Abstract
-
Background
- Although gastric reserve volume (GRV) is a surrogate marker of gastrointestinal dysfunction and feeding intolerance, there is ambiguity in its estimation due to problems associated with its measurement. Introduction of point-of-care ultrasound as a tool for anesthetists kindled interest in its use for GRV estimation.
-
Methods
- In this prospective observational study, we recruited 57 critically ill patients and analyzed 586 samples of GRV obtained by both ultrasonography (USG) and manual aspiration.
-
Results
- The analysis showed that USG-guided GRV was significantly correlated (r=0.788, P<0.001) and in positive agreement with manual aspiration based on Bland-Altman plot, with a mean difference of 8.50±14.84 (95% confidence interval, 7.389–9.798). The upper and lower limits of agreement were 37.7 and –20.5, respectively, within the ±1.96 standard deviation (P<0.001). The respective sensitivity and positive predictive value, specificity and negative predictive value, and area under the curve of USG for feeding intolerance were 66.67%, 98.15%, and 0.82%, with 96.49% diagnostic accuracy.
-
Conclusions
- Ultrasonographic estimation of GRV was positively, significantly correlated and in agreement with the manual aspiration method and estimated feeding intolerance earlier. Routine use of gastric USG could avoid clinical situations where feeding status is unclear and there is high risk of aspiration and could become a standard practice of critical care.
-
Keywords: critical illness; enema; enteral; logistic models; ultrasonic diagnosis; vomiting
INTRODUCTION
The incidence of feed intolerance to enteral nutrition is 31% to 46% in intensive care unit (ICU) patients [1]. The American Society of Parenteral and Enteral Nutrition guidelines recommend daily monitoring of the feed intolerance by observing symptoms of vomiting, flatulence, bowel movement, abdominal distension with pain, and radiological evaluations [2]. In most of the studies these symptoms are non-specific and increased gastric reserve volume (GRV) is still the most frequently occurring proxy marker of gastric intolerance (GI) [3]. Hence manual aspiration technique despite its shortcomings is still the frequently utilized technique by the ICU nursing staff (97%) the world over to decide the feeding practice to be followed in the ICU setup [1-4] Thus, we did this study to see the diagnostic accuracy of ultrasonographic (USG) guided over manual aspiration technique for evaluation of GRV in critically ill patients in our ICU.
MATERIALS AND METHODS
Research Ethics
The study was performed according to the Helsinki Declaration and approved by the Research Ethics Committee or the Institutional Review Board of Indra Gandhi Medical College as well as Clinical Trials Registry of India (CTRI/2020/10/028261). The study was conducted between October 2020 and September 2021 on 57 adult critically ill patients. Informed consent was taken from patients' attendants or patients as applicable.
Patients between 18 and 80 years of either sex who were enterally fed, not on any prokinetics for feed intolerance and with an anticipated ICU stay of at least 5 days were enrolled in the study. Patients having bowel surgery within 24 hours, GI bleed, obstruction, perforation, malabsorption syndrome, abnormal liver function tests; serum glutamic-oxaloacetic transaminase/serum glutamic pyruvic transaminase more than three times normal and/or total bilirubin more than three times normal and morbid obesity/pregnancy (unable to achieve right lateral position) were excluded from the study.
Patients meeting the criteria were identified and written informed consent was taken from the attendant. A brief history was taken regarding the comorbidities like hypertension, type 2 diabetes mellitus, acute kidney injury, and hypothyroidism. The patient’s age, weight, sex, and baseline vitals were also noted. All patients were subjected to bedside ultrasonography for measurement of GRV followed by manual aspiration for the same measurement twice a day in the morning and evening prior to feeding with last feed at least 4 hours prior to estimation of GRV.
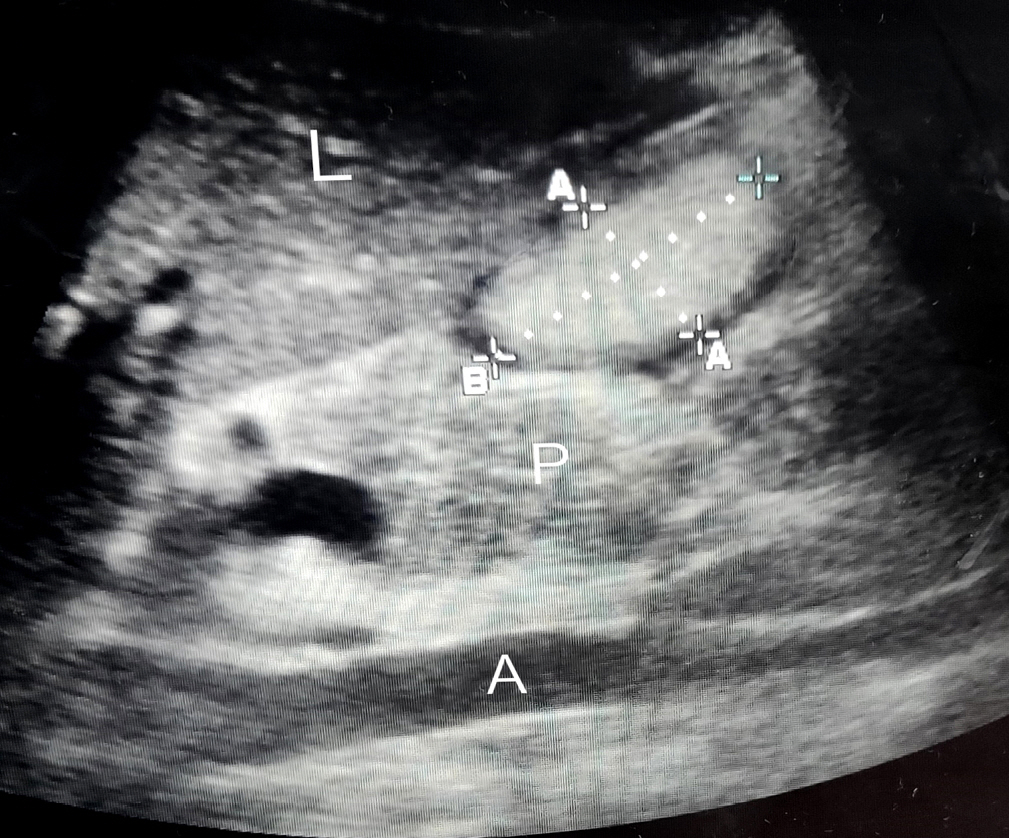
A curved array, low frequency (2–5 MHz) probe of portable Sonosite ultrasound machine was used in abdominal scan mode settings. For the initial few USG readings, the help of a radiologist was taken. Scanning of the epigastrium in a sagittal plane was done while moving the transducer from the left to right subcostal margins. The gastric antrum was thus identified just below the left lobe of the liver and pancreas with the aorta/superior mesenteric artery acting as an important landmark, a still good image of the antrum was taken between peristalsis. Cross‑sectional area (CSA) was measured using the free‑hand tracing tool built into the ultrasound machine. Gastric volume was calculated using the formula described by Perlas et al. [5] (Figure 1).
USG gastric volume (ml)=27.0+14.6×right‑lateral CSA−1.28×age.
Manual aspiration was done by a 50 ml syringe through a 14-G enteral tube and the contents were kept in a sterile container and were reposed back. The study was continued every day and prokinetics were added guided by either of these measurements. If a single GRV ≥150 ml was measured by these techniques, levosulpiride 25 mg QID (4 times a day) was started. If GRV ≥250 ml was obtained, next feed was omitted. Effects of the prokinetic drug were assessed for the next 2 days and after that study stopped in that patient as it would change the GRV volume. Bowel movements were recorded along with any episodes of diarrhea, constipation, vomiting, stomach pain, and increased flatulence. The use of agents like noradrenaline, dopamine, opioids, and enema were not controlled and were recorded. In our institution, the enteral feed is given in a bolus technique over 10–15 minutes and the same was given to our patients and the bed was elevated at the head end to 30°–40° at the time of giving bolus feed. The patients were subjected to chlorhexidine mouth wash in morning and evening.
Power of the study was done on the basis of the study by Perlas et al. [5], with a correlation of r=0.86, power of 90%, and significance of P<0.05, thus sample size was 36 but we recruited 69 patients in the study duration, and 12 patients were excluded in total with 4 patients expired, Ryles tube removed in 2 and 6 were, discharged from ICU before 5 days thus 57 patients with 586 measurements over the days were finally analyzed.
The presentation of the Categorical variables was done in the form of number and percentage (%). On the other hand, the quantitative data were presented as the mean± standard deviation (SD) and as median with 25th and 75th percentiles (interquartile range) and were analyzed using Paired t-test. The sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV) of the USG method were calculated for predicting feed intolerance, Pearson correlation coefficient and Bland-Altman plot was used to see the correlation and agreement between the two methods. The data entry was done in the Microsoft Excel spreadsheet and the final analysis was done with the use of IBM SPSS ver. 21.0 (IBM Corp.). For statistical significance, P-value of less than 0.05 was considered statistically significant.
RESULTS
In our study out of 57 patients 25 (43.86%) were females and 32 (56.14%) were males with a mean age of 50.49±14.1 years and mean weight of 60.67±9.15 kg (Table 1). Most patients in our study were undergoing neurosurgical procedures, i.e., 46 patients (80.70%), 10 patients (17.54%) were being treated for medical disease, and 1 patient (1.75%) was undergoing general surgery. Patients who had increased GRV were from neurosurgery (three patients), and one patient with organophosphorus poisoning was diagnosed with USG estimation only. Patients were having various comorbidities like hypertension (19.30%), diabetes mellitus (7.02%), acute kidney injury (1.75%), single kidney (1.75%), and hypothyroidism (1.75%). None of the patients with these comorbidities had increased GRV during the study period.
The baseline hemodynamic parameters were recorded at admission and were comparable (P>0.05). Forty-five patients (78.95%) were intubated and thus mechanically ventilated and 12 (21.05%) were on non-invasive ventilation or on a venturi mask. All four patients (8.80%) in our study who were diagnosed with increased GRV were mechanically ventilated. Mean Glasgow Coma Scale was 6.7±3.26 and was 5.70 in four patients with increased GRV (Table 1).
We noted other features of feed intolerance and nine patients (15.79%) had vomiting, four (7.02%) had episodes of stomach ache, 15 (26.32%) had constipation and four (7.02%) had episodes of increased flatulence. Out of four patients of increased GRV vomiting, constipation, and flatulence were seen in three, two, and two patients, respectively. Thirty patients (52.63%) received noradrenaline and three patients (5.26%) received both dopamine and noradrenaline for hypotension. Only three out of 30 patients (10%) of the patients on noradrenaline and none of the patients on dual inotropes had increased GRV. Forty-four patients (77.19%) were given opioids while 13 (22.81%) were given non-opioid–based sedation in ICU. Only two out of these 44 patients (4.50%) on opioid sedation had increased GRV in our study (Table 1).
Levosulpiride was used in four patients during the study period, and we observed a mean 87.02%±4.55% (range, 81.25–92.00) fall in GRV after its use. USG imaging was difficult in 21.05% (12 patients) while the resolution was optimal in rest 45 patients (78.95%). Total of 586 measurements of gastric aspirates taken over the days by USG and manual aspiration technique were recorded and the following observations were withdrawn (Table 2).
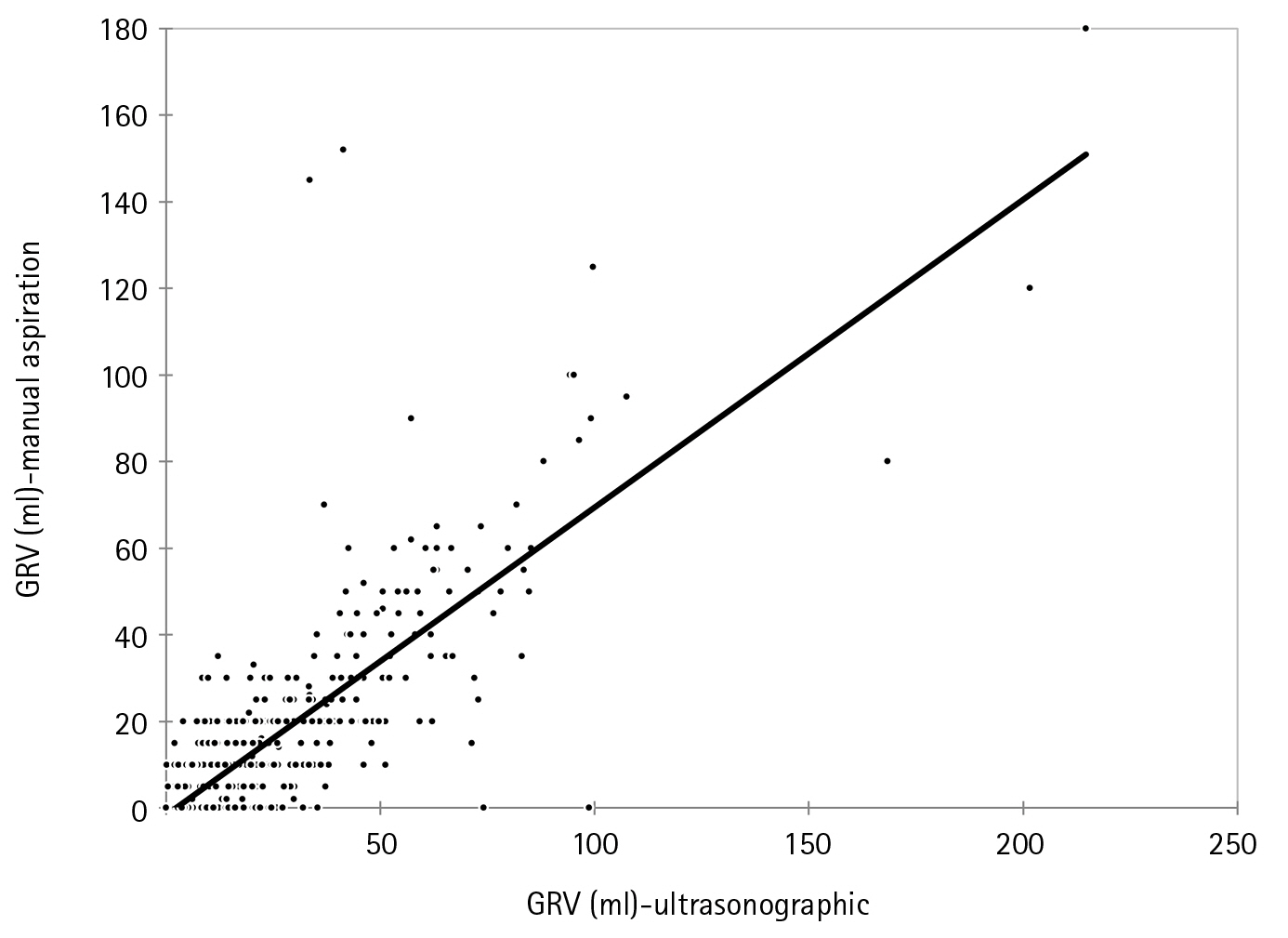
We got significantly higher values of GRV (8.60±14.85 ml arithmetic mean) with USG as compared to the manual aspiration in our study with no diurnal variation (P<0.001). USG-guided GRV was significantly correlated with manual technique by Pearson correlation coefficient, with the strength of association of r=0.788 (P<0.001) (Figure 2).
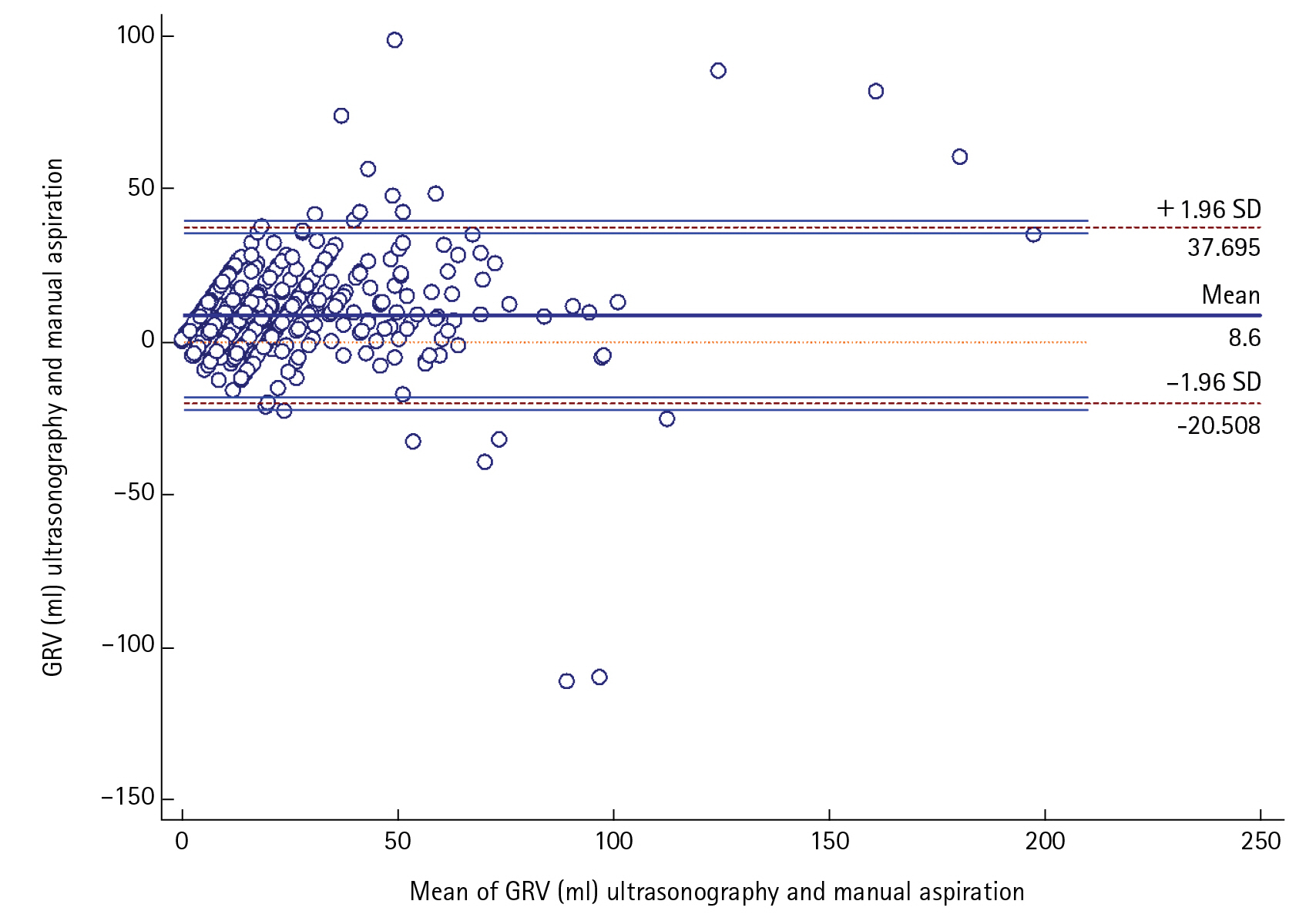
We further confirmed the positive agreement between the two methods by Bland-Altman plot using the data of all 586 times the measurements of GRV done over the days. The mean average of difference thus obtained was 8.50±14.84 (95% confidence interval [CI], 7.389–9.798) The upper and lower limit of the agreement are 37.7 and –20.5 which too lie within the±1.96SD (P<0.001) (Figure 3). As manual aspiration is the gold standard technique for evaluating GRV, the incidence of feed intolerance included only patients with the manual aspiration technique. Thus, 5.26% (three patients) had feed intolerance in our study by this method. When using USG this percentage would have been 7% (four patients).
The sensitivity and PPV, specificity and NPV, area under the curve (AUC; 95% CI) of the USG for finding out the feed intolerance was (66.67%, 98.15%, 0.82%) and diagnostic accuracy of USG for detecting feed intolerance was 96.49%. We categorized the GRV obtained into two groups of >0.8 ml/kg labeled as risk stomach and ≤0.8 ml/kg. The sensitivity, PPV, specificity, NPV, and AUC of the USG for ≤0.8 ml/kg stomach were 89.66%, 45.22%, 76.58%, 97.17%, and 0.83 with a diagnostic accuracy of 78.90%. It was (75% and 33.33%, 92.28% and 98.63%, 0.84%) with diagnostic accuracy of 91.44% for USG method in detecting >0.8 ml/kg stomach (Table 3).
DISCUSSION
Despite all the controversies regarding the estimation of GRV, it is still considered a definitive marker of gastrointestinal dysfunction and feeding intolerance followed by other features like vomiting, diarrhea, stomach ache, and increased flatulence [3,4]. Although The NUTRIREA-2 trials (Impact of not measuring GRV on nosocomial pneumonia rates) have discredited the role of GRV estimation and have laid more stress on parenteral nutrition over the enteral route, but as the surgical patients included in these trials were less, hence the results cannot be generalized to predominantly surgical ICU like ours. They have reported high incidence of vomiting 39.6% over 27% in patients where GRV was not measured but their primary outcome was overall patients’ morbidity and caloric intake over the days [6,7].
When we searched the literature for various methods used for GRV estimation, we observed that people used manual aspiration, scintigraphy, acetaminophen absorption assay method, George method in which dye refractometry was used, gastric impedance monitoring various radiological methods like computed tomography (CT) scan [8,9]. Most of these methods are not validated and expensive hence cannot be done daily in patients to find the gastric volume to decide the next feed. So, the thrust lies on the manual technique which is time-consuming, gives lower values in single aspiration over continuous pressure aspiration [10], and leads to loss of gastric juices as aspirates are frequently discarded by the nurses and frequent aspirations lead to clogging of the NG tube. Parenteral nutrition is not a viable method in third-world countries due to the cost factor hence the focus again points to the optimization of enteral nutrition and the justifiable use of prokinetic therapy guided by its estimation.
Perlas et al. [5] is credited with assessing the feasibility of USG for measuring gastric contents in 18 healthy volunteers in phase one and 36 volunteers in phase two trial in 2009 and found good correlation between CSA lateral and volume (rho=0.731, P<0.001) for predicted volumes of 0–300 ml only. Gastric sonography could be a bedside diagnostic tool to measure GRV, but its routine use has been limited by its inclusion mostly in the domain of the radiologist.
Sharma et al. [11] did a preoperative assessment of CSA in both supine and lateral positions and inferred that the recorded gastric volume was significantly more in right lateral position (P<0.001). Hence, we also took measurements in this position in our patients. Hamada et al. [12] compared USG with CT-guided gastric volume estimations and concluded that the results were positively correlating when performed in good conditions (65% [r=0.43]) and there was a clinically acceptable agreement between these two measurements (bias of –0.12 cm2) and limit of agreement of (–2.21 to –1.96). They found that there was good reproducibility of volumes by ICU physicians over radiologist produced data in CT scan, but all measurements which were obtained out of agreement were where USG was done with poor window.
Sharma et al. [13] studied the gastric contents in the supine position with the aorta and IVC as a guideline in 30 critically ill patients. Gastric antrum CSA using aorta as landmark correlated with aspirated volume (R2=0.86, P<0.001) and a craniocaudal diameter of gastric antrum of <10 cm predicted a volume of <500 ml and <5 cm ml predicted GRV of <150 ml.
In our study, we obtained a statistically significant positive linear relation between the two techniques with the strength of association of r=0.788 (P<0.001). They were in positive agreement as seen by Bland-Altman plot. The sensitivity and PPV were 66.67% and were low in our study as the number of patients taken was small (57 patients) and the prevalence of the diseases was also low in our study as we studied the patients on admission with study ending on 5 days of admission in ICU and researcher believe that the median time for beginning of feed intolerance is 3rd day of ICU admission [3]. The specificity and NPV were high (98.1%) thus the USG could accurately diagnose the feed intolerance in all patients in our study who were diagnosed by manual aspiration technique.
In our study, we did not find an association between other known features of GI with increased GRV values. Six patients (11%) had vomiting, two (3.7%) had an increased history of flatulence and stomach ache, 27 (51%) were on noradrenaline drip, three (5.26%) were on dual inotropes, 42 (79%) on opioid sedation, and 13 (24%) received enema without increased GRV in our study. Hamada et al. [12] also found no association between vasopressor use and increased GRV in his patients. Like our study, Sharma et al. [13] encountered difficulty in 21% of patients, Hamada et al. [12] could find good USG conditions in 65% of their patients after 4 hours of training in USG of ICU physicians and Sharma et al. [11] encountered some difficulty initially but once learned they, and Perlas et al. [5] found it was an easy method in all patients.
The study had some limitations. Firstly, our study was conducted on critically ill patients receiving enteral nutrition so study findings can partially be applied to other subject populations (e.g., healthy adults, children, morbidly obese, and patients coming for emergency surgeries. Secondly, we did not study ventilator-associated pneumonia and mortality indicators of the included patients. Finally, there was a small learning curve of ultrasonography which has to be mastered initially in the presence of a radiologist followed by individual practice. So, with practice, one learns to obtain an appropriate soft tissue window to obtain images of the gastric antrum.
Bedside gastric ultrasonography can provide accurate information regarding GRV and feed intolerance as USG estimations were positively, significantly correlated and was in agreement with manual aspiration method of GRV calculation. USG-guided enteral nutrition and prokinetic use can curb its incessant use and resultant tachyphylaxis in ICU.
KEY MESSAGES
▪ Ultrasonographic (USG) estimation of gastric reserve volume is significantly correlated with the manual aspiration technique and can estimate feeding intolerance earlier.
▪ Use if USG could avoid clinical situations where feeding status is unclear and there is a high risk of aspiration.
▪ Our USG estimation study comprised of large 586 samples could be included in American Society of Parenteral and Enteral Nutrition guidelines for feeding intolerance.
NOTES
-
CONFLICT OF INTEREST
No potential conflict of interest relevant to this article was reported.
-
FUNDING
None.
-
AUTHOR CONTRIBUTIONS
Conceptualization: JP, RKD. Data curation: RS. Formal analysis: RKD. Methodology: JP, RKD, RS. Project administration: JP. Visualization: JP, RKD, RS. Writing–original draft: RS, JP. Writing–review & editing: JP, RS, RKD.
Acknowledgments
This article was part of thesis submitted for grant of MD Anesthesia degree.
Figure 1.Ultrasonogram of the gastric antrum. L: liver; A: aorta; P: pancreas; A–A: a gastric antrum anteroposterior diameter; B: craniocaudal diameter.
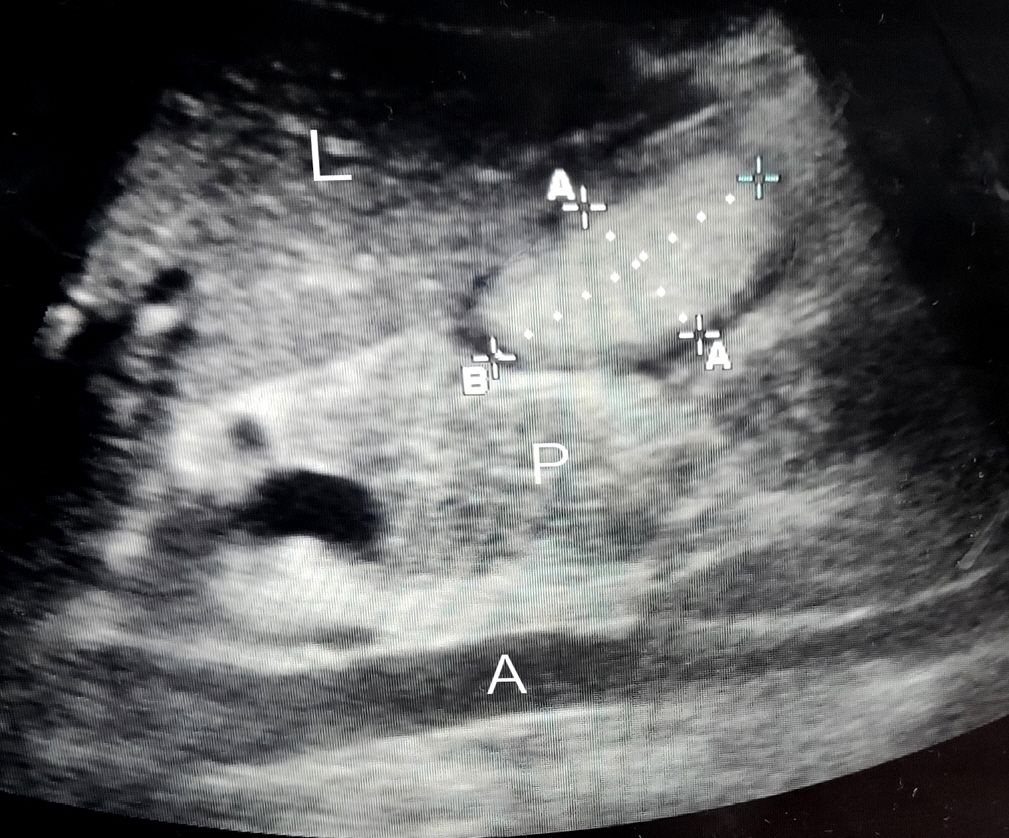
Figure 2.Correlation of gastric reserve volume (GRV, ml) between ultrasonography and manual aspiration. Pearson’s correlation coefficient: r=0.788 (P<0.001).
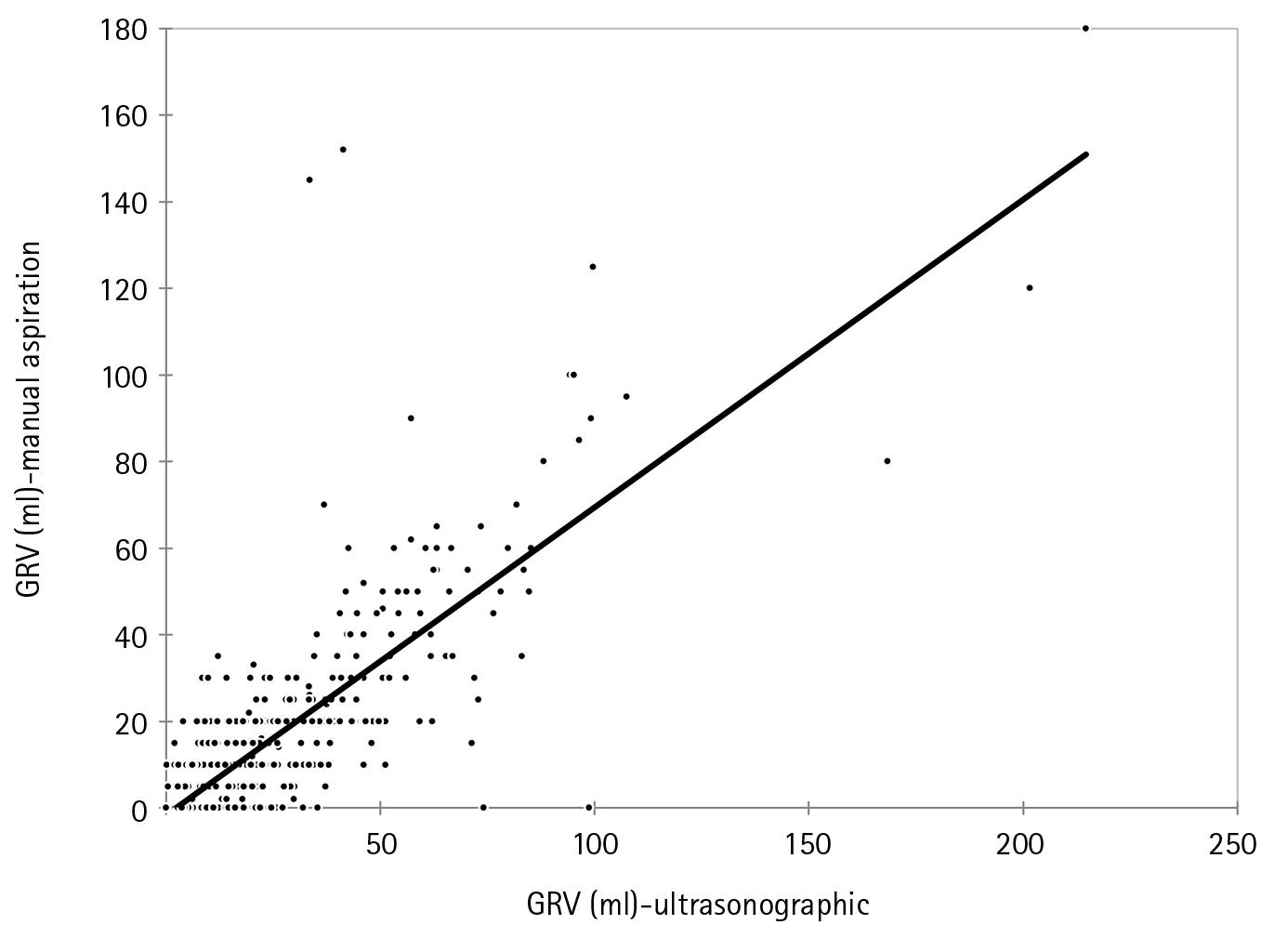
Figure 3.Bland-Altman plot of gastric reserve volume (GRV) measured by ultrasonography compared to manual aspiration. Sample size: 586; P (H0: mean=0) <0.001; 95% confidence interval [CI], 7.389–9.798; arithmetic mean, 8.6; standard deviation, 14.85; lower limit, –20.508 (95% CI, –2.568 to –18.448); upper limit, 37.695 (95% CI, 35.635 to 39.755).

Table 1.Demographic factors and other symptoms that could affect gastric reserve volume estimation
|
Parameter |
Total patients (n=57) |
Normal GRV (n=53) |
Increased GRV (n=4) |
|
Age (yr) |
50.50±14.10 |
50.27±11.89 |
51.25±13.07 |
|
Weight (kg) |
60.67±9.15 |
60.37±7.98 |
64.50±6.65 |
|
Female |
25 (43.86) |
23 (92.00) |
2 (8.00) |
|
Male |
32 (56.14) |
30 (93.75) |
2 (6.25) |
|
Glasgow coma scale |
6.70±3.26 |
- |
5.70 |
|
Mechanical ventilation |
45 (78.95) |
41 (71.92) |
4 (7.01) |
|
Vomiting |
9 (15.79) |
6 (10.50) |
3 (5.20) |
|
Flatulence |
4 (7.02) |
2 (3.50) |
2 (3.50) |
|
Stomach ache |
4 (7.02) |
2 (3.50) |
2 (3.50) |
|
Enema |
15 (26.32) |
13 (22.80) |
2 (3.50) |
|
Vasopressors or adrenaline |
30 (52.63) |
27 (47.30) |
3 (10.00) |
|
Dual inotropes |
3 (5.26) |
3 (5.26) |
- |
|
Opioid sedation |
44 (77.19) |
42 (73.68) |
2 (3.50) |
Table 2.Comparison of mean gastric reserve volume (ml) and average gastric residual volume (mean/kg body weight) measured by USG and manual aspiration over the study period
|
Gastric reserve volume (ml) |
USG |
Manual aspiration |
P-value |
|
Day 1 (n=57) |
|
|
<0.001 |
|
Morning |
31.98±24.32 |
20.65±18.28 |
|
|
Evening |
24.54±18.79 |
16.89±14.55 |
|
Average |
0.47±0.30 |
0.30±0.24 |
|
Day 2 (57) |
|
|
<0.001 |
|
Morning |
27.72±22.13 |
20.93±23.21 |
|
|
Evening |
26.39±31.07 |
18.33±27.46 |
|
Average |
0.44±0.38 |
0.31±0.37 |
|
Day 3 (n=57) |
|
|
<0.001 |
|
Morning |
24.99±21.06 |
17.44±25.95 |
|
|
Evening |
25.09±16.51 |
16.88±23.99 |
|
Average |
0.41±0.29 |
0.27±0.36 |
|
Day 4 (n=57) |
|
|
<0.001 |
|
Morning |
29.81±32.88 |
15.88±23.58 |
|
|
Evening |
23.94±24.65 |
11.32±15.66 |
|
Average |
0.45±0.47 |
0.22±0.32 |
|
Day 5 (n=56) |
|
|
<0.001 |
|
Morning |
21.45±20.18 |
11.11±17.42 |
|
|
Evening |
15.52±15.04 |
9.11±14.37 |
|
Average |
0.30±0.27 |
0.18±0.32 |
|
Day 6 (n=26) |
|
|
0.002 |
|
Morning |
21.30±39.77 |
12.65±29.05 |
|
|
Evening |
13.27±10.85 |
8.04±12.97 |
|
Average |
0.28±0.33 |
0.16±0.28 |
|
Day 7 (n=11) |
|
|
0.017 |
|
Morning |
13.99±8.78 |
11.55±6.73 |
|
|
Evening |
12.97±7.05 |
7.78±5.65 |
|
Average |
0.20±0.08 |
0.14±0.08 |
|
Day 8 (n=3) |
|
|
0.669 |
|
Morning |
8.69±1.31 |
11.67±2.89 |
|
|
Evening |
8.36±2.24 |
8.33±7.64 |
|
Average |
0.14±0.02 |
0.16±0.08 |
|
Day 9 (n=2) |
|
|
0.251 |
|
Morning |
7.85±6.29 |
0±0 |
|
|
Evening |
10.15±12.80 |
2.50±3.54 |
|
Average |
0.14±0.04 |
0.02±0.03 |
|
Day 10 (n=1) |
|
|
- |
|
Morning |
50.4±0 |
90±0 |
|
|
Evening |
55.2±0 |
60±0 |
|
Average |
0.81±0 |
1.15±0 |
Table 3.Sensitivity, specificity, positive predictive value, and negative predictive value of average GRV of ultrasonography for predicting feeding intolerance
|
Ultrasonographic vs. manual aspiration |
Sensitivity (95% CI), % |
Specificity (95% CI), % |
AUC (95% CI), % |
Positive predictive value (95% CI), % |
Negative predictive value (95% CI), % |
Diagnostic accuracy, % |
|
Feeding intolerance |
66.67 (9.43–99.16) |
98.15 (90.11–99.95) |
0.82 (0.70–0.91) |
66.67 (9.43–99.16) |
98.15 (90.11–99.95) |
96.49 |
|
Average GRV (≤0.8 ml/kg) |
89.66 (78.83–96.11) |
76.58 (71.06–81.51) |
0.83 (0.79–0.87) |
45.22 (35.92–54.77) |
97.17 (93.94–98.95) |
78.90 |
|
Average GRV (>0.8 ml/kg) |
75.00 (47.62–92.73) |
92.28 (88.74–94.99) |
0.84 (0.79–0.87) |
33.33 (18.56–50.97) |
98.63 (96.52–99.62) |
91.44 |
References
- 1. MacLaren R, Kiser TH, Fish DN, Wischmeyer PE. Erythromycin vs metoclopramide for facilitating gastric emptying and tolerance to intragastric nutrition in critically ill patients. JPEN J Parenter Enteral Nutr 2008;32:412-9.ArticlePubMedPDF
- 2. McClave SA, Taylor BE, Martindale RG, Warren MM, Johnson DR, Braunschweig C, et al. Guidelines for the provision and assessment of nutrition support therapy in the adult critically ill patient: Society of Critical Care Medicine (SCCM) and American Society for Parenteral and Enteral Nutrition (A.S.P.E.N.). JPEN J Parenter Enteral Nutr 2016;40:159-211.ArticlePubMedPDF
- 3. Gungabissoon U, Hacquoil K, Bains C, Irizarry M, Dukes G, Williamson R, et al. Prevalence, risk factors, clinical consequences, and treatment of enteral feed intolerance during critical illness. JPEN J Parenter Enteral Nutr 2015;39:441-8.ArticlePubMedPDF
- 4. Elke G, Felbinger TW, Heyland DK. Gastric residual volume in critically ill patients: a dead marker or still alive? Nutr Clin Pract 2015;30:59-71.ArticlePubMedPDF
- 5. Perlas A, Chan VW, Lupu CM, Mitsakakis N, Hanbidge A. Ultrasound assessment of gastric content and volume. Anesthesiology 2009;111:82-9.ArticlePubMedPDF
- 6. Reignier J, Mercier E, Le Gouge A, Boulain T, Desachy A, Bellec F, et al. Effect of not monitoring residual gastric volume on risk of ventilator-associated pneumonia in adults receiving mechanical ventilation and early enteral feeding: a randomized controlled trial. JAMA 2013;309:249-56.ArticlePubMed
- 7. Reignier J, Boisramé-Helms J, Brisard L, Lascarrou JB, Ait Hssain A, Anguel N, et al. Enteral versus parenteral early nutrition in ventilated adults with shock: a randomised, controlled, multicentre, open-label, parallel-group study (NUTRIREA-2). Lancet 2018;391:133-43.PubMed
- 8. Beckers EJ, Rehrer NJ, Brouns F, Ten Hoor F, Saris WH. Determination of total gastric volume, gastric secretion and residual meal using the double sampling technique of George. Gut 1988;29:1725-9.ArticlePubMedPMC
- 9. Chang WK, McClave SA, Lee MS, Chao YC. Monitoring bolus nasogastric tube feeding by the Brix value determination and residual volume measurement of gastric contents. JPEN J Parenter Enteral Nutr 2004;28:105-12.ArticlePubMed
- 10. Bartlett Ellis RJ, Fuehne J. Examination of accuracy in the assessment of gastric residual volume: a simulated, controlled study. JPEN J Parenter Enteral Nutr 2015;39:434-40.PubMed
- 11. Sharma G, Jacob R, Mahankali S, Ravindra MN. Preoperative assessment of gastric contents and volume using bedside ultrasound in adult patients: a prospective, observational, correlation study. Indian J Anaesth 2018;62:753-8.ArticlePubMedPMC
- 12. Hamada SR, Garcon P, Ronot M, Kerever S, Paugam-Burtz C, Mantz J. Ultrasound assessment of gastric volume in critically ill patients. Intensive Care Med 2014;40:965-72.ArticlePubMedPDF
- 13. Sharma V, Gudivada D, Gueret R, Bailitz J. Ultrasound-assessed gastric antral area correlates with aspirated tube feed volume in enterally fed critically ill patients. Nutr Clin Pract 2017;32:206-11.ArticlePubMedPDF
Citations
Citations to this article as recorded by

- Correlación de volumen residual gástrico medido y ultrasonido gástrico para identificar intolerancia gástrica en pacientes neurocríticos con nutrición enteral en la unidad de cuidados intensivos
Mauricio Pérez-Macías, Roxana Vázquez-Ramírez, Marcela Pinedo-Lechuga
Medicina Crítica.2024; 38(2): 102. CrossRef - Gastrointestinal function in critically ill patients
Annika Reintam Blaser, Kaspar F. Bachmann, Adam M. Deane
Current Opinion in Clinical Nutrition & Metabolic Care.2023; 26(5): 463. CrossRef
 , Ravi Kant Dogra2
, Ravi Kant Dogra2 , Jyoti Pathania1
, Jyoti Pathania1 , Arti Sharma1
, Arti Sharma1







 KSCCM
KSCCM
 PubReader
PubReader ePub Link
ePub Link Cite
Cite