilitation and then weekly throughout rehabilitation according to criteria from our institutional pulmonary rehabilitation program: grade 1, bedside (G1); grade 2, dangling (G2); grade 3, standing (G3); and grade IV, gait (G4).
-
Results
- The median age of patients was 53 years (range, 25 to 73 years). Fourteen patients (64%) were males. The initial level of physical function was G1 in nine patients, G2 in seven patients, G3 in four patients, and G4 in two patients. Patients started pulmonary rehabilitation at a median of 7.5 days (range, 1 to 29 days) after lung transplantation. We did not observe any rehabilitation-related complications during follow-up. The final level of physical function was G1 in six patients, G3 in two patients, and G4 in 14 patients. Fourteen of the 22 patients were able to walk with or without assistance, and 13 of them maintained G4 until discharge; the eight remaining patients never achieved G4.
-
Conclusions
- Our results suggest the feasibility of early pulmonary rehabilitation initiated in the ICU within a few days after lung transplantation.
INTRODUCTION
MATERIALS AND METHODS
RESULTS
DISCUSSION
KEY MESSAGES

| Characteristics | Value |
|---|---|
| Age (yr) | 52.6±12.4 (25-73) |
| Male sex | 14 (64) |
| Body mass index (kg/m2) | 19.9 (16.25-23.4) |
| Smoker | 13 (59) |
| Underlying lung disease | |
| IPF | 12 (54) |
| COPD | 1 (5) |
| PPAH | 1 (5) |
| AIP | 2 (9) |
| Bronchiectasis | 2 (9) |
| Post-SCT BOS | 3 (13) |
| CVD-ILD | 1 (5) |
| mMRC grade | |
| II or III/IV | 9 (41)/13 (59) |
| NYHA grade | |
| II or III/IV | 7 (32)/15 (68) |
| Comorbidity | 10 (45) |
| Osteopenia/osteoporosis | 7 (32)/5 (23) |
| 25-Hydroxy vitamin D (ng/ml) | 9.0 (6.1-19.0) |
| Pulmonary function | |
| FVC (l) | 1.5 (1.1-2.4) |
| FEV1 (l) | 1.1 (0.8-1.9) |
| FEV1 (% predicted) | 43 (30-62) |
| Cardiac function | |
| LVEF (%) | 64 (58-68) |
| Emergency statusa | |
| Status 0 | 10 (45.5) |
| Status 1 | 11 (50) |
| Status 2 | 1 (4.5) |
| Time on waiting list (day) | 35 (10-168) |
| Donor age (yr) | 42.2±12.9 (16-59) |
| Donor sex, male | 15 (68) |
| Donor body mass index (kg/m2) | 22.8 (15.8-32) |
| Donor PaO2/FiO2 | 428 (195-669) |
| Preoperative status | |
| Tracheostomy | 6 (27) |
| Hospitalization | 3 (14) |
| ICU care | 11 (50) |
| Mechanical ventilator | 11 (50) |
| ECMO support | 7 (32) |
Values are presented as mean±standard deviation (range), number (%), or median (interquartile range).
IPF: idiopathic pulmonary fibrosis; COPD: chronic obstructive pulmonary disease; PPAH: primary pulmonary hypertension; AIP: acute interstitial pneumonia; SCT: stem cell transplantation; BOS: bronchiolitis obliterans syndrome; CVD-ILD: collagen vascular disease related interstitial lung disease; mMRC: modified Medical Research Council; NYHA: New York Heart Association; FVC: forced vital capacity; FEV1: forced expiratory volume in 1 second; LVEF: left ventricular ejection fraction; PaO2: arterial partial pressure of oxygen; FiO2: fraction of inspired oxygen; ICU: intensive care unit; ECMO: extracorporeal membrane oxygenation.
a Lungs are allocated by the emergency status of the recipient (Korean lung allocation score).
| Patient no. | Age (yr) | Sex | Diagnosis | Start of PR | Days of PR | Initial functional grade | Best functional grade | Final functional grade | O2 support at start of PR | O2 support at finish of PR | Status, survival months | Outcome (cause of death) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 54 | F | CVD-ILD | POD 14 | 10 | 4-2 | 4-3 | 4-3 | Nasal | Nasal | Alive, 14.8 | - |
| 2 | 63 | M | IPF | POD 7 | 48 | 2-1 | 4-2 | 4-2 | HFNC | Nasal | Alive, 16.0 | - |
| 3 | 46 | F | Bronchiectasis | POD 11 | 27 | 3-2 | 4-3 | 4-3 | Ventilator | Room air | Alive, 15.7 | - |
| 4a | 52 | M | IPF | POD 7 | 13 | 2-1 | 1-1 | 1-1 | T-mask | Ventilator | Died, 1.4 | Sudden cardiac arrest |
| 5 | 49 | F | AIP | POD 1 | 59 | 1-1 | 1-1 | 1-1 | Ventilator | Ventilator | Died, 2.9 | Bleeding |
| 6 | 32 | F | Post-SCT BOS | POD 8 | 217 | 3-1 | 4-2 | 4-2 | Ventilator | Room air | Alive, 14.6 | - |
| 7 | 62 | F | Bronchiectasis | POD 6 | 103 | 1-2 | 1-1 | 1-1 | Ventilator | Ventilator | Died, 4.0 | Pneumonia (Pseudomonas aeruginosa) |
| 8 | 26 | M | Post-SCT BOS | POD 6 | 85 | 1-2 | 3-2 | 3-2 | Nasal | Room air | Died, 12.7 | Sepsis (Acinetobacter baumannii) |
| 9 | 58 | F | IPF | POD 12 | 5 | 3-3 | 4-2 | 4-2 | Ventilator | Nasal | Died, 5.4 | Thrombotic thrombocytopenic purpura |
| 10 | 73 | M | IPF | POD 7 | 26 | 2-1 | 4-3 | 4-3 | Nasal | Room air | Died, 10.3 | Pneumonia (A. baumannii) |
| 11 | 58 | M | IPF | POD 1 | 14 | 1-1 | 4-2 | 4-2 | Nasal | Nasal | Died, 6.5 | Pneumonia (pathogen unknown) |
| 12 | 64 | M | IPF | POD 6 | 42 | 1-2 | 4-3 | 4-3 | Nasal | Room air | Alive, 13.4 | - |
| 13 | 60 | M | IPF | POD 6 | 59 | 1-2 | 3-1 | 3-1 | Ventilator | Ventilator | Died, 4.9 | Sepsis (A. baumannii) |
| 14 | 61 | M | COPD | POD 7 | 10 | 2-1 | 4-1 | 4-1 | Nasal | Nasal | Died, 5.6 | Pneumonia (A. baumannii) |
| 15 | 61 | M | IPF | POD 7 | 5 | 2-1 | 1-1 | 1-1 | Ventilator | Ventilator | Died, 1.5 | Sepsis (pathogen unknown) |
| 16 | 46 | M | AIP | POD 6 | 8 | 1-2 | 1-1 | 1-1 | Ventilator | Ventilator | Died, 2.6 | Sepsis (pathogen unknown) |
| 17 | 55 | F | IPF | POD 13 | 7 | 4-1 | 4-3 | 4-3 | Nasal | Nasal | Alive, 10.6 | - |
| 18 | 53 | M | IPF | POD 1 | 27 | 1-1 | 4-3 | 4-3 | Ventilator | Room air | Alive, 10.1 | - |
| 19 | 41 | M | Post-SCT BOS | POD 7 | 75 | 2-1 | 4-3 | 4-2 | Ventilator | Nasal | Alive, 9.4 | - |
| 20 | 25 | F | PPHN | POD 6 | 43 | 1-2 | 1-2 | 1-2 | Ventilator | Ventilator | Died, 2.3 | Sepsis (A. baumannii) |
| 21 | 60 | M | IPF | POD 7 | 42 | 2-1 | 4-3 | 4-3 | Nasal | Room air | Died, 5.5 | Sepsis |
| 22 | 58 | M | IPF | POD 8 | 10 | 3-1 | 4-3 | 4-3 | Nasal | Nasal | Alive, 7.0 | - |
PR: pulmonary rehabilitation; CVD-ILD: collagen vascular disease related interstitial lung disease; POD: postoperative day; IPF: idiopathic pulmonary fibrosis; HFNC: high flow nasal cannula; AIP: acute interstitial pneumonia; SCT: stem cell transplantation; BOS: bronchiolitis obliterans syndrome; COPD: chronic obstructive pulmonary disease; PPHN: primary pulmonary hypertension.
a Patient#4 had received liver transplantation simultaneously.
- 1. Myles PS, Snell GI, Westall GP. Lung transplantation. Curr Opin Anaesthesiol 2007;20:21-6.ArticlePubMed
- 2. Lands LC, Smountas AA, Mesiano G, Brosseau L, Shennib H, Charbonneau M, et al. Maximal exercise capacity and peripheral skeletal muscle function following lung transplantation. J Heart Lung Transplant 1999;18:113-20.ArticlePubMed
- 3. Reinsma GD, ten Hacken NH, Grevink RG, van der Bij W, Koëter GH, van Weert E. Limiting factors of exercise performance 1 year after lung transplantation. J Heart Lung Transplant 2006;25:1310-6.ArticlePubMed
- 4. Finlen Copeland CA, Vock DM, Pieper K, Mark DB, Palmer SM. Impact of lung transplantation on recipient quality of life: a serial, prospective, multicenter analysis through the first posttransplant year. Chest 2013;143:744-50.ArticlePubMed
- 5. Yusen RD, Christie JD, Edwards LB, Kucheryavaya AY, Benden C, Dipchand AI, et al. The Registry of the International Society for Heart and Lung Transplantation: thirtieth adult lung and heart-lung transplant report--2013; focus theme: age. J Heart Lung Transplant 2013;32:965-78.ArticlePubMed
- 6. Strueber M. Bridges to lung transplantation. Curr Opin Organ Transplant 2011;16:458-61.ArticlePubMed
- 7. Javidfar J, Brodie D, Iribarne A, Jurado J, Lavelle M, Brenner K, et al. Extracorporeal membrane oxygenation as a bridge to lung transplantation and recovery. J Thorac Cardiovasc Surg 2012;144:716-21.ArticlePubMed
- 8. Tomaszek SC, Fibla JJ, Dierkhising RA, Scott JP, Shen KR, Wigle DA, et al. Outcome of lung transplantation in elderly recipients. Eur J Cardiothorac Surg 2011;39:726-31.ArticlePubMedPDF
- 9. Nici L, Donner C, Wouters E, Zuwallack R, Ambrosino N, Bourbeau J, et al. American Thoracic Society/European Respiratory Society statement on pulmonary rehabilitation. Am J Respir Crit Care Med 2006;173:1390-413.ArticlePubMed
- 10. Wickerson L, Rozenberg D, Janaudis-Ferreira T, Deliva R, Lo V, Beauchamp G, et al. Physical rehabilitation for lung transplant candidates and recipients: an evidence-informed clinical approach. World J Transplant 2016;6:517-31.ArticlePubMedPMC
- 11. Maury G, Langer D, Verleden G, Dupont L, Gosselink R, Decramer M, et al. Skeletal muscle force and functional exercise tolerance before and after lung transplantation: a cohort study. Am J Transplant 2008;8:1275-81.ArticlePubMed
- 12. Munro PE, Holland AE, Bailey M, Button BM, Snell GI. Pulmonary rehabilitation following lung transplantation. Transplant Proc 2009;41:292-5.ArticlePubMed
- 13. Adler J, Malone D. Early mobilization in the intensive care unit: a systematic review. Cardiopulm Phys Ther J 2012;23:5-13.Article
- 14. Kayambu G, Boots R, Paratz J. Physical therapy for the critically ill in the ICU: a systematic review and meta-analysis. Crit Care Med 2013;41:1543-54.ArticlePubMed
- 15. Dowdy DW, Eid MP, Dennison CR, Mendez-Tellez PA, Herridge MS, Guallar E, et al. Quality of life after acute respiratory distress syndrome: a meta-analysis. Intensive Care Med 2006;32:1115-24.ArticlePubMed
- 16. Yu WS, Paik HC, Haam SJ, Lee CY, Nam KS, Jung HS, et al. Transition to routine use of venoarterial extracorporeal oxygenation during lung transplantation could improve early outcomes. J Thorac Dis 2016;8:1712-20.ArticlePubMedPMC
- 17. Jeong YH, Choi S, Park SI, Kim DK, Asan Medical Center Lung Transplantation Team. Clinical outcomes of lung transplantation: experience at Asan Medical Center. Korean J Thorac Cardiovasc Surg 2018;51:22-8.ArticlePubMedPMCPDF
- 18. Dierich M, Tecklenburg A, Fuehner T, Tegtbur U, Welte T, Haverich A, et al. The influence of clinical course after lung transplantation on rehabilitation success. Transpl Int 2013;26:322-30.ArticlePubMed
- 19. Walsh JR, Chambers DC, Davis RJ, Morris NR, Seale HE, Yerkovich ST, et al. Impaired exercise capacity after lung transplantation is related to delayed recovery of muscle strength. Clin Transplant 2013;27:E504-11.ArticlePubMed
- 20. Walsh JR, Chambers DC, Yerkovich ST, Hopkins PM, Morris NR. Low levels of physical activity predict worse survival to lung transplantation and poor early post-operative outcomes. J Heart Lung Transplant 2016;35:1041-3.ArticlePubMed
- 21. Calvo-Ayala E, Khan BA, Farber MO, Ely EW, Boustani MA. Interventions to improve the physical function of ICU survivors: a systematic review. Chest 2013;144:1469-80.ArticlePubMedPMC
- 22. Castro-Avila AC, Serón P, Fan E, Gaete M, Mickan S. Effect of early rehabilitation during intensive care unit stay on functional status: systematic review and meta-analysis. PLoS One 2015;10:e0130722.ArticlePubMedPMC
- 23. Lee CM, Fan E. ICU-acquired weakness: what is preventing its rehabilitation in critically ill patients? BMC Med 2012;10:115. ArticlePubMedPMCPDF
- 24. Nordon-Craft A, Moss M, Quan D, Schenkman M. Intensive care unit-acquired weakness: implications for physical therapist management. Phys Ther 2012;92:1494-506.ArticlePubMedPMCPDF
- 25. Wieske L, Dettling-Ihnenfeldt DS, Verhamme C, Nollet F, van Schaik IN, Schultz MJ, et al. Impact of ICU-acquired weakness on post-ICU physical functioning: a follow-up study. Crit Care 2015;19:196. ArticlePubMedPMC
- 26. Rozenberg D, Wickerson L, Singer LG, Mathur S. Sarcopenia in lung transplantation: a systematic review. J Heart Lung Transplant 2014;33:1203-12.ArticlePubMed
- 27. Fuller LM, Button B, Tarrant B, Steward R, Bennett L, Snell G, et al. Longer versus shorter duration of supervised rehabilitation after lung transplantation: a randomized trial. Arch Phys Med Rehabil 2017;98:220. -6. e3.ArticlePubMed
- 28. Langer D. Rehabilitation in patients before and after lung transplantation. Respiration 2015;89:353-62.ArticlePubMed
References
Figure & Data
References
Citations

- Post-operative, inpatient rehabilitation after lung transplant evaluation (PIRATE): A feasibility randomized controlled trial
Benjamin J Tarrant, Elizabeth Quinn, Rebecca Robinson, Megan Poulsen, Louise Fuller, Greg Snell, Bruce R Thompson, Brenda M Button, Anne E Holland
Physiotherapy Theory and Practice.2023; 39(7): 1406. CrossRef - Early Gait Function After Lung Transplantation in Patients With and Without Pretransplant Extracorporeal Membrane Oxygenation Support
Junghwa Do, Hyojin Lim, Kyung Cheon Seo, Suyoung Park, HyeRin Joo, Junghoon Lee, Eunjae Ko, Jaehwal Lim, Ho Cheol Kim, Dongkyu Oh, Sang-Bum Hong, Won Kim
Transplantation Proceedings.2023; 55(3): 616. CrossRef

 KSCCM
KSCCM
 PubReader
PubReader ePub Link
ePub Link Cite
Cite



