Articles
- Page Path
- HOME > Acute Crit Care > Volume 35(3); 2020 > Article
-
Original Article
Surgery Association between postoperative fluid balance and mortality and morbidity in critically ill patients with complicated intra-abdominal infections: a retrospective study -
Joohyun Sim1
 , Jae Young Kwak2, Yun Tae Jung2
, Jae Young Kwak2, Yun Tae Jung2
-
Acute and Critical Care 2020;35(3):189-196.
DOI: https://doi.org/10.4266/acc.2020.00031
Published online: August 19, 2020
1Department of Surgery, Ajou University School of Medicine, Suwon, Korea
2Department of Surgery, Gangneung Asan Hospital, University of Ulsan College of Medicine, Gangneung, Korea
- Corresponding author Yun Tae Jung Department of Surgery, Gangneung Asan Hospital, University of Ulsan College of Medicine, 38 Bangdong-gil, Gangneung 25440, Korea Tel: +82-33-610-3230 Fax: +82-33-610-3229 E-mail: paysan@gnah.co.kr
- *The first authors contributed equally to this study.
Copyright © 2020 The Korean Society of Critical Care Medicine
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
-
Background
- Postoperative fluid overload may increase the risk of developing pulmonary complications and other adverse outcomes. We evaluated the impact of excessive fluid administration on postoperative outcomes in critically ill patients.
-
Methods
- We reviewed the medical records of 320 patients admitted to intensive care unit (ICU) after emergency abdominal surgery for complicated intra-abdominal infection (cIAI) between January 2013 and December 2018. The fluid balance data of the patients were reviewed for a maximum of 7 days. The patients were grouped based on average daily fluid balance with a cutoff value of 20 ml/kg/day. Propensity score matching was performed to reduce the underlying differences between the groups.
-
Results
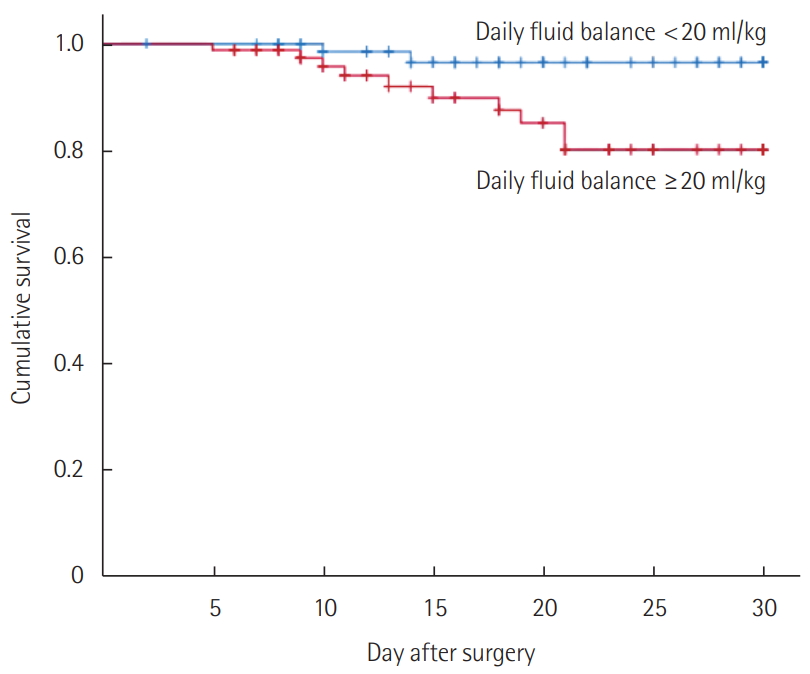
- Patients with an average daily fluid balance of ≥20 ml/kg/day were associated with higher rates of 30-day mortality (11.8% vs. 2.4%; P=0.036) than those with lower fluid balance (<20 ml/kg/day). Kaplan-Meier survival curves for 30-day mortality in these groups also showed a better survival rate in the lower fluid balance group with a statistical significance (P=0.020). The percentage of patients who developed pulmonary consolidation during ICU stay (47.1% vs. 24.7%; P=0.004) was higher in the fluid-overloaded group. Percentages of newly developed pleural effusion (61.2% vs. 57.7%; P=0.755), reintubation (18.8% vs. 10.6%; P=0.194), and infectious complications (55.3% vs. 49.4%; P=0.539) showed no significant differences between the two groups.
-
Conclusions
- Postoperative fluid overload in patients who underwent emergency surgery for cIAI was associated with higher 30-day mortality and more frequent occurrence of pulmonary consolidation. Postoperative fluid balance should be adjusted carefully to avoid adverse clinical outcomes.
INTRODUCTION
MATERIALS AND METHODS
RESULTS
DISCUSSION
KEY MESSAGES
Acknowledgments
-
AUTHOR CONTRIBUTIONS
Conceptualization: YTJ. Data curation: YTJ, JS. Formal analysis: JS. Methodology, YTJ, JS, JYK. Visualization: JS, JYK. Writing–original draft: JS. Writing–review & editing: YTJ, JYK.
NOTES
SUPPLEMENTARY MATERIALS
Supplementary Figure 1.
| Variable |
Daily fluid balance |
P-value | |
|---|---|---|---|
| <20 ml/kg (n=85) | ≥20 ml/kg (n=85) | ||
| Age (yr) | 72 (57–80) | 65 (55–77) | 0.077a |
| Sex (male:female) | 46 (54.12):39 (45.88) | 45 (52.94):40 (47.06) | 1.000 |
| Body weight (kg) | 56.00 (50.00–65.00) | 56.50 (51.00–63.00) | 0.840a |
| Height (m) | 165.0 (155.5–170.0) | 164.0 (155.0–170.0) | 0.572a |
| BMI (kg/m2) | 21.21 (19.54–23.14) | 21.47 (19.33–23.73) | 0.697a |
| CCI (n) | 4.0 (2.0–5.0) | 3.0 (2.0–5.0) | 0.070a |
| Comorbidity | |||
| HTN | 44 (51.76) | 46 (54.12) | 0.878 |
| CAOD | 6 (7.06) | 8 (9.41) | 0.780 |
| DM | 23 (27.06) | 17 (20.0) | 0.366 |
| CRF | 3 (3.53) | 1 (1.18) | 0.747b |
| Malignancy | 12 (14.12) | 17 (20.0) | 0.301 |
| COPD | 6 (6.3) | 4 (4.2) | 0.745b |
| LC | 5 (5.88) | 0 | 0.059b |
| Diagnosis | 0.099 | ||
| Mechanical | 26 (30.59) | 34 (40.00) | |
| Vascular | 11 (12.94) | 13 (15.29) | |
| Ulceration | 31 (36.47) | 32 (37.65) | |
| Infection | 17 (20.0) | 6 (7.06) | |
| Location | 0.656 | ||
| Stomach | 24 (28.24) | 23 (27.06) | |
| Duodenum | 3 (3.53) | 5 (5.88) | |
| Small bowel | 25 (29.41) | 32 (37.65) | |
| Large bowel | 31 (36.47) | 24 (28.24) | |
| Multifocal | 2 (2.35) | 1 (1.18) | |
| Perforation | 69 (81.18) | 68 (80.00) | 1.000 |
| Laparoscopic:open surgery | 7 (8.24):78 (91.76) | 17 (20.0):68 (80.0) | 0.047 |
Values are presented as median (interquartile range) or number (%)
BMI: body mass index; CCI: Charlson comorbidity index; HTN: hypertension; CAOD: coronary artery occlusive disease; DM: diabetes mellitus; CRF: chronic renal failure; COPD: chronic obstructive pulmonary disease; LC: liver cirrhosis.
a Mann-Whitney U-test;
b Fisher’s exact test.
| Variable |
Daily fluid balance |
P-value | |
|---|---|---|---|
| <20 ml/kg (n=85) | ≥20 ml/kg (n=85) | ||
| APACHE II | 12.0 (10.0–17.0) | 12.0 (7.0–16.5) | 0.316a |
| ASA | 0.478b | ||
| 1 | 21 (24.71) | 20 (23.53) | |
| 2 | 47 (55.29) | 42 (49.41) | |
| 3 | 16 (18.82) | 23 (27.06) | |
| 4 | 1 (1.18) | 0 (0.0) | |
| qSOFA | 0.967b | ||
| 0 | 56 (65.88) | 57 (67.06) | |
| 1 | 23 (27.06) | 21 (24.71) | |
| 2 | 5 (5.88) | 6 (7.06) | |
| 3 | 1 (1.18) | 1 (1.18) | |
| fSOFA | 3.0 (2.0–6.0) | 2.0 (1.0–4.0) | 0.086a |
| SIRS | 0.380b | ||
| 0 | 4 (4.71) | 9 (10.59) | |
| 1 | 24 (28.24) | 29 (34.12) | |
| 2 | 37 (43.53) | 27 (31.76) | |
| 3 | 19 (22.35) | 19 (22.35) | |
| 4 | 1 (1.18) | 1 (1.18) | |
| SBP (mm Hg) | 114.0 (100.0–140.0) | 111.0 (100.0–133.0) | 0.381a |
| Respiration rate (n/min) | 16.0 (14.0–18.0) | 16.0 (14.0–18.0) | 0.575a |
| Preoperative shock | 28 (32.94) | 32 (37.65) | 0.630 |
| Preoperative vasopressor use | 15 (17.65) | 10 (11.76) | 0.386 |
Values are presented as median (interquartile range) or number (%).
APACHE: Acute Physiology and Chronic Health Evaluation; ASA: American Society of Anesthesiology; qSOFA: Quick Sequential Organ Failure Assessment; fSOFA: full Sequential Organ Failure Assessment; SIRS: systemic inflammatory response syndrome; SBP: systolic blood pressure.
a Mann-Whitney U-test;
b Fisher’s exact test.
| Variable |
Daily fluid balance |
P-value | |
|---|---|---|---|
| <20 ml/kg (n=85) | ≥20 ml/kg (n=85) | ||
| No. of vasopressors | 0.105a | ||
| 0 | 46 (54.12) | 40 (47.06) | |
| 1 | 23 (27.06) | 32 (37.65) | |
| 2 | 14 (16.47) | 7 (8.24) | |
| 3 | 2 (2.35) | 6 (7.06) | |
| NE day | 2.0 (2.0–3.0) | 4.5 (2.0–7.0) | 0.013b |
| Initial NE dose (μg/kg/min) | 0.10 (0.05–0.14) | 0.11 (0.07–0.21) | 0.252b |
| Highest NE dose (μg/kg/min) | 0.26 (0.12–0.41) | 0.30 (0.13–0.69) | 0.104b |
| RRT | 6 (7.06) | 11 (12.94) | 0.183 |
| Average daily intake (ml) | 3,122.80 (2,742.60–3,793.43) | 4,011.14 (3,358.75–4,766.67) | <0.001b |
| Average daily urine (ml) | 1,877.0 (1,538.86–2,208.20) | 1,625.0 (1,237.86–2,098.14) | 0.060b |
| Average daily fluid balance (ml) | 675.80 (332.20–829.14) | 1,600.50 (1,354.50–1,992.29) | <0.001b |
| Daily fluid balance/bw (ml/kg) | 11.67 (6.20–15.77) | 28.31 (23.02–35.26) | <0.001b |
| Initial fSOFA | 3.0 (2.0–6.0) | 2.0 (1.0–4.0) | 0.086b |
| fSOFA at ICU discharge | 2.0 (1.0–5.0) | 3 (2.0–5.0) | 0.038b |
| Delta SOFA (n/day) | –0.17 (–0.67 to 0.25) | 0.00 (–0.50 to 0.50) | 0.008b |
Values are presented as number (%) or median (interquartile range).
NE: norepinephrine; RRT: renal replacement therapy; bw: body weight; fSOFA: full Sequential Organ Failure Assessment; ICU: intensive care unit; SOFA: Sequential Organ Failure Assessment.
a Fisher’s exact test;
b Mann-Whitney U-test.
| Variable |
Daily fluid balance |
P-value | |
|---|---|---|---|
| <20 ml/kg (n=85) | ≥20 ml/kg (n=85) | ||
| HLOS (day) | 15 (10–25) | 15 (9–28) | 0.731a |
| ICU LOS (day) | 3 (1–8) | 3 (1–10) | 0.459a |
| MV day | 0 (0–2) | 0 (0–5) | 0.472a |
| In-hospital mortality | 7 (8.2) | 11 (12.9) | 0.455 |
| 30-day mortality | 2 (2.4) | 10 (11.8) | 0.036b |
| Variable |
Daily fluid balance |
P-value | |
|---|---|---|---|
| <20 ml/kg (n=85) | ≥20 ml/kg (n=85) | ||
| Pre-existing pleural effusion | 31 (36.5) | 29 (34.1) | 0.872 |
| Newly developed pleural effusion | 49 (57.7) | 52 (61.2) | 0.755 |
| PCD for pleural effusion | 14 (16.5) | 22 (25.9) | 0.189 |
| Pre-existing pulmonary consolidation | 4 (4.7) | 10 (11.8) | 0.163a |
| Newly developed pulmonary consolidation | 21 (24.7) | 40 (47.1) | 0.004 |
| Reintubation | 9 (10.6) | 16 (18.8) | 0.194 |
| Infectious complication | 42 (49.4) | 47 (55.3) | 0.539 |
| Reoperation for anastomosis leakage | 2 (2.4) | 6 (7.1) | 0.277 |
- 1. Balakumar V, Murugan R, Sileanu FE, Palevsky P, Clermont G, Kellum JA. Both positive and negative fluid balance may be associated with reduced long-term survival in the critically ill. Crit Care Med 2017;45:e749-57.ArticlePubMedPMC
- 2. Lee J, de Louw E, Niemi M, Nelson R, Mark RG, Celi LA, et al. Association between fluid balance and survival in critically ill patients. J Intern Med 2015;277:468-77.ArticlePubMed
- 3. Brandstrup B, Tønnesen H, Beier-Holgersen R, Hjortsø E, Ørding H, Lindorff-Larsen K, et al. Effects of intravenous fluid restriction on postoperative complications: comparison of two perioperative fluid regimens: a randomized assessor-blinded multicenter trial. Ann Surg 2003;238:641-8.ArticlePubMedPMC
- 4. Myles PS, Bellomo R, Corcoran T, Forbes A, Peyton P, Story D, et al. Restrictive versus liberal fluid therapy for major abdominal surgery. N Engl J Med 2018;378:2263-74.ArticlePubMed
- 5. Wang N, Jiang L, Zhu B, Wen Y, Xi XM, Beijing Acute Kidney Injury Trial (BAKIT) Workgroup. Fluid balance and mortality in critically ill patients with acute kidney injury: a multicenter prospective epidemiological study. Crit Care 2015;19:371. ArticlePubMedPMC
- 6. Rhodes A, Evans LE, Alhazzani W, Levy MM, Antonelli M, Ferrer R, et al. Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock: 2016. Intensive Care Med 2017;43:304-77.ArticlePubMedPDF
- 7. Zhu AC, Agarwala A, Bao X. Perioperative fluid management in the Enhanced Recovery after Surgery (ERAS) pathway. Clin Colon Rectal Surg 2019;32:114-20.ArticlePubMedPMCPDF
- 8. Sartelli M, Abu-Zidan FM, Catena F, Griffiths EA, Di Saverio S, Coimbra R, et al. Global validation of the WSES Sepsis Severity Score for patients with complicated intra-abdominal infections: a prospective multicentre study (WISS Study). World J Emerg Surg 2015;10:61. ArticlePubMedPMC
- 9. Rivers E, Nguyen B, Havstad S, Ressler J, Muzzin A, Knoblich B, et al. Early goal-directed therapy in the treatment of severe sepsis and septic shock. N Engl J Med 2001;345:1368-77.ArticlePubMed
- 10. Kelm DJ, Perrin JT, Cartin-Ceba R, Gajic O, Schenck L, Kennedy CC. Fluid overload in patients with severe sepsis and septic shock treated with early goal-directed therapy is associated with increased acute need for fluid-related medical interventions and hospital death. Shock 2015;43:68-73.ArticlePubMedPMC
- 11. Barmparas G, Liou D, Lee D, Fierro N, Bloom M, Ley E, et al. Impact of positive fluid balance on critically ill surgical patients: a prospective observational study. J Crit Care 2014;29:936-41.ArticlePubMed
- 12. Shim HJ, Jang JY, Lee SH, Lee JG. The effect of positive balance on the outcomes of critically ill noncardiac postsurgical patients: a retrospective cohort study. J Crit Care 2014;29:43-8.ArticlePubMed
- 13. Mazuski JE, Tessier JM, May AK, Sawyer RG, Nadler EP, Rosengart MR, et al. The Surgical Infection Society Revised Guidelines on the Management of Intra-Abdominal Infection. Surg Infect (Larchmt) 2017;18:1-76.ArticlePubMed
- 14. Sartelli M, Chichom-Mefire A, Labricciosa FM, Hardcastle T, Abu-Zidan FM, Adesunkanmi AK, et al. The management of intra-abdominal infections from a global perspective: 2017 WSES guidelines for management of intra-abdominal infections. World J Emerg Surg 2017;12:29. ArticlePubMedPMCPDF
- 15. McNarry AF, Goldhill DR. Simple bedside assessment of level of consciousness: comparison of two simple assessment scales with the Glasgow Coma scale. Anaesthesia 2004;59:34-7.ArticlePubMed
- 16. Calandra T, Cohen J, International Sepsis Forum Definition of Infection in the ICU Consensus Conference. The international sepsis forum consensus conference on definitions of infection in the intensive care unit. Crit Care Med 2005;33:1538-48.ArticlePubMed
- 17. Levy MM, Evans LE, Rhodes A. The surviving sepsis campaign bundle: 2018 update. Crit Care Med 2018;46:997-1000.ArticlePubMed
- 18. RENAL Replacement Therapy Study Investigators; Bellomo R, Cass A, Cole L, Finfer S, Gallagher M, et al. An observational study fluid balance and patient outcomes in the randomized evaluation of normal vs. augmented level of replacement therapy trial. Crit Care Med 2012;40:1753-60.ArticlePubMed
- 19. Samoni S, Vigo V, Reséndiz LI, Villa G, De Rosa S, Nalesso F, et al. Impact of hyperhydration on the mortality risk in critically ill patients admitted in intensive care units: comparison between bioelectrical impedance vector analysis and cumulative fluid balance recording. Crit Care 2016;20:95. ArticlePubMedPMC
- 20. Silversides JA, Fitzgerald E, Manickavasagam US, Lapinsky SE, Nisenbaum R, Hemmings N, et al. Deresuscitation of patients with iatrogenic fluid overload is associated with reduced mortality in critical illness. Crit Care Med 2018;46:1600-7.ArticlePubMed
- 21. Saugel B, Vincent JL, Wagner JY. Personalized hemodynamic management. Curr Opin Crit Care 2017;23:334-41.ArticlePubMed
- 22. Alvarado Sánchez JI, Amaya Zúñiga WF, Monge García MI. Predictors to intravenous fluid responsiveness. J Intensive Care Med 2018;33:227-40.ArticlePubMed
- 23. Saugel B, Ringmaier S, Holzapfel K, Schuster T, Phillip V, Schmid RM, et al. Physical examination, central venous pressure, and chest radiography for the prediction of transpulmonary thermodilution-derived hemodynamic parameters in critically ill patients: a prospective trial. J Crit Care 2011;26:402-10.ArticlePubMed
- 24. Monnet X, Teboul JL. Passive leg raising: five rules, not a drop of fluid! Crit Care 2015;19:18. ArticlePubMedPMCPDF
- 25. Jones SL, Tanaka A, Eastwood GM, Young H, Peck L, Bellomo R, et al. Bioelectrical impedance vector analysis in critically ill patients: a prospective, clinician-blinded investigation. Crit Care 2015;19:290. ArticlePubMedPMC
- 26. Dewitte A, Carles P, Joannès-Boyau O, Fleureau C, Roze H, Combe C, et al. Bioelectrical impedance spectroscopy to estimate fluid balance in critically ill patients. J Clin Monit Comput 2016;30:227-33.ArticlePubMedPDF
- 27. Murphy CV, Schramm GE, Doherty JA, Reichley RM, Gajic O, Afessa B, et al. The importance of fluid management in acute lung injury secondary to septic shock. Chest 2009;136:102-9.ArticlePubMed
- 28. Cordemans C, De Laet I, Van Regenmortel N, Schoonheydt K, Dits H, Huber W, et al. Fluid management in critically ill patients: the role of extravascular lung water, abdominal hypertension, capillary leak, and fluid balance. Ann Intensive Care 2012;2(Suppl 1 Diagnosis and management of intra-abdominal hyperten):S1. ArticlePubMedPMC
- 29. Hjortrup PB, Haase N, Bundgaard H, Thomsen SL, Winding R, Pettilä V, et al. Restricting volumes of resuscitation fluid in adults with septic shock after initial management: the CLASSIC randomised, parallel-group, multicentre feasibility trial. Intensive Care Med 2016;42:1695-705.ArticlePubMedPDF
- 30. Besen BA, Gobatto AL, Melro LM, Maciel AT, Park M. Fluid and electrolyte overload in critically ill patients: an overview. World J Crit Care Med 2015;4:116-29.ArticlePubMedPMC
- 31. O’Connor ME, Prowle JR. Fluid overload. Crit Care Clin 2015;31:803-21.ArticlePubMed
References
Figure & Data
References
Citations

- Complications during Veno-Venous Extracorporeal Membrane Oxygenation in COVID-19 and Non-COVID-19 Patients with Acute Respiratory Distress Syndrome
Andrea Bruni, Caterina Battaglia, Vincenzo Bosco, Corrado Pelaia, Giuseppe Neri, Eugenio Biamonte, Francesco Manti, Annachiara Mollace, Annalisa Boscolo, Michele Morelli, Paolo Navalesi, Domenico Laganà, Eugenio Garofalo, Federico Longhini
Journal of Clinical Medicine.2024; 13(10): 2871. CrossRef - Fluid balance following laparotomy for hollow viscus perforation: A study of morbidity and mortality
James Tankel, David Chayen, Sharon Einav
Surgery in Practice and Science.2023; 12: 100146. CrossRef - Risk of fluid accumulation after cardiac surgery
Atte Koskinen, Jenni Aittokallio, Jarmo Gunn, Joonas Lehto, Arto Relander, Emma Viikinkoski, Tuija Vasankari, Juho Jalkanen, Maija Hollmén, Tuomas O. Kiviniemi
JTCVS Open.2023; 16: 602. CrossRef - Loop diuretics in adult intensive care patients with fluid overload: a systematic review of randomised clinical trials with meta-analysis and trial sequential analysis
Sine Wichmann, Marija Barbateskovic, Ning Liang, Theis Skovsgaard Itenov, Rasmus Ehrenfried Berthelsen, Jane Lindschou, Anders Perner, Christian Gluud, Morten Heiberg Bestle
Annals of Intensive Care.2022;[Epub] CrossRef - Goal directed fluid removal with furosemide versus placebo in intensive care patients with fluid overload: A trial protocol for a randomised, blinded trial (GODIF trial)
Sine Wichmann, Theis S. Itenov, Rasmus E. Berthelsen, Theis Lange, Anders Perner, Christian Gluud, Pia Lawson‐Smith, Lars Nebrich, Jørgen Wiis, Anne C. Brøchner, Thomas Hildebrandt, Meike T. Behzadi, Kristian Strand, Finn H. Andersen, Thomas Strøm, Mikko
Acta Anaesthesiologica Scandinavica.2022; 66(9): 1138. CrossRef - Lung Ultrasound-Guided Fluid Management versus Standard Care in Surgical ICU Patients: A Randomised Controlled Trial
Daniel-Mihai Rusu, Ioana Grigoraș, Mihaela Blaj, Ianis Siriopol, Adi-Ionut Ciumanghel, Gigel Sandu, Mihai Onofriescu, Olguta Lungu, Adrian Constantin Covic
Diagnostics.2021; 11(8): 1444. CrossRef
- Figure
- Related articles
-
- Association of malnutrition status with 30-day mortality in patients with sepsis using objective nutritional indices: a multicenter retrospective study in South Korea
- Erratum to "Adjuvant intravenous immunoglobulin administration on postoperative critically ill patients with secondary peritonitis: a retrospective study"
- Adjuvant intravenous immunoglobulin administration on postoperative critically ill patients with secondary peritonitis: a retrospective study
- Association between timing of intubation and mortality in patients with idiopathic pulmonary fibrosis
- Association between the National Health Insurance coverage benefit extension policy and clinical outcomes of ventilated patients: a retrospective study

 KSCCM
KSCCM

 PubReader
PubReader ePub Link
ePub Link Cite
Cite



