Articles
- Page Path
- HOME > Acute Crit Care > Volume 36(3); 2021 > Article
-
Original Article
Pulmonary The Mount Sinai Hospital Institute for critical care medicine response to the COVID-19 pandemic -
Jennifer Wang1,2, Evan Leibner1,3
 , Jaime B. Hyman4, Sanam Ahmed1,2, Joshua Hamburger4, Jean Hsieh1,5, Neha Dangayach1,6
, Jaime B. Hyman4, Sanam Ahmed1,2, Joshua Hamburger4, Jean Hsieh1,5, Neha Dangayach1,6 , Pranai Tandon1,5, Umesh Gidwani1,7
, Pranai Tandon1,5, Umesh Gidwani1,7 , Andrew Leibowitz4
, Andrew Leibowitz4 , Roopa Kohli-Seth1,2
, Roopa Kohli-Seth1,2 ,
, -
Acute and Critical Care 2021;36(3):201-207.
DOI: https://doi.org/10.4266/acc.2021.00402
Published online: August 10, 2021
1Institute for Critical Care Medicine, Icahn School of Medicine at Mount Sinai, New York, NY, USA
2Department of Surgery, Icahn School of Medicine at Mount Sinai, New York, NY, USA
3Department of Emergency Medicine, Icahn School of Medicine at Mount Sinai, New York, NY, USA
4Department of Anesthesiology, Perioperative, and Pain Medicine, Icahn School of Medicine at Mount Sinai, New York, NY, USA
5Division of Pulmonary Medicine, Icahn School of Medicine at Mount Sinai, New York, NY, USA
6Department of Neurosurgery, Icahn School of Medicine at Mount Sinai, New York, NY, USA
7Division of Cardiology, Icahn School of Medicine at Mount Sinai, New York, NY, USA
- Corresponding author Evan Leibner Institute for Critical Care Medicine, Mount Sinai Hospital, 1468 Madison Ave, New York, NY 10029, USA Tel: +1-212-241-8867 Fax: +1-212-860-3669 E-mail: evan.leibner@mountsinai.org
Copyright © 2021 The Korean Society of Critical Care Medicine
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
-
Background
- The coronavirus disease 2019 (COVID-19) pandemic resulted in a surge of critically ill patients. This was especially true in New York City. We present a roadmap for hospitals and healthcare systems to prepare for a Pandemic.
-
Methods
- This was a retrospective review of how Mount Sinai Hospital (MSH) was able to rapidly prepare to handle the pandemic. MSH, the largest academic hospital within the Mount Sinai Health System, rapidly expanded the intensive care unit (ICU) bed capacity, including creating new ICU beds, expanded the workforce, and created guidelines.
-
Results
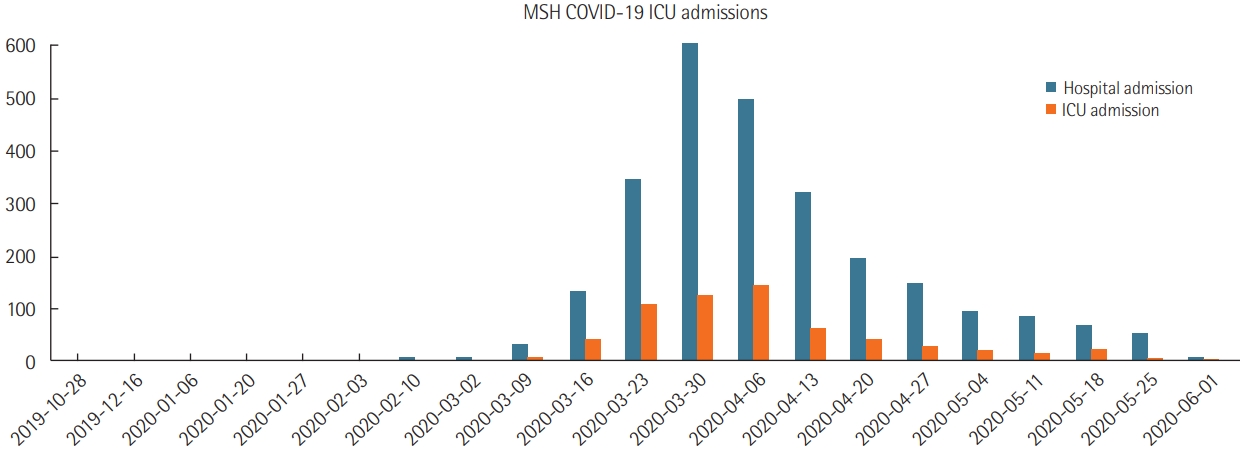
- MSH a 1,139-bed quaternary care academic referral hospital with 104 ICU beds expanded to 1,453 beds (27.5% increase) with 235 ICU beds (126% increase) during the pandemic peak in the first week of April 2020. From March to June 2020, with follow-up through October 2020, MSH admitted 2,591 COVID-19-positive patients, 614 to ICUs. Most admitted patients received noninvasive support including a non-rebreather mask, high flow nasal cannula, and noninvasive positive pressure ventilation. Among ICU patients, 68.4% (n=420) received mechanical ventilation; among the admitted ICU patients, 42.8% (n=263) died, and 47.8% (n=294) were discharged alive.
-
Conclusions
- Flexible bed management initiatives; teamwork across multiple disciplines; and development and implementation of guidelines were critical accommodating the surge of critically ill patients. Non-ICU services and staff were deployed to augment the critical care work force and open new critical care units. This approach to rapidly expand bed availability and staffing across the system helped provide the best care for the patients and saved lives.
INTRODUCTION
MATERIALS AND METHODS
RESULTS
DISCUSSION
KEY MESSAGES
-
CONFLICT OF INTEREST
No potential conflict of interest relevant to this article was reported.
-
AUTHOR CONTRIBUTIONS
Conceptualization: JW, EL, JH (Joshua Hamburger), SA, JH (Jean Hsieh). Data curation: EL, PT. Formal analysis: EL, JW. Methodology: JH (Joshua Hamburger), JH (Jean Hsieh). Project administration: RKS, AL, UG. Visualization: RKS, AL. Writing–original draft: JW, EL, JH, SA, JH, JH. Writing–review & editing: ND, UG.
NOTES
SUPPLEMENTARY MATERIALS
Supplementary Material 1.
Supplementary Material 2.
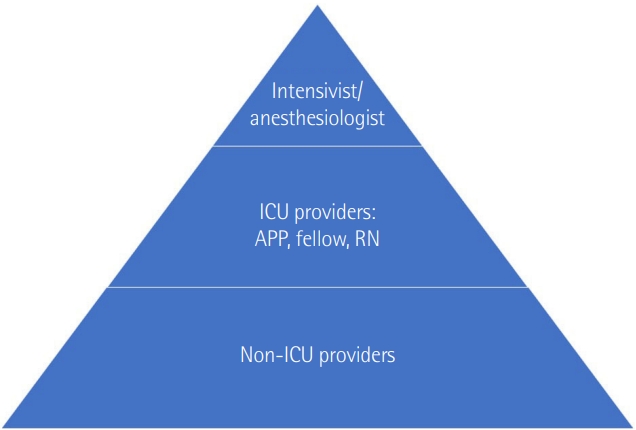
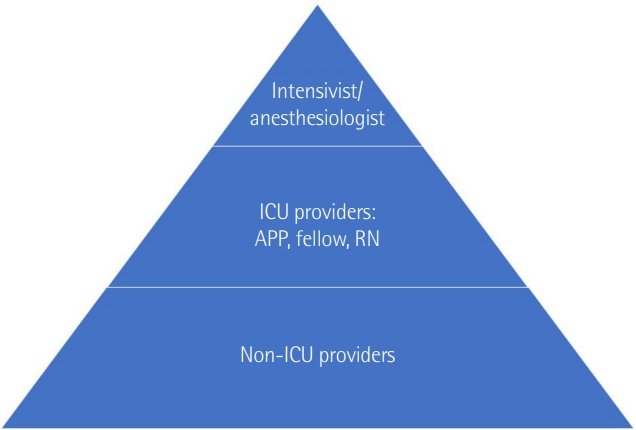
| Unit |
Pre-COVID-19 |
COVID-19 |
||
|---|---|---|---|---|
| Patient population | No. of bedsa | Patient population | No. of bedsa | |
| PACU | ||||
| Main OR PACU | OR Post Op | 30 | MICU/SICU/medicine stepdown (COVID negative) | 30 |
| Annenberg 6 PACU | Ambulatory Post Op | 12 | Transplant critically Ill (COVID negative) | 12 |
| Annenberg 7 PACU | Neurosurgical Post Op | 12 | OR PACU (COVID negative) | 12 |
| ICU | ||||
| MICU | Medical critically ill | 14 | COVID-19 critically ill | 28 |
| SICU | Surgical critically ill | 14 | COVID-19 critically ill | 28 |
| TICU | Transplant critically ill | 12 | COVID-19 critically ill | 24 |
| CICU | Cardiac critically ill | 14 | COVID-19 critically ill | 28 |
| NSICU | Neurosurgical critically ill | 18 | COVID-19 critically ill | 36 |
| CVU | Cardiac surgical Critically ill | 14 | COVID-19 critically ill | 28 |
| CTICU | Cardiac surgical Critically ill | 12 | COVID-19 critically ill | 24 |
| Step down unit | ||||
| Medicine stepdown | Medicine stepdown | 14 | COVID-19 critically ill | 14 |
| Cardiac stepdown | Cardiac stepdown | 6 | COVID-19 critically ill | 12 |
| Surgical stepdown | Surgical stepdown | 14 | COVID-19 critically ill | 14 |
| Medicine overflow | General medicine patients | 16 | Neurosurgical critically ill | 16 |
COVID-19: coronavirus disease 2019; ICU: intensive care unit; PACU: post-anesthesia care unit; OR: operating room; Post Op: postoperative patients; MICU: medical ICU; SICU: surgical ICU; TICU: transplant ICU; CICU: cardiac ICU; NSICU: neuroscience ICU; CVU: cardio vascular; CTICU: cardio thoracic ICU.
a ICU rooms were set up for two patients but varied depending on patient needs and nursing staff.
Values are presented as mean±standard deviation or number (%).
COVID-19: coronavirus disease 2019; ICU: intensive care unit; HTN: hypertension; CKD: chronic kidney disease; COPD: chronic obstructive pulmonary disease; NRB: non-rebreather; HFNC: high-flow nasal cannula; NIPPV: non-invasive positive pressure ventilation.
- 1. Johns Hopkins University. Covid-19 dashboard by the center for systems [Internet]. Baltimore (MD), Johns Hopkins University. 2020 [cited 2020 Apr 17]. Available from: https://www.covidtracker.com/.
- 2. Halpern NA, Pastores SM. Critical care medicine beds, use, occupancy, and costs in the United States: a methodological review. Crit Care Med 2015;43:2452-9.PubMedPMC
- 3. Halpern NA, Tan KS, DeWitt M, Pastores SM. Intensivists in U.S. acute care hospitals. Crit Care Med 2019;47:517-25.ArticlePubMedPMC
- 4. Halpern NA, Tan KS. United States resource availability for COVID-19 [Internet]. Mount Prospect (IL), Society of Critical Care Medicine. 2020 [cited 2020 Apr 17]. Available from: https://sccm.org/getattachment/Blog/March-2020/United-States-Resource-Availability-for-COVID-19/United-States-Resource-Availability-for-COVID-19.pdf?lang=en-US.
- 5. Kumaraiah D, Yip N, Ivascu N, Hill L. Innovative ICU physician care models: Covid-19 pandemic at NewYork-Presbyterian. NEJM Catal Innov Care Deliv 2020;1.
- 6. Farmer JC, Wax RS MD, Baldisseri MR. Preparing your ICU for disaster response [Internet]. Mount Prospect (IL), Society of Critical Care Medicine. 2020 [cited 2020 Apr 17]. Available from: https://www.sccm.org/getattachment/Disaster/PreparingforDisasterResponse.pdf?lang=en-US.
- 7. Halpern NA, Kaplan LJ, Rausen M, Yang JJ. Configuring ICUs in the COVID-19 Era. Mount Prospect (IL), Society of Critical Care Medicine. 2020.
- 8. Scholten EL, Beitler JR, Prisk GK, Malhotra A. Treatment of ARDS with prone positioning. Chest 2017;151:215-24.ArticlePubMed
- 9. Cherian SV, Kumar A, Akasapu K, Ashton RW, Aparnath M, Malhotra A. Salvage therapies for refractory hypoxemia in ARDS. Respir Med 2018;141:150-8.ArticlePubMedPMC
- 10. Kumar P, Kattan O, Broome B, Singhal S. Reassessing Covid-19 needs: how providers can reexamine their surge capacity, supply availability, workforce readiness, and financial resiliency. NEJM Catal Innov Care Deliv 2020;1.
- 11. Kim MK, Rabinowitz LG, Nagula S, Dunn A, Chalil J, Tao Xu, et al. A primer for clinician deployment to the medicine floors from an epicenter of Covid-19. NEJM Catal Innov Care Deliv 2020;1.
- 12. Alhazzani W, Møller MH, Arabi YM, Loeb M, Gong MN, Fan E, et al. Surviving Sepsis Campaign: guidelines on the management of critically ill adults with coronavirus disease 2019 (COVID-19). Intensive Care Med 2020;46:854-87.ArticlePubMedPMC
- 13. Evan S, Leibner M, Elvera L, Baron M, Ronak S, Shah M, et al. Critical care simulation education program during the COVID-19 pandemic. J Patient Saf Forthcoming 2021.
- 14. Christian MD, Wax R, Lazar N. Critical care during a pandemic: final report of the Ontario health plan for an influenza pandemic (OHPIP) working group on adult critical care admission, discharge and triage criteria. Toronto, Ontario, Canada, Toronto, Ontario Health System. 2006.
- 15. Mount Sinai. Faculty and staff education during COVID-19 [Internet]. New York (NY), Mount Sinai. 2020 [cited 2020 May 27]. https://www.mountsinai.org/about/covid19/staff-resources/critical-care-education.
- 16. Caputo KM, Byrick R, Chapman MG, Orser BJ, Orser BA. Intubation of SARS patients: infection and perspectives of healthcare workers. Can J Anaesth 2006;53:122-9.ArticlePubMed
- 17. Peng PW, Ho PL, Hota SS. Outbreak of a new coronavirus: what anaesthetists should know. Br J Anaesth 2020;124:497-501.ArticlePubMed
- 18. Zucco , Liana , Levy N, Ketchandji D, Aziz M, Ramachandran SK. An Update on the Perioperative Considerations for COVID-19 Severe Acute Respiratory Syndrome Coronavirus-2 (SARS-CoV-2) [Internet]. Rochester (MN), Anesthesia Patient Safety Foundation. 2020 [cited 2021 Aug 24]. Available from: https://www.apsf.org/article/an-update-on-the-perioperative-considerations-for-covid-19-severe-acute-respiratory-syndrome-coronavirus-2-sars-cov-2/.Article
- 19. Ranney ML, Griffeth V, Jha AK. Critical supply shortages: the need for ventilators and personal protective equipment during the COVID-19 pandemic. N Engl J Med 2020;382:e41. ArticlePubMed
- 20. Respiratory care committee of Chinese Thoracic Society. Expert consensus on preventing nosocomial transmission during respiratory care for critically ill patients infected by 2019 novel coronavirus pneumonia. Zhonghua Jie He Hu Xi Za Zhi 2020;17:E020.
- 21. Hui DS, Chow BK, Lo T, Tsang OT, Ko FW, Ng SS, et al. Exhaled air dispersion during high-flow nasal cannula therapy versus CPAP via different masks. Eur Respir J 2019;53:1802339. ArticlePubMed
- 22. Apfelbaum JL, Hagberg CA, Caplan RA, Blitt CD, Connis RT, Nickinovich DG, et al. Practice guidelines for management of the difficult airway: an updated report by the American Society of Anesthesiologists Task Force on Management of the Difficult Airway. Anesthesiology 2013;118:251-70.PubMed
- 23. Association of Anaesthetists. Anaesthetic management of patients during a COVID-19 outbreak [Internet]. London, Association of Anaesthetists. 2020 [cited 2020 May 27]. Available from: https://anaesthetists.org/Home/Resources-publications/Anaesthetic-Management-of-Patients-During-a-COVID-19-Outbreak.
References
Figure & Data
References
Citations

- The COVID-19 Tracheostomy Experience at a Large Academic Medical Center in New York during the First Year
Dhruv Patel, Anthony Devivo, Evan Leibner, Atinuke Shittu, Usha Govindarajulu, Pranai Tandon, David Lee, Randall Owen, Gustavo Fernandez-Ranvier, Robert Hiensch, Michael Marin, Roopa Kohli-Seth, Adel Bassily-Marcus
Journal of Clinical Medicine.2024; 13(7): 2130. CrossRef - Why are there so many hospital beds in Germany?
Matthias Brunn, Torsten Kratz, Michael Padget, Marie-Caroline Clément, Marc Smyrl
Health Services Management Research.2023; 36(1): 75. CrossRef - Flow-Sizing Critical Care Resources*
Joseph L. Nates, John M. Oropello, Neeraj Badjatia, Gregory Beilman, Craig M. Coopersmith, Neil A. Halpern, Daniel L. Herr, Judith Jacobi, Roozehra Kahn, Sharon Leung, Nitin Puri, Ayan Sen, Stephen M. Pastores
Critical Care Medicine.2023; 51(11): 1552. CrossRef - Sustaining the Australian respiratory workforce through the COVID‐19 pandemic: a scoping literature review
Emily Stone, Louis B. Irving, Katrina O. Tonga, Bruce Thompson
Internal Medicine Journal.2022; 52(7): 1115. CrossRef - Outcomes of hip fracture surgery during the COVID-19 pandemic
Sherrie Wang, MaKenzie Chambers, Kelsey Martin, Grace Gilbert, Pietro M. Gentile, Rock Hwang, Rakesh Mashru, Kenneth W. Graf, Henry J. Dolch
European Journal of Orthopaedic Surgery & Traumatology.2022; 33(6): 2453. CrossRef - Rapid communication for effective medical resource allocation in the COVID-19 pandemic
Kwangha Lee
Acute and Critical Care.2021; 36(3): 262. CrossRef - Triage: Medical Details and Words Matter
Jolion McGreevy, Rosamond Rhodes
The American Journal of Bioethics.2021; 21(11): 64. CrossRef

 KSCCM
KSCCM
 PubReader
PubReader ePub Link
ePub Link Cite
Cite